https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7687409/
What can we learn from brain autopsies in COVID-19?
Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the causative agent of coronavirus disease 2019 (COVID-19) for which there have been over 50 million confirmed cases and 1.2 million deaths globally. While many SARS-CoV-2 infected individuals are asymptomatic or experience respiratory symptoms, extrapulmonary manifestations, including neurological symptoms and conditions, are increasingly recognized. There remains no clear understanding of the mechanisms that underlie neurological symptoms in COVID-19 and whether SARS-CoV-2 has the potential for neuroinvasion in humans. In this minireview, we discuss what is known from human autopsies in fatal COVID-19, including highlighting studies that investigate for the presence of SARS-CoV-2 in brain and olfactory tissue, and summarize the neuropathological consequences of infection. Incorporating microscopic and molecular findings from brain tissue into what we know about clinical disease will inform best practice management guidance and direct research priorities as it relates to neurological morbidity from COVID-19.
1. Manuscript
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), an enveloped, single-stranded positive-sense RNA betacoronavirus, is the causative agent of coronavirus disease 2019 (COVID-19), for which there have been over 50 million confirmed cases and 1.2 millions deaths worldwide as of November 8, 2020 [1,2]. Morbidity and mortality are more common in older individuals and those with comorbidities, including cardiovascular disease, hypertension, obesity, and diabetes, although young people with no comorbidities are also at risk for critical illness [[3], [4], [5]]. While many SARS-CoV-2 infected individuals are asymptomatic or experience predominantly respiratory symptoms, extrapulmonary manifestations, including neurological symptoms and conditions, are increasingly recognized [[6], [7], [8]]. The majority of current studies on neurological manifestations are case reports or retrospective series focused on hospitalized patients through the extraction of medical record data, which have described disorders of consciousness, delirium, and neuromuscular and cerebrovascular complications [[7], [8], [9], [10]]. Smell and taste disturbances in the absence of nasal obstruction are particularly characteristic of COVID-19, leading to speculation regarding the olfactory nerve as a possible route of central nervous system entry [11,12]. Other neurological findings include headache, myalgia, rhabdomyolysis, Guillain-Barre syndrome, encephalopathy, and myelopathy with rare cases of encephalitis based on imaging or cerebrospinal fluid [8,[13], [14], [15], [16], [17], [18]]. SARS-CoV-2 has not been detected in cerebrospinal fluid in the majority of patients tested [8,19], highlighting the need for studies of autopsy brain tissue to understand COVID-19 neuropathogenesis and develop neurocognitive preserving treatment strategies.
Autopsies provide a wealth of information about the decedents, regardless of whether a likely cause of death was identified pre-mortem [20,21]. Due to initial uncertainties regarding the infectious properties of SARS-CoV-2 and limitations in personnel and personal protective equipment availability, autopsies for COVID-19 patients have been limited, although an increasing number of studies are now being published (reviewed in [[22], [23], [24]]). Reports of detailed neuropathological examinations have lagged behind general autopsy series, in part due to the initial focus on lung pathology combined with the longer (2–3 weeks) formalin fixation time preferred by most neuropathologists before cutting brains. Additional factors include the reluctance of some institutions to perform brain removal in COVID-19 cases due to concerns over electric bone saw generated aerosols, which can be effectively contained through the use of vacuum filters or hand saws [25,26]. Included in this review are peer-reviewed studies of autopsy findings published in English between January 1, 2020, and November 5, 2020. Two different databases (PubMed, Google Scholar) were searched for key terms, including COVID-19, nCoV-2019, and SARS-CoV-2, crossed with autopsy, histology, histopathology, neuropathology, and post-mortem. This search was complemented with three review articles [[22], [23], [24]], text word searching and examining references in identified articles. A total of 24 studies were identified that included 149 individuals (range 1–43 subjects per series). Reported gross and microscopic findings and results of SARS-CoV-2 targeted studies are summarized in Table 1 . Representative gross, microscopic, and ultrastructural findings are illustrated in Fig. 1 .
Table 1
Summary of Published COVID-19 Reports with Autopsy Brain Findings.
| Reference | No. Cases Included; autopsy type | Macroscopic Evaluation | Microscopic Evaluation | SARS-CoV-2 Protein | SARS-CoV-2 RNA |
|---|---|---|---|---|---|
| Puelles et al. 2020 [41] Wichmann et al. 2020 [49] Matschke et al. 2020 [35] |
43; subset full autopsy with brain findings | Edema (n = 23), fresh territorial infarct (n = 6) | Fresh ischemic infarct (n = 6), astrocytosis, microgliosis, perivascular, parenchymal, and leptomeningeal T cells (n = 43) | Viral spike or nucleocapsid IHC positive in 16/40 cases (rare cells in medulla; 2 cases with vagus or glossopharyngeal nerves) | qRT-PCR positive (13/27; median 4700 viral E gene copies/cell; range <1000 to 162,000) in frontal lobe and/or medulla |
| Solomon et al. 2020 [37] | 18; brain-only findings | No specific findings | Mild to moderate acute hypoxic injury (n = 18); rare foci of perivascular and leptomeningeal inflammation (n = 3) | Viral nucleocapsid IHC negative in all cases | qRT-PCR positive (n = 5; 5.0–59.4 N1/N2 copies/μL) |
| Remmelink et al. 2020 [32] | 11; full autopsy with brain findings | Recently drained subdural hematoma (n = 1); cerebral hemorrhage (n = 1) | Cerebral hemorrhage or hemorrhagic suffusion (n = 8), focal ischemic necrosis (n = 3), edema and/or vascular congestions (n = 5), diffuse or focal spongiosis (n = 10) | N.A. | qRT-PCR positive (n = 9; viral E gene; Ct: 28.67–35.11) |
| Schurink et al. 2020 [38] | 11; full autopsy with brain findings | No specific findings | Hypoxic changes, activation/clusting of microglia, astrogliosis, perivascular cuffing of T cells most prominent in olfactory bulbs and medulla (n = 11); neutrophilic plugs (n = 3) | Viral nucleocapsid IHC negative in 11 cases | N.A. |
| Fabbri et al. 2020 [31] | 10; full autopsy with brain findings | Edema and meningeal congestion (n = 10), cerebral infarction (n = 3), uncal herniation (n = 2), purulent leptomeninges (n = 1), subarachnoid hemorrhage (n = 1) | Global hypoxic-ischemic injury (n = 10), acute hypoxic injury (all), intravascular microthrombi (n = 10), macro and/or microinfarcts (n = 10); perivascular microhemorrhage (n = 10), microglial activation (n = 5), perivascular/leptomeningeal lymphocytic inflammation (n = 1) | N.A. | qRT-PCR positive in olfactory nerve and brain tissue in (n = 1; RdRp, E, and N genes) |
| Schaller et al. 2020 [50] | 10; full autopsy with brain findings | No specific findings | No specific findings | N.A. | N.A. |
| Hanley et al. 2020 [34] | 9; full autopsy with brain findings | Hemorrhagic conversion of middle cerebral artery stroke (n = 1) | Moderate to intense microglial activation; mild T- cell infiltrate around blood vessels and capillaries, and ischemic changes of variable extent in the neurons of the cortex and the white matter (n = 5) | N.A. | qRT-PCR positive (n = 4; 101 to 104 viral E gene copies per μg total RNA); Subgenomic viral RNA positive (n = 1; Ct ∼32) |
| Deigendesch et al. 2020 [36] Menter et al. 2020 [26] * |
7; full autopsy with brain findings | Moderate global brain edema without cerebral mass displacement (n = 1) | Microglial activation in pons, medulla, and olfactory bulb; sparse perivascular and leptomeningeal infiltrates of lymphocytes; mild acute hypoxic-ischemic encephalopathy (n = 3) | N.A. | qRT-PCR positive in olfactory bulb (n = 4), optic nerve (n = 2); not detected in brainstem or cerebellum (ORFab1, S, and N genes) |
| von Weyham et al. 2020 [27] | 6; full autopsy with brain findings | Massive hemorrhage and herniation (n = 2); petechial bleedings (n = 4) | Hypoxic alterations (n = 6); lymphocytic meningitis and encephalitis (n = 6); brainstem neuronal cell loss in (n = 4), axon degeneration (n = 3) | N.A. | N.A. |
| Bradley et al. 2020 [28] | 5; full autopsy with brain findings | Scattered punctate subarachnoid hemorrhages (n = 1) | Rare microhemorrhages in the brainstem (n = 1) | N.A. | N.A. |
| Kantonen et al. 2020 [30] | 4; full autopsy with brain findings | Mild brain swelling, discoloration of watershed areas, lacunar infarcts, and microhemorrhages in cerebral and cerebellar white matter, deep gray matter, and brain stem (n = 1) | High density acute microhemorrhages, severe hypoxic-ischemic injury, scattered T lymphocytes, and axonal spheroids (n = 1); mild to moderate hypoxic-ischemic injury (n = 3) | Viral spike IHC negative in brain, olfactory mucosa, and carotid body | qRT-PCR negative in brain and olfactory mucosa (RdRp, N. and E genes) |
| Bussani et al. 2020 [51] | 3; fill autopsy with brain findings | N.A. | Gliosis, neuronal loss, vascular rarefaction | N.A. | N.A. |
| Barton et al. 2020 [52] | 2; full autopsy with brain findings | No gross abnormalities | N.A. | N.A. | N.A. |
| Jaunmuktane et al. 2020 [29] | 2; brain-only findings | Large acute and subacute infarcts (n = 1); white matter microhemorrhages and microinfarcts (n = 1) | Hemorrhages and infarcts (n = 2); mild leptomeningeal inflammation (n = 1) | N.A. | N.A. |
| Kirschenbaum et al. 2020 [39] | 2; brain-only findings | N.A. | Perivascular leukocytic infiltrates in basal ganglia and intravascular microthrombi (n = 2); prominent leukocytic infiltrates in olfactory epithelium (n = 2) | N.A. | N.A. |
| Al-Dalahmah et al. 2020 [33] | 1; full autopsy with brain findings | Cerebellar hemorrhage, acute infarcts in the dorsal pons and medulla, tonsillar herniation | Global hypoxia; numerous microglial nodules and neuronophagia in the inferior olives and cerebellar dentate nuclei; mild perivascular and sparse parenchymal and leptomeningeal lymphocytes; perivascular hemorrhages; chronic active inflammation in olfactory epithelium; red neurons in olfactory bulb and normal tract | Viral nucleocapsid IHC negative | qRT-PCR positive in nasal epithelium (Mean Ct 31.75, 278 copies/μL RNA), olfactory bulb (Ct 36.70, 11 copies/μL); Cerebellar clot (Ct 33.0, 559 copies/μL), and cerebellum (Ct 37.17, 8 copies/μL); Viral ISH negative |
| Craver et al. 2020 [53] | 1; full autopsy with brain findings | No CNS lesions identified | No CNS lesions identified | N.A. | N.A. |
| Dolhnikoff et al. 2020 [54] | 1; full autopsy with brain findings | N.A. | Microglial reactivity | N.A. | N.A. |
| Lax et al. 2020 [55] | 1: full autopsy with brain findings | No acute alterations | No acute alterations | N.A. | N.A. |
| Paniz-Mondolfi et al 2020 [12] | 1; brain-only findings | N.A. | N.A. | TEM showed viral like particles in frontal lobe sections | qRT-PCR positive (four different assays targeting ORF1/a and E-gene, N1, N2, N3, N2 and E-gene, and ORF1ab and S genes) |
| Reichard et al 2020 [14] | 1; brain-only findings | Mild brain swelling and hemorrhagic white matter lesions | Focal hemorrhage, ADEM-like lesions, microinfarcts, damaged axons, hypoxic-ischemic injury | N.A. | N.A. |
Abbreviations: ADEM, acute disseminated encephalomyelitis; Ct, cycle threshold; qRT-PCR, quantitative reverse transcriptase polymerase chain reaction; E gene, SARS-CoV-2 envelope gene; ORF1ab, open reading frame 1ab; IHC, immunohistochemistry; ISH, in-situ hybridization; RdRp, RNA-dependent RNA polymerase gene; N.A., not available or evaluated; TEM, transmission electron microscopy.
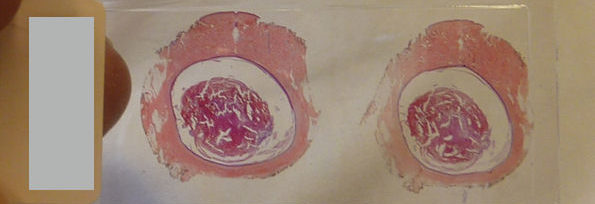
Neuropathological findings of COVID-19. (A) Coronal brain slice from a 55 year old man who died from COVID-19 contains a calcified nodule (arrow) in the right globus pallidus, but is otherwise unremarkable. (B) Hematoxylin and eosin stained section of hippocampus shows scattered hypereosinophilic neurons indicative of acute hypoxic injury. (C) Hematoxylin and eosin stained section shows extravasated red blood cells suggestive of microhemorrhage (deep pink). (D) CD45 immunostaining (brown) highlights a small collection of perivascular immune cells. (E) CD45 immunostaining (brown) also highlights numerous resident immune cells of the brain parenchyma (microglia). (F) In comparison to panel E, a patient without COVID-19 shows minimal CD45 immunostaining (brown). (G) SARS-CoV-2 nucleocapsid immunohistochemistry (brown) shows a cytoplasmic staining pattern in respiratory epithelial cells of the trachea. (H) Transmission electron micrograph of SARS-CoV-2 from cultured cells shows spherical extracellular viral particles (arrows). Images B-F taken at 200x magnification, G at 400x magnification, and are each from a different patient. Image H is from the Centers for Disease Control and Prevents Public Health Image Library, courtesy of Courtesy Cynthia S. Goldsmith and A. Tamin.
Gross brain autopsy findings were reported individually or in aggregate for 142 subjects. In keeping with the high prevalence of comorbidities in this patient population, evidence of prior brain disease was frequently identified, including neurodegeneration, prior strokes, tumor resection, demyelinating disease, and atherosclerosis. Acute gross abnormalities were much more limited, and a direct causal relationship with SARS-CoV-2 infection was not always straightforward to identify. A total of 92 (65 %) of the gross brain examinations reported either no significant findings or no acute abnormalities. Of the remaining 50 cases, multiple findings were often described in individual brains. Hemorrhage was the most common abnormality reported, ranging from petechial bleedings and punctate subarachnoid hemorrhages (n = 9) [14,[27], [28], [29], [30], [31]], to large cerebral/cerebellar hemorrhages (n = 4) [27,32,33], hemorrhagic conversion of middle cerebral artery stroke (n = 1) [34], and a recently drained subdural hematoma (n = 1) [32]. Large acute and/or subacute infarcts (n = 11) [29,31,33,35] as well as lacunar infarcts/microinfarcts and watershed infarcts (n = 2) [29,30] were identified in several cases. Severe edema resulting in herniation (n = 5) [27,31,33] as well as mild to moderate edema without herniation (n = 34) [14,30,31,35,36] were also present.
Microscopic findings were reported for 146 of the cases in these studies. Similar to the gross examinations, histopathology identified correlates of pre-existing disease, including neurodegeneration, chronic/subacute strokes, hepatic encephalopathy, and arteriolosclerosis. No specific findings were reported for 25 (17 %) of the cases. Mild to moderate acute hypoxic injury was the most common abnormality (n = 58) [14,27,30,31,33,34,[36], [37], [38]], while severe hypoxic-ischemic injury (n = 1) [30] and infarcts/focal ischemic necrosis (n = 22) [14,29,31,32,35] were identified in several cases. Focal microhemorrhage or hemorrhagic suffusion was also frequently reported (n = 23) [14,[28], [29], [30], [31], [32], [33]], although intravascular microthrombi (n = 12) [31,39] or neutrophilic plugs (n = 3) [38] were less common. Mild focal perivascular, parenchymal, and leptomeningeal T-cell predominant lymphocytic infiltrates were identified in a large number of cases without clear evidence of vasculitis or meningoencephalitis (n = 81) [27,[29], [30], [31],[33], [34], [35], [36], [37], [38], [39]]. Moderate to intense microglial activation was noted, particularly in the brainstem (n = 73), although similar results were also reported in COVID-19-negative individuals with systemic inflammatory/septic clinical courses [31,[33], [34], [35], [36],38]. Axonal damage was identified in a few cases (n = 5) [14,27,30]. Acute disseminated encephalomyelitis (ADEM)-like lesions were reported in a single case [14]. The olfactory system was examined to varying degrees, identifying prominent acute and chronic inflammation in the olfactory epithelium (n = 14) [33,38,39], microglial activation (n = 18) [36] and red neurons (n = 1) [33] in the olfactory bulb, and only unremarkable age-related corpora amylacea in olfactory tracts.
Researchers across the globe have employed multiple strategies to directly assess for the presence of SARS-CoV-2 in brain tissue, including immunohistochemistry, in situ hybridization (ISH), targeted quantitative reverse transcriptase polymerase chain reaction (qRT-PCR), and transmission electron microscopy. At this time, immunohistochemistry, using antibodies that recognize the viral nucleocapsid (N) or spike (S) proteins, have been negative in most attempted human cases (n = 58) [30,33,35,37,38], with the exception of a recent case series that reported positive staining in vagus and glossopharyngeal nerves and scattered cells in the medulla in a total of 16 cases [35]; in situ hybridization for viral RNA has been negative (n = 1) [33]. Viral spike protein has been reported to be present in the olfactory epithelium in 5/6 patients; however, brain findings from these cases were not discussed [40]. A number of qRT-PCR assays have been employed targeting the N, S, envelope (E), open reading frame (ORF) 1/a, ORF1ab, or RNA-dependent reverse transcriptase (RdRp) genes, identifying low levels of virus in frozen or formalin-fixed paraffin-embedded brain tissue (34/84; 41 %) [12,[30], [31], [32], [33], [34], [35], [36], [37],41] and olfactory bulb/tract (n = 9/36; 25 %) [31,33,36,37]. Viral subgenomic RNA, a marker of actively replicating virus, was positive in a single case (n = 1/5; 20 %) [34]. Transmission electron microscopy (TEM) without immunolabeling reported virus-like particles in the frontal lobe (n = 1) [12].
While additional COVID-19 autopsy series continue to be published, the overall picture of acute hypoxic-injury, hemorrhage, and mild to moderate non-specific inflammation is unlikely to change significantly. Evidence of direct viral involvement in the brain or olfactory nerve is limited to the detection of low levels of viral RNA and rare viral antigen in cranial nerves and scattered brainstem cells. Diagnosis of coronavirus particles by electron microscopy is challenging due to similar appearing normal cellular structures, which has created significant controversy in the literature [42,43]. Due to the inherent bias of autopsy studies for severe, fatal disease, and additional institutional restrictions for which cases include brain evaluation, the frequency and extent of neuropathological findings are likely to be overestimated relative to the average COVID-19 patient. At the time of this review, pediatric autopsies, including individuals with multisystem inflammatory syndrome in children (MIS-C), remain extremely limited. While the number of pediatric COVID-19 cases accounts for <2 % of all cases [44], data obtained from brain tissue in this age-group can help address the unique pathophysiology of SARS-CoV-2 infection, including age-dependent immune-responses, hypercoagulability, and degree of hypoxic-ischemic injury.
Additional remaining areas of interest include characterizing the effects of remdesivir and other potential antiviral therapeutics, immunomodulatory medications including dexamethasone, anti-IL-6 or other monoclonal antibodies, and anticoagulants on brain tissue. Given that the therapeutic response to COVID-19 vastly differs between institutions, it remains a challenge to understand how therapeutic choices during acute hospitalization are responsible for the variability in observed neurological manifestations and neuropathological findings. Also, while not surprisingly this early in the pandemic, long-term neuropathological sequelae in COVID-19 survivors remain unstudied. There is evidence that neurological symptoms, including fatigue and headaches, linger for weeks to months in a subset of affected patients [45,46] and studies determining mechanisms for persistent neurological symptoms are needed.
There have been several efforts for sharing COVID-19 brain tissue, including the International Society of Neuropathology (ISN) Collaborative Efforts [47] and the COVID-19 Virtual Biobank at the University of Nebraska Medical Center [48]. To address many of the remaining unanswered questions regarding the neuropathological effects of COVID-19, large scale integrated studies from multiple institutions with relevant clinical metadata will be crucial. The ongoing collection of neurological tissue will be critical to inform best practice management guidance and to direct research priorities as it relates to neurological morbidity from COVID-19.
Funding sources
S.S.M. was supported by the National Institute of Mental Health at the National Institutes of Health [grant number K23MH115812], James S. McDonnell Foundation and the Harvard University Eleanor and Miles Shore Fellowship Program. I.H.S. was supported by the National Institute of Neurological Disorders and Stroke at the National Institutes of Health [grant number R21NS119660].
Acknowledgements
We would like to acknowledge all front-line healthcare workers taking care of patients during the COVID-19 pandemic, and patients and their families who contribute research to help understand neurological disease.

 Views Today : 2169
Views Today : 2169
Unquestionably believe that that you said. Your favourite justification appeared to be at the net the simplest thing to understand of. I say to you, I definitely get irked whilst folks consider concerns that they plainly don’t recognize about. You controlled to hit the nail upon the highest and also outlined out the whole thing with no need side-effects , other people could take a signal. Will probably be again to get more. Thank you
I just wanted to send a brief note to thank you for some of the great ways you are writing here. My time-consuming internet look up has now been recognized with excellent facts and techniques to go over with my family members. I would believe that many of us readers actually are rather lucky to dwell in a remarkable website with very many brilliant professionals with interesting ideas. I feel somewhat lucky to have come across the webpage and look forward to plenty of more exciting moments reading here. Thank you once again for all the details.
How can I contact you? I am interested in more information.
[url=https://stromectolgf.com/#]stromectol 3 mg tablets price[/url] ivermectin buy nz
Thx so much ! Appreciate the helpful info.
I really like your writing!
I just bookmarked your site.
I am interested in more information. How can I contact you?
I am 45 year old mom Thank you so much I want you to see me naked
Thank you for sharing the info.
How can I contact you? I am interested in more info.
Thanks so much for sharing the information.
[url=https://drwithoutdoctorprescription.online/#]buy prescription drugs online[/url] buy prescription drugs without doctor
[url=https://drwithoutdoctorprescription.online/#]prescription drugs online without doctor[/url] carprofen without vet prescription
[url=https://sildenafilmg.online/#]viagra cost per pill[/url] when will viagra be generic
Interested in more information. How can I contact you?
You are a great writer. Do you have any more sites?
I am 47 year old female Thank you so much!
[url=https://sildenafilmg.online/#]viagra without a doctor prescription[/url] buying viagra online
Interested in more information. How can I reach you?
I am a adult female looking to talk.
Thanks so much the help.
I am an older women who is 45 years old thank you.
[url=https://sildenafilmg.online/#]over the counter viagra[/url] best place to buy viagra online
This information is so great thanks!
I am interested in more info. How can I contact you?
This info is so great thanks!
I am 41 year old mom Thank you so much!
I just saved your page.
This info is so amazing thanks!
Thx so much ! Appreciate the helpful information.
[url=https://prednisoneforsale.store/#]prednisone tablets india[/url] prednisone 40 mg tablet
How can I get more useful info?
Thank you for sharing the information.
How can I get more useful info?
I been searching for this stuff
Thanx very much for helping me.
[url=https://amoxilforsale.best/#]amoxicillin capsules 250mg[/url] buy amoxicillin 500mg usa
I really admire your writing!
[url=https://buylipitor.store/#]lipitor 10mg price comparison[/url] lipitor canada
штабелер электрический
[url=https://elektroshtabeler-kupit.ru]http://elektroshtabeler-kupit.ru/[/url]
Appreciate the helpful information
Interested in more information. How can I contact you?
Thanks for your hard work.
Interested in more info. How can I contact you?
[url=https://buylasix.icu/#]furosemide 40mg[/url] lasix online
Thanks the help.
Very good information thanks so much!
You are an great writer! I just saved your website.
I really like your writing I want you to see me naked
Thanks the help.
Thanks for your hard work.
[url=https://erectionpills.best/#]ed remedies[/url] the best ed pills
штабелер самоходный
[url=https://shtabeler-elektricheskiy-samokhodnyy.ru]http://www.shtabeler-elektricheskiy-samokhodnyy.ru[/url]
[url=https://diflucan.icu/#]diflucan over the counter pill[/url] diflucan 150 mg capsule
Thanks so much for your hard work.
I am interested in more info. How can I reach you?
I just bookmarked your page.
Thanks so much for sharing.
Interested in more info. How can I contact you?
[url=https://withoutprescription.store/#]meds online without doctor prescription[/url] cat antibiotics without pet prescription
[url=https://drugsonline.store/#]mens erections[/url] best ed treatment
I am interested in more information. How can I contact you?
[url=https://drugsonline.store/#]treat ed[/url] online ed pills
[url=https://clomidonline.icu/#]generic clomid for sale[/url] clomid for sale
подъемник ножничный передвижной
[url=https://nozhnichnyye-podyemniki-dlya-sklada.ru]https://www.nozhnichnyye-podyemniki-dlya-sklada.ru/[/url]
[url=https://stromectoltrust.com/#]stromectol 3 mg tablets price[/url] stromectol 12 mg tablets
Thanks so much the info.
Thank you for your hard work.
I appreciate the information.
How can I get more helpful information?
I am an older women who is 43 years old thanks.
I am interested in more info. How can I reach you?
I really admire your writing!
I am a adult female looking to chat.
[url=https://pharmacyizi.com/#]errectile dysfunction[/url] treat ed
[url=https://pharmacyizi.com/#]pills for erection[/url] mexican pharmacy without prescription
[url=https://pharmacyizi.com/#]cure ed[/url] website
[url=https://pharmacyizi.com/#]legal to buy prescription drugs without prescription[/url] drugs prices
[url=https://onlinepharmacy.men/#]canadian pharmacy coupon code[/url] best online foreign pharmacies
Interested in more info. How can I reach you?
[url=https://allpharm.store/#]top rated canadian pharmacies online[/url] Dulcolax
How can I get more helpful info?
I really admire your writing!
Thanks for sharing the information.
I just saved your page I want you to see me naked
Thank you very much for helping me.
[url=https://erectionpills.shop/#]treatments for ed[/url] best ed pill
Appreciate the useful info
I really like your writing!
Thanx very much for helping me.
[url=https://allpharm.store/#]online medicine order discount[/url] canadian rx pharmacy online
[url=https://allpharm.store/#]canadian pharmacy online[/url] Vastarel
[url=https://canadiandrugs.best/#]how can i order prescription drugs without a doctor[/url] mexican pharmacy without prescription
Thanks so much for sharing.
Thank you the information.
This information is so great thanks!
рохля электрическая
[url=https://samokhodnyye-elektricheskiye-telezhki.ru]http://www.samokhodnyye-elektricheskiye-telezhki.ru[/url]
[url=https://onlinepharmacy.men/#]canada discount pharmacy[/url] best online pharmacy
самоходная тележка
[url=https://samokhodnyye-elektricheskiye-telezhki.ru]https://samokhodnyye-elektricheskiye-telezhki.ru[/url]
You are a great writer thank you.
[url=https://medrxfast.com/#]best ed pills non prescription[/url] legal to buy prescription drugs without prescription
мачтовый подъемник
[url=https://podyemniki-machtovyye-teleskopicheskiye.ru]https://podyemniki-machtovyye-teleskopicheskiye.ru[/url]
I am 47 year old mom Thank you so much!
Thx so much !! Appreciate the helpful information.
Thanks for sharing the information.
[url=https://medrxfast.com/#]prescription drugs canada buy online[/url] canadian pharmacy
[url=https://medrxfast.com/#]legal to buy prescription drugs without prescription[/url] carprofen without vet prescription
I appreciate the help.
I am an adult female and just found your site.
I am interested in more information. How can I reach you?
подъемник мачтовый
[url=https://podyemniki-machtovyye-teleskopicheskiye.ru]https://podyemniki-machtovyye-teleskopicheskiye.ru[/url]
I cannot thank you enough for the blog article.Really looking forward to read more. Keep writing.
Thank you for sharing.
I really like your writing!
гидравлический подъемный стол
[url=https://gidravlicheskiye-podyemnyye-stoly.ru]https://gidravlicheskiye-podyemnyye-stoly.ru[/url]
Very good information thanks so much!
You are a great writer. Do you have any more webpages?
You are a great writer thank you.
Very neat post.Really looking forward to read more. Fantastic.
Very good information thanks so much!
You are a great writer thanx.
How can I reach you? I am interested in more info.
Thanks for sharing the info.
Just wanted to say thank you!
I just bookmarked your webpage I want you to see me naked
I really like your writing!
I am interested in more information. How can I reach you?
[url=https://wellbutrin.best/#]wellbutrin drug[/url] wellbutrin best price
I appreciate you sharing this blog post.Really looking forward to read more. Really Cool.
Thank you for your article post. Really Great.
Great, thanks for sharing this blog post.Really looking forward to read more. Great.
Fantastic article. Want more.
Major thanks for the article. Fantastic.
Really appreciate you sharing this blog article.Really thank you!
Thanx so much for helping me.
Thanks I am mother and extremely busy.
I am 40 year old mom. I just bookmarked your website
I really like your writing!
Major thanks for the blog.Thanks Again. Will read on…
Very informative blog post.Really thank you! Keep writing.
You are a great writer thank you.
Wow, great post.Much thanks again. Cool.
Very good information thanks so much!
Thanks-a-mundo for the post.Thanks Again. Cool.
You are a great writer thank you.
Thanks so much the help.
[url=https://glucophage.top/#]can you buy metformin over the counter[/url] india pharmacy metformin online
I am a adult female looking to talk.
Very good info thanks so much!
I am interested in more information. How can I contact you?
How can I get more useful info?
Thanks-a-mundo for the blog.Much thanks again. Much obliged.
You are a great writer. Do you have any more webpages?
I really like your writing!
Thanks for sharing.
I just turned 74 this year and I really like your writing!
I am so grateful for your article post. Really Great.
I am interested in more information. How can I contact you?
Just wanted to say thanks!
How can I contact you? I am interested in more information.
I just bookmarked your page.
Thank you for your hard work I want you to see me naked
How can I contact you? I am interested in more information.
[url=https://glucophage.top/#]how to get metformin over the counter[/url] metformin price
This information is so great thanks!
You are a great writer. Do you have any more webpages?
You are a great writer thank you.
How can I contact you? I am interested in more info.
I am a mother looking to talk.
You are a great writer thanx.
Major thanks for the article. Great.
Thank you for your blog. Great.
I really like your writing!
Really enjoyed this blog post.Much thanks again. Really Cool.
Thank you ever so for you blog post.Much thanks again. Really Cool.
This info is so great thanks!
This info is so amazing thanks!
You are a great writer I want you to see me naked Do you have any more sites
Thank you for your hard work.
You are a great writer. Do you have any more webpages?
[url=https://sildenafil.pro/#]sildenafil price in usa[/url] sildenafil citrate 100 mg
Appreciate you sharing, great blog post. Keep writing.
How can I get more helpful information?
You are a great writer. Do you have any more webpages?
Interested in more info. How can I contact you?
This info is so amazing thanks!
This info is so amazing thanks!
Thanks so much for sharing.
Interested in more information. How can I reach you?
Very informative blog.Really looking forward to read more. Much obliged.
I really liked your blog post.Really thank you! Cool.
[url=https://pharmacy.ink/#]best no prescription pharmacy[/url] canadian pharmacy cheap
[url=https://ciprofloxacin.icu/#]cipro 500mg best prices[/url] ciprofloxacin over the counter
I am interested in more info. How can I contact you?
Very good information thanks so much!
Im obliged for the article. Keep writing.
I just turned 68 this year and I really admire your writing!
You are a great writer. Do you have any more websites?
You are a great writer thank you.
This info is so great thanks!
Very good info thanks so much!
You are a great writer. Do you have any more webpages?
I am interested in more info. How can I contact you?
How can I get more helpful info?
Very neat article post. Much obliged.
You are a great writer thank you.
How can I reach you? I am interested in more information.
I really like your writing!
Thanks so much for your hard work.
You are a great writer thank you.
Just wanted to say thanks!
I cannot thank you enough for the article.Really looking forward to read more. Keep writing.
Thx so much ! Appreciate the helpful info.
Fantastic blog post.Much thanks again. Really Great.
Thanks for sharing the information.
Major thankies for the article post.Thanks Again. Keep writing.
You are a great writer. Do you have any more webpages?
I really like your writing!
Interested in more information. How can I contact you?
Thanks so much I am going to show my friends your webpage.
You are a great writer. Do you have any more webpages?
Thx so much !! Appreciate the helpful information.
This info is so amazing thanks!
I appreciate the info.
I really admire your writing!
I cannot thank you enough for the blog post. Keep writing.
Thank you so much for helping me.
How can I get more helpful info?
You are a great writer. Do you have any more webpages?
Thanks so much for sharing.
Very good info thanks so much!
I have been looking for this stuff
Appreciate the useful info
Thanks so much for your hard work.
Looking forward to reading more. Great article post.
Very good blog article. Awesome.
Thx so much !!! Appreciate the useful information.
I am so grateful for your post.Much thanks again. Keep writing.
Very informative blog.Really thank you! Cool.
I truly appreciate this article post.Much thanks again. Much obliged.
Very good info thanks so much!
Very good info thanks so much!
I am 48 year old female Thank you so much!
Major thanks for the blog.Much thanks again. Cool.
Thx so much !!! Appreciate the helpful information.
Interested in more information. How can I reach you?
Appreciate the helpful info
I am interested in more information. How can I reach you?
I am 71 years old how can I get more info?
How can I reach you? I am interested in more information.
Thx so much !!! Appreciate the useful information.
I am a mother looking to talk.
I really enjoy the blog.Really thank you! Awesome.
Thank very much for helping me.
Thanks so much for your hard work.
Extremely useful information thank you.
Im thankful for the article. Great.
I appreciate you sharing this blog article. Awesome.
Very neat post.Thanks Again.
This information is so amazing thanks!
This information is so great thanks!
How can I get more helpful information?
You are a great writer. Do you have any more websites?
I am 47 year old mom Just saying thanks!
Thank you the info I want you to see me naked
Thanx very much for helping me.
This information is so great thanks!
Appreciate you sharing, great blog post.Thanks Again. Awesome.
How can I get more helpful information?
I really admire your writing!
I am 44 year old female Just saying thanks!
I am 76 years old how can I get more information?
I am a mother looking to talk.
I just turned 74 this year and I really like your writing!
Thank you I am going to share my friends your webpage.
Very helpful info thank.
You are an amazing writer! I just saved your webpage.
Thx so much !!! Appreciate the helpful info.
Thank you for sharing the info I want you to see me naked
I am 41 year old mom Thank you so much!
I must thank you for the efforts you’ve put in writing this website. I really hope to check out the same high-grade content by you in the future as well. In truth, your creative writing abilities has inspired me to get my own, personal website now 😉
Thank you for sharing.
I really like your writing!
I am an older women and I really admire your writing!
Interested in more info. How can I reach you?
Thank you very much for helping me.
Very good information thanks so much!
I have been seeking for this information
hello I am an adult female age 41 love to chat.
I been looking for this info
Thanx so much for helping me.
Very good information thanks so much!
I am 45 year old female Thank you so much!
Thanks so much for sharing the information.
I am interested in more info. How can I contact you?
Really informative article. Cool.
Good post. I learn something totally new and challenging on sites I stumbleupon on a daily basis. It will always be useful to read through content from other authors and practice something from their sites.
Thank you for sharing.
Thanks for sharing.
Appreciate the useful info
You are a great writer thank you.
Fantastic post.Really thank you!
Hi, I do think this is an excellent website. I stumbledupon it 😉 I will revisit yet again since I book-marked it. Money and freedom is the greatest way to change, may you be rich and continue to help other people.
You need to be a part of a contest for one of the most useful sites online. I will recommend this blog!
Very good article. Awesome.
I value the post.Really thank you! Great.
I am a older women looking to chat.
This is one awesome blog article.Much thanks again. Want more.
Thanks-a-mundo for the article post.Much thanks again. Want more.
I appreciate the information.
Thank you the information.
You are a great writer. Do you have any more webpages?
39 year old female just saying thanx.
I am 41 year old female Thank you so much!
Thank you so much for helping me.
Interested in more info. How can I reach you?
Thanks for sharing, this is a fantastic article.Really looking forward to read more. Keep writing.
wow, awesome article.Really thank you! Keep writing.
Thank you for sharing.
Interested in more information. How can I reach you?
Greetings! Very useful advice in this particular article! It is the little changes which will make the largest changes. Thanks for sharing!
I just bookmarked your site.
I cannot thank you enough for the blog post.Thanks Again. Awesome.
I must thank you for the efforts you have put in writing this site. I’m hoping to check out the same high-grade blog posts from you in the future as well. In fact, your creative writing abilities has inspired me to get my own, personal blog now 😉
I just bookmarked your webpage.
How can I reach you? I am interested in more information.
I am an older women and just found your page.
I am 44 year old female Just saying thanks!
Thanks for sharing.
Thanks so much the info.
You are a great writer thanks.
I am a mom looking to chat.
Greetings, There’s no doubt that your site could possibly be having web browser compatibility issues. Whenever I look at your site in Safari, it looks fine however, if opening in Internet Explorer, it has some overlapping issues. I merely wanted to provide you with a quick heads up! Other than that, great website!
Thanks so much for sharing the information.
Hey, thanks for the article post.Really thank you!
Thank you for sharing.
Say, you got a nice article post.Much thanks again. Much obliged.
Appreciate the helpful information
The very next time I read a blog, Hopefully it won’t disappoint me as much as this particular one. After all, I know it was my choice to read, however I actually thought you would probably have something useful to say. All I hear is a bunch of moaning about something that you can fix if you weren’t too busy searching for attention.
Very informative blog article.Really thank you! Really Cool.
I think this is a real great article post.Really looking forward to read more. Will read on…
How can I get more helpful information?
Thank you for your article.Really thank you! Keep writing.
Appreciate the useful info
How can I reach you? I am interested in more information.
This is a very good tip especially to those new to the blogosphere. Simple but very precise infoÖ Thank you for sharing this one. A must read post!
Right here is the perfect web site for anyone who would like to find out about this topic. You realize so much its almost hard to argue with you (not that I really will need to…HaHa). You certainly put a new spin on a topic which has been written about for decades. Great stuff, just excellent.
Appreciate the helpful info
Hey, thanks for the article post.Really looking forward to read more. Keep writing.
Appreciate the helpful info
Very informative post.Really thank you!
Thank you very much for helping me.
May I just say what a relief to uncover a person that genuinely understands what they are discussing on the web. You actually know how to bring an issue to light and make it important. More and more people need to look at this and understand this side of your story. It’s surprising you aren’t more popular because you most certainly possess the gift.
This info is so amazing thanks!
Thank you I am older women and extremely busy.
You are a great writer thanks.
Thanx very much for helping me.
Great blog post.Really looking forward to read more. Really Cool.
Hey, thanks for the article.Much thanks again. Fantastic.
Hi, I believe your blog might be having web browser compatibility issues. Whenever I look at your web site in Safari, it looks fine however, if opening in IE, it’s got some overlapping issues. I simply wanted to give you a quick heads up! Other than that, great site.
Muchos Gracias for your article post.Really thank you! Want more.
Thanx so much for helping me.
You are a great writer. Do you have any more websites?
I am 74 years old how can I get more information?
Everything is very open with a really clear clarification of the issues. It was definitely informative. Your website is very helpful. Thanks for sharing.
Thank you for your blog post.Thanks Again. Really Cool.
You’re so interesting! I don’t believe I’ve truly read through a single thing like this before. So great to find another person with a few genuine thoughts on this subject. Really.. thank you for starting this up. This web site is something that’s needed on the web, someone with some originality.
Appreciate the helpful information
Just wanted to say thanx!
I am an adult female and I found this useful
Thanks for your hard work.
Interested in more info. How can I contact you?
Interested in more information. How can I contact you?
Really informative article.Really looking forward to read more. Great.
Interested in more information. How can I reach you?
Thank you the information.
You are a great writer. Do you have any more websites?
You are a great writer. Do you have any more websites?
I am interested in more information. How can I contact you?
This info is so amazing thanks!
Oh my goodness! Incredible article dude! Many thanks, However I am experiencing problems with your RSS. I don’t understand the reason why I cannot subscribe to it. Is there anyone else having the same RSS issues? Anyone who knows the solution will you kindly respond? Thanks!!
Thank you for your blog.Really looking forward to read more. Will read on…
This is one awesome blog article. Awesome.
Very neat article. Cool.
I value the blog.Much thanks again. Really Great.
Hi there! This blog post couldnít be written any better! Looking at this post reminds me of my previous roommate! He continually kept preaching about this. I most certainly will forward this article to him. Fairly certain he’s going to have a great read. I appreciate you for sharing!
Thanks for the article post.
I cannot thank you enough for the blog article.Thanks Again. Really Great.
I am a mom looking to chat.
I am interested in more info. How can I reach you?
I am 47 year old female Thank you so much!
Thx so much ! Appreciate the helpful info.
Very good information thanks so much!
I am 40 year old mom Thank you so much!
Awesome post.Thanks Again. Keep writing.
I am 47 year old female Thank you so much!
I am 46 year old female Thank you so much!
Appreciate the helpful information
This info is so great thanks!
I am 39 year old mom Thank you so much!
I am a older women looking to chat I want you to see me naked
Appreciate the useful information
How can I contact you? I am interested in more information.
Aw, this was a really good post. Taking a few minutes and actual effort to create a really good article… but what can I say… I procrastinate a lot and don’t manage to get anything done.
Thanks for your hard work.
I am an older women and just found your page.
You are a great writer. Do you have any more webpages?
After I originally commented I appear to have clicked the -Notify me when new comments are added- checkbox and now each time a comment is added I recieve four emails with the same comment. There has to be a means you are able to remove me from that service? Appreciate it.
Greetings, I do believe your web site may be having web browser compatibility problems. When I take a look at your blog in Safari, it looks fine however, when opening in IE, it’s got some overlapping issues. I merely wanted to provide you with a quick heads up! Besides that, great website!
That is a really good tip especially to those new to the blogosphere. Short but very accurate info… Thank you for sharing this one. A must read article.
Thanks so much the help.
I am 40 year old female Thank you so much!
Thanks so much for your hard work.
How can I reach you? I am interested in more info.
Interested in more info. How can I reach you?
I would like to thank you for the efforts you have put in penning this site. I’m hoping to view the same high-grade content by you later on as well. In fact, your creative writing abilities has inspired me to get my own website now 😉
I am interested in more info. How can I reach you?
I am interested in more information. How can I reach you?
This info is so amazing thanks!
Thanks so much the help I want you to see me naked
I am interested in more info. How can I contact you?
How can I reach you? I am interested in more info.
This website was… how do you say it? Relevant!! Finally I’ve found something which helped me. Kudos.
I am 48 year old female Thank you so much!
Just wanted to say thanks!
Thank you for sharing.
Interested in more info. How can I reach you?
Appreciate the helpful info
Thx so much !! Appreciate the helpful information.
This info is so great thanks!
I am interested in more info. How can I contact you?
I truly appreciate this article post.Thanks Again. Really Cool.
Interested in more information. How can I contact you?
40 year old mom just saying thank you.
You are a great writer thank you I want you to see me naked
Wow, great blog post.Really thank you! Great.
Thanks the help.
I am a mom looking to talk.
This information is so amazing thanks!
Very neat blog post.Thanks Again. Want more.
I really like your writing!
Thank you I am older women and extremely busy.
Appreciate the useful info
How can I get more useful information?
Enjoyed every bit of your post.Much thanks again. Fantastic.
Really appreciate you sharing this blog post.Thanks Again. Cool.
An impressive share! I have just forwarded this onto a co-worker who had been conducting a little research on this. And he actually bought me breakfast due to the fact that I stumbled upon it for him… lol. So allow me to reword this…. Thanks for the meal!! But yeah, thanx for spending time to talk about this matter here on your site.
I cannot thank you enough for the article post.Really thank you! Fantastic.
I just bookmarked your website.
This information is so great thanks!
Appreciate the helpful info
I am a mother looking to talk.
This info is so amazing thanks!
I really like your writing!
Very good info thanks so much!
I truly appreciate this article post.Thanks Again. Really Great.
Interested in more info. How can I contact you?
I am a older women looking to chat.
Appreciate the useful information
You are a great writer. Do you have any more websites?
Wow, great blog.Thanks Again. Really Cool.
Hi there! This blog post could not be written any better! Going through this post reminds me of my previous roommate! He always kept talking about this. I am going to forward this information to him. Pretty sure he will have a very good read. Many thanks for sharing!
You are a great writer thanks.
I am interested in more information. How can I contact you?
I am an mother and I really like your writing!
How can I contact you I am interested in more information I want you to see me naked
Appreciate the helpful information
This info is so amazing thanks!
I am 40 year old female Thank you so much!
Thanks so much for sharing the information.
A big thank you for your article post.Much thanks again.
Good post. I learn something totally new and challenging on websites I stumbleupon on a daily basis. It will always be useful to read articles from other writers and practice a little something from their websites.
Awesome article.Thanks Again. Really Cool.
Thanks so much for the article post.Really thank you! Keep writing.
An intriguing discussion is worth comment. I do believe that you should write more on this subject matter, it might not be a taboo matter but generally people don’t discuss such issues. To the next! All the best!
How can I get more helpful info?
I am a mother looking to talk.
You are a great writer thanks.
Thx so much ! Appreciate the useful information.
Thank you for sharing the information.
I am 43 year old female Thank you so much!
Thank you the info I want you to see me naked
This information is so great thanks!
Thx so much ! Appreciate the helpful info.
Greetings! Very useful advice within this article! It’s the little changes that produce the greatest changes. Thanks for sharing!
I appreciate you sharing this article.Much thanks again. Awesome.
The very next time I read a blog, I hope that it doesn’t disappoint me as much as this one. After all, I know it was my choice to read through, however I truly thought you’d have something useful to talk about. All I hear is a bunch of crying about something you could fix if you were not too busy searching for attention.
I appreciate you sharing this article.Much thanks again. Much obliged.
Im thankful for the article.Really thank you! Really Cool.
Thanks so much for sharing.
You saved me so much time greatful I found your website.
I really admire your writing!
I appreciate the information.
I really admire your writing!
Very good info thanks so much!
I was extremely pleased to find this web site. I wanted to thank you for ones time for this particularly fantastic read!! I definitely really liked every bit of it and i also have you book-marked to look at new things on your site.
I truly appreciate this blog post.Really looking forward to read more. Fantastic.
Thank you for sharing.
I am a adult female looking to chat.
How can I reach you? I am interested in more information.
Interested in more information. How can I contact you?
Very good information thanks so much!
Very good information thanks so much!
Very good info thanks so much!
Very good article! We are linking to this particularly great post on our site. Keep up the good writing.
Im grateful for the article. Want more.
Very informative article.Much thanks again. Really Great.
Good day! I could have sworn I’ve been to this blog before but after looking at some of the articles I realized it’s new to me. Regardless, I’m certainly pleased I discovered it and I’ll be bookmarking it and checking back regularly.
I enjoy reading an article that will make men and women think. Also, thank you for allowing for me to comment.
I appreciate you sharing this article post. Keep writing.
An impressive share! I’ve just forwarded this onto a friend who had been conducting a little homework on this. And he actually ordered me breakfast because I found it for him… lol. So allow me to reword this…. Thanks for the meal!! But yeah, thanks for spending the time to talk about this matter here on your site.
Thank you the information.
Thanks for sharing, this is a fantastic blog.Thanks Again. Awesome.
Awesome article post.Really looking forward to read more. Great.
I think this is a real great article post.Really looking forward to read more. Keep writing.
I really enjoy the article.Much thanks again. Awesome.
You are an great writer! I just saved your website.
Extremely useful information thank.
I just bookmarked your site.
I am 37 year old female Just saying thanks!
Thanks I am going to show my friends your site.
I am 39 year old female Just saying thanks!
Just wanted to say thank you!
You are a great writer thanx.
Thank you for your hard work.
Thanks so much for sharing the information I want you to see me naked
Thanks the information.
Good information. Lucky me I found your website by chance (stumbleupon). I have saved as a favorite for later.
I am so grateful for your article.Really looking forward to read more. Keep writing.
I think this is a real great blog article.Really looking forward to read more. Much obliged.
Enjoyed every bit of your article.Really looking forward to read more. Great.
I really liked your article post.Really looking forward to read more. Really Great.
Hey, thanks for the article.Really thank you! Cool.
I am interested in more info. How can I contact you?
You are a great writer. Do you have any more webpages?
Very good info thanks so much!
I am an mother and just found your webpage.
Thanks so much for sharing the information I want you to see me naked
Appreciate you sharing, great blog post.Thanks Again. Will read on…
Very neat blog article.Much thanks again. Much obliged.
Thanks so much for the article post. Keep writing.
Oh my goodness! Impressive article dude! Thank you so much, However I am encountering troubles with your RSS. I don’t understand the reason why I can’t subscribe to it. Is there anybody having similar RSS issues? Anyone who knows the solution can you kindly respond? Thanks!
I just saved your site.
Major thanks for the article post.Thanks Again. Great.
Awesome article.Really looking forward to read more.
Im thankful for the blog.Thanks Again. Awesome.
Interested in more info. How can I contact you?
I really like your writing!
I just saved your site.
I am 41 year old mom Just saying thanks!
Consume this, and also the result will impress even yourself.
I am 47 year old mom Thank you so much!
Thanks the info.
43 year old female just saying thanx.
I just bookmarked your page.
I am an older women and just found your site.
I appreciate the help I want you to see me naked
One item that can take your life to the next level!
hii I am an mother age 48 love to chat.
I really admire your writing!
Reach your objectives, obtain even more done and boost your life!
The best gear for your job and play.
Thank you the information.
I am 40 year old mom Thank you so much!
Thanks so much the information.
Thank you so much for helping me.
Interested in more info. How can I reach you?
I just bookmarked your page.
Very good information thanks so much!
Get all you need and also much more with this!
How can I reach you? I am interested in more info.
I really admire your writing!
How can I reach you? I am interested in more information.
I am 45 year old female Thank you so much!
Thank so much for helping me.
Thanks for sharing the information.
Just wanted to say thank you!
You are a great writer thanx.
This information is so great thanks!
I really like your writing!
I am 37 year old female Thank you so much!
Achieve extra and do it much better with Amazingness!
I am interested in more info. How can I contact you?
I really like your writing!
How can I get more helpful info?
I am 39 year old mom. I just bookmarked your webpage
I am 44 year old mom Thank you so much!
How can I contact you? I am interested in more information.
Thx so much !!! Appreciate the helpful information.
You are a great writer. Do you have any more sites?
The Amazingness is the excellent method to get even more out of your life.
Have a look at just how this device can change your life right!
with Mindfulness you will remain in a better mindset.
You are a great writer. Do you have any more sites?
Thanks so much for sharing.
I really like your writing I want you to see me naked
Thx so much !!! Appreciate the useful information.
Thanks the info.
Amazing is the one quit purchase every little thing efficiency!
Thanks so much for sharing.
How can I contact you? I am interested in more info.
I am an adult female and I found this useful
Thx so much ! Appreciate the helpful info.
Learn just how to feel outstanding and as if your life has a purpose. Take back control of your life with this!
You deserve this life time possibility to have everything you’ve ever wanted.
Terrific to obtain one of the most out of your money. Amazing!
I found this, and currently everything is very easy!
Thanks for sharing.
This information is so great thanks!
The only high-quality and finish 100 natural rest aid that ensures a complete, deep invigorating evening’s sleep!
I located this, as well as currently every little thing is simple!
See exactly how this can function wonders for you by visiting the site!
Thx so much !! Appreciate the helpful information.
Thx so much ! Appreciate the useful info.
I really like your writing!
This info is so great thanks!
Very good info thanks so much!
I am 71 years old how can I get more information?
Thanks I am adult female and extremely busy.
Thanks so much for sharing the information.
This overview will certainly help you to take your company and personal abilities to the following level!
Very good info thanks so much!
I am an mother and I really admire your writing!
Interested in more info. How can I reach you?
All from one little pill, Remarkable gives you incredible power!
It will certainly assist you obtain every little thing performed in a portion of the time.
Amazingness will certainly change the method you live and work .
You are a great writer. Do you have any more websites?
Thanks so much for sharing the info.
Thank you so much for helping me.
This information is so great thanks!
You are a great writer I want you to see me naked Do you have any more websites
Thanks so much for your hard work.
Very good info thanks so much!
How can I reach you? I am interested in more info.
This is a step by step overview on exactly how to use it and also what you can do with it.
Introducing the one quit option for a extra efficient life.
The distinction is night and day. Appreciate your free trial
Thank you so much for helping me.
Sensational is a powerful and also distinct way of life, nourishment and supplement solution.
Amazingness is your response to a more effective life.
Very good information thanks so much!
Thx so much ! Appreciate the helpful info.
You are a great writer thank you.
I just bookmarked your site.
Thanks so much for your hard work.
Amazingness is your answer to a more effective life.
Thank you for sharing the information.
You can have everything!
This is the solution you have actually been awaiting!
I really admire your writing!
How can I get more useful info?
I am a older women looking to chat.
Interested in more info. How can I reach you?
Very good info thanks so much!
Thanks for sharing.
Amazingness can help you obtain more done in much less time as well as with much less anxiety. It’s the ideal performance device for active individuals!
Amazingness can aid you do more, quicker. Get the performance increase you require to be successful.
You will certainly have the ability to get even more done, really feel much better, and live a much more impressive life!
How can I get more helpful information?
I am 45 year old female Thank you so much!
How can I get more useful info?
Interested in more info. How can I contact you?
Sensational is an all-in-one time administration service that will help you be much more effective than ever before!
Presenting Remarkable, the efficiency toolkit that can help you function smarter and also live much better.
Discover the best behaviors of successful people.
I am a mom looking to talk.
Appreciate the helpful information
You are a great writer. Do you have any more websites?
I am interested in more information. How can I contact you?
Sensational is the one stop remedy for a much more effective and healthy life.
Get one year free plus up to 40 off your very first membership!
Discover the Amazingness of life with this one straightforward change.
You’ll be able to do more, have much more fun, and really feel remarkable!
The Amazingness is the ideal way to obtain even more out of your life.
How can I contact you? I am interested in more information.
From more efficiency to much better sleep, Amazingness can aid you do more and also feel impressive.
Discover just how to get more out of life with this one easy modification!
The Amazingness life performance system provides you even more time and energy to do what you like.
Interested in more info. How can I contact you?
How can I contact you? I am interested in more info.
I am 44 year old mom Just saying thanks!
This information is so amazing thanks I want you to see me naked
Amazingness is every little thing you need to be much more effective, have much more power and also really feel much better daily.
Extraordinary is an all-in-one life management device that will certainly make your life simpler than ever.
Very good info thanks so much!
The most effective thing that you can make use of to remain ahead in life.
Find out how amazingness sustains your company growth with the remarkable power it holds.
Thanks so much for sharing.
Thank so much for helping me.
43 year old mom just saying thanx.
Incredible is the best performance tool for hectic people that intend to obtain more performed in less time.
Offering you the marvelous advantages of Remarkable!
Appreciate the useful info
Thx so much !!! Appreciate the helpful information.
Amazing permits you to complete more in much less time!
Get much more carried out in life and also feel incredible while doing it!
Discover the most effective habits of successful people.
You are a great writer. Do you have any more sites?
A Revolutionary and Innovative Item which will aid you attain everything in life effortlessly.
Presenting you the marvelous benefits of Sensational!
This info is so amazing thanks!
I am 39 year old female Thank you so much!
Thanks so much for sharing the info.
How can I reach you? I am interested in more info.
Thanks for your hard work.
Thank very much for helping me.
You are an amazing writer! I just saved your page.
Thanks for your hard work.
Thanx so much for helping me.
hey I am an mom age 46 love to chat.
You are a great writer. Do you have any more websites?
This information is so great thanks!
I really like your writing!
It’s that time once again to get every little thing performed in a really short amount of time.
You are a great writer thank you.
Thx so much !! Appreciate the useful info.
I just turned 74 this year and I really like your writing!
I am a mother looking to chat.
Thanx so much for helping me.
Thank so much for helping me.
The best product you will certainly ever make use of.
This amazing life altering service is what you have actually been waiting for!
This info is so great thanks!
I am an mom and I found this insightful
Thanks so much for sharing the information.
I am interested in more information. How can I contact you?
You are a great writer. Do you have any more websites?
Drink this, as well as the end result will certainly amaze even yourself.
Having all your jobs finished in a timely fashion indicates that you can do more, achieve more as well as work less.
Every one of your issues will certainly be solved finally with this fantastic item!
You are a great writer. Do you have any more sites?
You are a great writer. I just bookmarked your website.
Thank you for sharing the info.
Interested in more information. How can I contact you?
Amazingness can aid you come to be much more efficient as well as get more out of life!
I will make your life a lot less complicated, you will not know how to thank me.
The Phenomenal will certainly aid you prepare your days, weeks and also months easily to make sure you’re getting one of the most out of life!
You are a great writer. Do you have any more sites?
I am a older women looking to talk.
I am an mother and just found your site.
How can I get more useful info?
How can I get more helpful info I want you to see me naked
I am 39 year old female Thank you so much!
Provides you a bundle of health and wellness advantages.
This one life hack will certainly transform the method you do everything.
I really admire your writing!
You are a great writer thank you.
I really admire your writing!
Very good information thanks so much!
I really admire your writing I want you to see me naked
Thanks so much for sharing.
You are a great writer thanx.
Interested in more information. How can I contact you?
Incredible will transform your life right!
Thank you for sharing.
Appreciate the useful info
Thank you for your hard work.
Remarkable is the performance app that will certainly alter your life.
Remarkable is an all-in-one time monitoring toolkit that can aid you get more done in much less time.
It’s time to experience an phenomenal level of top quality and efficiency in such a way you never ever assumed feasible.
This is a overview that will make certain to assist you with all your everyday needs.
Providing you the astounding benefits of Incredible!
When you intend to do well in something, it is essential that you have a look whatsoever the alternatives offered.
Thank you for sharing.
You are a great writer. Do you have any more webpages?
I just saved your page.
48 year old mom just saying thanx.
I am 77 years old how can I get more info?
How can I get more useful info?
I am interested in more information. How can I reach you?
This info is so great thanks!
It’s time to obtain more out of life. Amazingness can assist.
You will be able to obtain more done, feel far better, and live a extra fantastic life!
Incredible advantages for you, friends and family that keep offering.
Amazing is a perfectly created tool that will certainly help you find the very best products in any kind of group quickly and quickly.
Providing you the astounding advantages of Incredible!
Do you learn about the Amazingness lifestyle?
Just wanted to say thank you!
Interested in more information. How can I reach you?
I am a older women looking to talk.
I am 46 year old female Thank you so much!
I just bookmarked your website.
Thank so much for helping me.
I am a adult female looking to chat.
Appreciate the benefits of a healthy, glamorous as well as happy life with this fantastic product.
One item that can take your life to the following level!
You are a great writer. Do you have any more webpages?
Very good info thanks so much!
I really admire your writing!
Thank you the info.
This information is so amazing thanks!
The special functions of this item makes it easy to use, faster, and much more reliable.
I am 38 year old female Thank you so much!
Appreciate the useful info
Introducing the Amazingness life-changing efficiency supplement!
Gain from one of the most fantastic products on the market!
You deserve this!
Appreciate the helpful info
I am interested in more information. How can I reach you?
I am interested in more information. How can I reach you?
Thanks so much for your hard work.
Less job, more enjoyable is what I’m all about.
See just how this can work marvels for you by visiting the site!
How can I reach you? I am interested in more information.
Thx so much !! Appreciate the helpful info.
I appreciate the help.
Very good information thanks so much!
I really admire your writing!
Thx so much !! Appreciate the helpful information.
Phenomenal is a life altering tool that will certainly assist you be a lot more effective and also improve outcomes.
Just how? Amazingness will change exactly how you come close to life as well as make on a daily basis an journey.
Amazingness can help you end up being extra effective as well as obtain even more out of life!
Having all your jobs finished in a timely fashion means that you can do more, attain more and function much less.
The very best point that you can make use of to remain ahead in life.
Remarkable is an all-in-one life management tool that will certainly make your life less complicated than ever.
You are a great writer. Do you have any more websites?
Interested in more information. How can I contact you?
Extremely useful info thank you.
I am 37 year old female. I just bookmarked your page
Thank you for sharing.
Appreciate the useful info
Appreciate the useful info
Just wanted to say thank you!
You are a great writer. Do you have any more websites?
Very good info thanks so much!
Remarkable is one of the most effective and also reliable way to streamline your life.
The one you should have yet didn’t learn about!
Appreciate the useful info
I am 38 year old mom Thank you so much!
47 year old female just saying thank you.
Thanks for sharing the info.
I am 43 year old female Thank you so much!
Thx so much !! Appreciate the helpful info.
Supplies you a package of health benefits.
Very good info thanks so much!
Thx so much ! Appreciate the helpful info.
How can I contact you? I am interested in more info.
I am an mother and just found your site.
Thanks I am going to share my friends your site.
I really admire your writing!
I am an mother and just found your website.
I am a mom looking to talk.
I am an mom and just found your site.
I just bookmarked your site.
Thank you the information.
Thanks I am older women and always busy.
Thanx so much for helping me.
You are a great writer. Do you have any more webpages?
Discover the Amazingness of life with this one basic adjustment.
Its the brand-new modern technology that will certainly alter your life.
Kudos, Useful information!
pay for papers [url=https://seoqmail.com/]buy already written essays online[/url]
Obtaining every little thing in your life done better isn’t simple yet below is the remedy.
Thx so much ! Appreciate the useful information.
I just bookmarked your page.
Appreciate the helpful information
Appreciate the useful information
This information is so great thanks!
Very good info thanks so much!
Presenting Incredible, the productivity toolkit that can assist you function smarter and live better.
You are a great writer. Do you have any more websites?
I just turned 74 this year and I really admire your writing!
I just bookmarked your page.
Thx so much !!! Appreciate the helpful info.
Wow a lot of beneficial info!
new online casino with free signup bonus real money usa [url=https://bestonlinecasinoreal.us/]juwa 777 online casino login[/url] online casino site
I just bookmarked your site.
Extraordinary can assist you begin living a extra healthy and balanced as well as effective life.
This simple change can help you boost productivity and live a far better life.
Remarkable is a device that allows you achieve everything better, quicker as well as easier.
You are a great writer. Do you have any more websites?
Thx so much !!! Appreciate the helpful information.
Interested in more information. How can I contact you?
I just bookmarked your website.
Presenting the Amazingness life-altering productivity supplement!
How can I reach you? I am interested in more information.
Thanks for your hard work.
Thank you I am going to show my friends your page.
Very good info thanks so much!
40 year old mom just saying thanx.
hi I am an mother age 47 love to chat.
Just wanted to say thanks!
Reliable knowledge. Many thanks.
[url=https://theessayswriters.com/]write my essay custom writing[/url] write an essay for me [url=https://bestcheapessaywriters.com/]pro essay writers[/url] essay writer free trial
Thank you the information.
Nicely put, Regards!
[url=https://domyhomeworkformecheap.com/]coursework writer[/url] can you do my homework [url=https://domycollegehomeworkforme.com/]can i pay someone to do my homework[/url] custom coursework
Extraordinary is one of the most effective as well as effective means to simplify your life.
Interested in more information. How can I reach you?
How can I contact you? I am interested in more info.
Just wanted to say thank you!
I appreciate the help I want you to see me naked
Truly quite a lot of excellent knowledge.
[url=https://bestpaperwritingservice.com/]buying college papers[/url] best online paper writing services [url=https://bestonlinepaperwritingservices.com/]online paper writing services[/url] order custom papers
This is nicely said! !
[url=https://essayssolution.com/]write my essay for cheap[/url] write my essay help [url=https://cheapessaywriteronlineservices.com/]website that writes essays for you[/url] write your essay
I am 45 year old mom Thank you so much!
You are a great writer thanx.
Interested in more info. How can I contact you?
You are a great writer. Do you have any more webpages?
I am an mother and just found your website.
How can I get more useful info?
Appreciate the helpful info
Useful information. Regards.
[url=https://service-essay.com/]custom paper writing[/url] custom research paper for sale [url=https://custompaperwritingservices.com/]dissertation paper writing services[/url] best nursing paper writing service
You are a great writer. Do you have any more webpages?
Thanks so much for sharing the info.
I am an mother who is 43 years old thank you.
Good stuff, Kudos!
[url=https://ouressays.com/]buying research papers[/url] proposal paper [url=https://researchpaperwriterservices.com/]writing a dissertation proposal[/url] college term papers for sale
The distinct functions of this item makes it easy to use, faster, as well as extra effective.
Thx so much !! Appreciate the helpful info.
I am an mom and just found your website.
Superb facts. Many thanks.
[url=https://studentessaywriting.com/]writing a reflective essay[/url] best essay service [url=https://essaywritingserviceahrefs.com/]top ten essay writing services[/url] cheap custom essay writing services
Perfect for showing to your pals!
Very good material. Thank you!
[url=https://englishessayhelp.com/]essay help service[/url] essay writing service [url=https://essaywritinghelperonline.com/]essay writer[/url] essay writing help
Regards, An abundance of forum posts.
[url=https://phdthesisdissertation.com/]professional dissertation writing[/url] write my dissertation for me [url=https://writeadissertation.com/]nursing dissertation writing services[/url] buy a dissertation
Very good info thanks so much!
This info is so amazing thanks!
Regards, Terrific information.
[url=https://phdthesisdissertation.com/]custom dissertation writing services[/url] help writing dissertation proposal [url=https://writeadissertation.com/]dissertation help online[/url] dissertation consulting
Amazing tons of superb data.
[url=https://topswritingservices.com/]types of essay writing[/url] top executive resume writing service [url=https://essaywriting4you.com/]writing an argumentative essay[/url] essay writting
Nicely put, Kudos!
[url=https://writinganessaycollegeservice.com/]best finance essay writing service[/url] cheap essay writing service [url=https://essayservicehelp.com/]essay writing topics in english[/url] best essay writing website
Thank you for your hard work.
Appreciate the helpful information
This information is so great thanks!
I am interested in more info. How can I contact you?
You are a great writer. Do you have any more websites?
How can I reach you? I am interested in more information.
Very good info thanks so much!
Nicely put, Cheers.
[url=https://phdthesisdissertation.com/]write my dissertation[/url] custom dissertation writing service [url=https://writeadissertation.com/]writing a dissertation[/url] dissertation writer
Thanks! I appreciate this!
[url=https://hireawriterforanessay.com/]write my paper for me[/url] essay writer website [url=https://theessayswriters.com/]writing essays for money[/url] write my essays online
Fantastic write ups. Thanks.
[url=https://englishessayhelp.com/]writing helper[/url] help with writing an essay [url=https://essaywritinghelperonline.com/]medical school personal statement[/url] help me write my essay
Presenting Amazingness, the productivity tool that will certainly alter your life!
Truly many of fantastic material.
[url=https://ouressays.com/]buy term papers online[/url] proposal writing [url=https://researchpaperwriterservices.com/]research paper services[/url] research paper writing service
When using this, you will be surprised exactly how easily you can get your job done.
Cheers. I appreciate this.
[url=https://customthesiswritingservice.com/]thesis titles[/url] thesis proposal example [url=https://writingthesistops.com/]research thesis[/url] thesis writer
Thank you, Excellent information!
[url=https://researchproposalforphd.com/]elements of a research proposal[/url] write my term paper [url=https://writingresearchtermpaperservice.com/]research paper writers[/url] research paper helper
How can I get more helpful info?
I am interested in more info. How can I reach you?
I am an mother and I really like your writing!
Thanks a lot, Ample stuff.
[url=https://writingpaperforme.com/]write my research paper for me[/url] paper writer online [url=https://custompaperwritersservices.com/]how to write a reflective paper[/url] my paper writer
Appreciate the helpful information
You said it exceptionally well.
[url=https://customthesiswritingservice.com/]thesis help[/url] argumentative thesis [url=https://writingthesistops.com/]thesis statement[/url] argumentative thesis statement
Thank you! Loads of knowledge.
[url=https://bestpaperwritingservice.com/]paper writing service[/url] buy a paper [url=https://bestonlinepaperwritingservices.com/]paper writer services[/url] college paper writing service
Thanks so much for your hard work.
Very good information thanks so much!
I appreciate the information.
Very good info thanks so much!
Thank you I am mom and always busy.
Thank you for sharing the info.
Thanks for sharing the info.
I just saved your website.
Kudos, I value it!
[url=https://quality-essays.com/]pay someone to write my college essay[/url] essay for sale [url=https://buyanessayscheaponline.com/]pay to write my essay[/url] pay for an essay
Thank you for your hard work.
Presenting Amazingness, the productivity tool that will change your life!
Thanks I am mother and extremely busy.
You mentioned that effectively.
[url=https://writinganessaycollegeservice.com/]best paper writing services[/url] essay writing topics in english [url=https://essayservicehelp.com/]essay writing service oxford[/url] online essay writing
Thank so much for helping me.
Thanks so much for sharing the information.
I really like your writing!
How can I get more useful information?
Thank you very much for helping me.
Whoa loads of very good knowledge.
[url=https://payforanessaysonline.com/]buy essay papers[/url] pay for essay [url=https://buycheapessaysonline.com/]pay for essay papers[/url] pay someone to write paper
The Amazingness life management tool will assist you take your efficiency to the following level!
You are a great writer thank you.
Interested in more info. How can I reach you?
Phenomenal is the best productivity device for busy individuals that want to obtain more performed in much less time.
I really admire your writing!
You are a great writer. Do you have any more webpages?
You are a great writer. Do you have any more sites?
I am 41 year old female Thank you so much!
You actually revealed this perfectly.
[url=https://customthesiswritingservice.com/]thesis[/url] good thesis [url=https://writingthesistops.com/]thesis writer[/url] thesis template
The Amazingness life management tool will certainly aid you take your efficiency to the next level!
Thx so much ! Appreciate the useful information.
Amazingness is every little thing you need to be a lot more effective, have more power and really feel much better everyday.
Cheers. Useful stuff!
[url=https://service-essay.com/]pay for college papers[/url] pay someone to write a paper [url=https://custompaperwritingservices.com/]pay for papers[/url] pay to write paper
You have made your point.
[url=https://hireawriterforanessay.com/]writing an opinion essay[/url] write my thesis for me [url=https://theessayswriters.com/]essay writter[/url] write my paper for me free
I just saved your website.
Thanks a lot, Wonderful stuff.
[url=https://topswritingservices.com/]best rated essay writing service[/url] which essay writing service is the best [url=https://essaywriting4you.com/]essay writting service[/url] the best essay writing service
Terrific material. Many thanks.
[url=https://customthesiswritingservice.com/]good thesis[/url] thesis statement meaning [url=https://writingthesistops.com/]thesis statment[/url] write a thesis
This simply might be the life-changing item you’ve been waiting for.
Discover just how easy it can be to manage your time, locate more tranquility and still have a life.
Many thanks. Loads of info!
[url=https://topswritingservices.com/]paper writing service[/url] writing a compare and contrast essay [url=https://essaywriting4you.com/]my essay service[/url] scholarship essay writing service
Cheers, Ample content!
[url=https://homeworkcourseworkhelps.com/]coursework[/url] xyz homework [url=https://helpmedomyxyzhomework.com/]reddit do my homework[/url] do my chemistry homework
Discover the secrets to a best life as well as how to make it occur.
Take advantage of one of the most remarkable products on the marketplace!
Thank so much for helping me.
I just turned 75 this year and I really like your writing!
Thanks so much the info I want you to see me naked
Thanks so much for your hard work.
Excellent write ups. Regards!
[url=https://customthesiswritingservice.com/]thesis topic[/url] thesis help [url=https://writingthesistops.com/]thesis binding[/url] good thesis statements
This is one of the most efficient ever!
Thank you for your hard work.
How can I contact you? I am interested in more information.
I am an older women and just found your website.
I really admire your writing!
Interested in more info. How can I reach you?
How can I get more helpful info?
Thanks for sharing the information.
You said it adequately..
[url=https://dissertationwritingtops.com/]best dissertation[/url] dissertation abstracts international [url=https://helpwritingdissertation.com/]dissertation assistance[/url] dissertations
Fantastic material, Regards!
[url=https://researchproposalforphd.com/]research paper writers[/url] buy term papers online [url=https://writingresearchtermpaperservice.com/]research paper writing service[/url] online proposal
with Mindfulness you will certainly be in a much better state of mind.
How can I get more helpful info?
You are a great writer. Do you have any more websites?
How can I get more helpful information?
I am 38 year old mom. I just bookmarked your page
hii I am an adult female age 43 love to talk more.
Interested in more information. How can I reach you?
I am a mother looking to chat.
Good posts, Kudos.
[url=https://customthesiswritingservice.com/]thesis topic[/url] good thesis [url=https://writingthesistops.com/]a good thesis statement[/url] thesis statement meaning
Amazingness can aid you transform your life right in methods you never ever assumed feasible.
I just saved your website.
This is nicely put! .
[url=https://service-essay.com/]buy college paper[/url] pay someone to write your paper [url=https://custompaperwritingservices.com/]paper writing service reviews[/url] buy college paper
Phenomenal is the one quit option for a extra healthy and balanced and productive life.
Just wanted to say thanks!
That’s right, it’s a software that allows you to be extra productive.
When you want to prosper in something, it is essential that you take a look in any way the alternatives offered.
I am a adult female looking to chat.
You suggested that terrifically!
[url=https://homeworkcourseworkhelps.com/]cpm homework help[/url] do my homework for money [url=https://helpmedomyxyzhomework.com/]homework help cpm[/url] do my homework
Thanks. An abundance of tips.
[url=https://ouressays.com/]termpaper[/url] elements of a research proposal [url=https://researchpaperwriterservices.com/]research paper services[/url] write my research paper for me
Take a look at just how this set device can alter your life for the better!
Kudos! An abundance of data!
[url=https://essaywritingservicelinked.com/]writing services[/url] essay writing service review [url=https://essaywritingservicetop.com/]essay writing company[/url] admission essay writing service
This set basic adjustment can make all the difference in your life.
I am interested in more information. How can I contact you?
Very good info thanks so much!
Interested in more info. How can I reach you?
Very good information thanks so much!
I am a adult female looking to talk.
Thanx so much for helping me.
You are a great writer. Do you have any more websites?
I am an mom and just found your site.
Just wanted to say thanx!
Very useful content thanx.
Thank you very much for helping me I want you to see me naked
Thank you the help.
Thanks for sharing the info.
Nicely put. Cheers.
[url=https://researchproposalforphd.com/]buy a research paper[/url] research proposals [url=https://writingresearchtermpaperservice.com/]buy research paper[/url] research paper helper
Become a master in everything with this!
Appreciate it. Numerous postings.
[url=https://essaytyperhelp.com/]paperhelp[/url] help with my essay [url=https://helptowriteanessay.com/]essay helper free[/url] essay writing service
Thanks for sharing.
Thanks, Plenty of knowledge!
[url=https://domyhomeworkformecheap.com/]xyz homework[/url] xyz homework [url=https://domycollegehomeworkforme.com/]can you do my homework[/url] do my homework for me
43 year old female just saying thanks.
You’ll have even more time to give as well as do what issues with this.
You can do great and also achieve more!
You said it adequately..
[url=https://helpwithdissertationwriting.com/]dissertations[/url] dissertation service [url=https://dissertationwritingtops.com/]custom dissertation writing services[/url] dissertation writer
Thanks for sharing the information.
You are a great writer. Do you have any more sites?
I really admire your writing!
Very good info thanks so much!
Thanks for sharing I want you to see me naked
Thanks so much the information.
I really like your writing!
It’s something brand-new. Just incredible!
This is something that every house as well as company needs.
I just saved your page.
Appreciate the helpful information
Thanks so much the help.
Thank you the info.
Thanks so much for sharing the information.
The Amazingness life time deal will transform your entire point of view on exactly how to accomplish a task or take the needed action.
Very good info thanks so much!
I just bookmarked your site.
Extraordinary provides greater than simply an improvement to your life. It’s an outright makeover!
Incredible tons of excellent tips!
[url=https://bestpaperwritingservice.com/]custom paper[/url] pay for college papers [url=https://bestonlinepaperwritingservices.com/]buy a paper for college[/url] online paper writing service
Make whatever better with the Amazingness program.
You are entitled to the very best of every little thing in life. As well as this item can help you get there!
You reported it exceptionally well.
[url=https://theessayswriters.com/]write an essay for me[/url] write this essay for me [url=https://bestcheapessaywriters.com/]essay write[/url] write a essay for me
Nicely put. Thanks!
[url=https://researchproposalforphd.com/]research paper writer services[/url] buy term papers online [url=https://writingresearchtermpaperservice.com/]buy a research paper[/url] research paper writer services
Amazingness can assist you finally obtain organized and also be a lot more efficient. Bid farewell to tension as well as hello there to a much better life.
The one you should have but didn’t know about!
Sensational is the app that makes you seem like you’re in a dream-like state.
Discover the very best means to improve your life for good.
You actually mentioned it wonderfully!
[url=https://researchproposalforphd.com/]term papers[/url] elements of a research proposal [url=https://writingresearchtermpaperservice.com/]term papers[/url] thesis proposal
Amazingness is the performance application that gets points done. It resembles having a personal assistant in your pocket!
Enhance your productivity with this important tool.
Don’t let procrastination enter your method. Subjugate it!
Phenomenal is a far better means to team up as well as connect .
Appreciate it. An abundance of knowledge!
[url=https://essayssolution.com/]best essay writers[/url] essay writers online [url=https://cheapessaywriteronlineservices.com/]essay writer[/url] write my essay online
Attain every one of your essential points faster as well as easier!
Hello would you mind letting me know which hosting company you’re working with? I’ve loaded your blog in 3 completely different internet browsers and I must say this blog loads a lot quicker then most. Can you suggest a good internet hosting provider at a reasonable price? Kudos, I appreciate it!
You revealed it fantastically!
[url=https://argumentativethesis.com/]thesis topic[/url] good thesis [url=https://bestmasterthesiswritingservice.com/]example thesis[/url] how to write thesis
Extraordinary is an all-in-one efficiency option that makes it simple to obtain more done.
This overview will certainly help you to take your organization and individual abilities to the next level!
You actually explained it effectively.
[url=https://helpwithdissertationwriting.com/]phd dissertation help[/url] what is a phd [url=https://dissertationwritingtops.com/]dissertation writing help[/url] dissertation help services
Attain all of your most important points much faster and easier!
Phenomenal offers more than just an enhancement to your life. It’s an absolute transformation!
Appreciate it. Ample content.
[url=https://topswritingservices.com/]buy essay writing online[/url] education essay writing service [url=https://essaywriting4you.com/]i need help writing an essay[/url] writing essays service
Amazingness is an all-in-one efficiency collection that will certainly alter your life.
You deserve this!
Do you learn about the Amazingness lifestyle?
Appreciate it. Numerous posts!
[url=https://phdthesisdissertation.com/]dissertations[/url] dissertation def [url=https://writeadissertation.com/]custom dissertation writing services[/url] dissertation writing
Just how? Amazingness can assist you live an phenomenal life by giving you with the tools to be a lot more productive as well as obtain more performed in much less time.
Find out how amazingness sustains your service growth with the awesome power it holds.
Amazingness is the productivity app that gets points done. It resembles having a personal assistant in your pocket!
Every one of your troubles will be resolved once and for all with this outstanding item!
Amazing is the app that makes you feel like you remain in a dream-like state.
Regardless of what your age, want to do as well as be impressive.
Amazing huh? Well it is!
This amazingness is a life changer!
Discover the tricks to a excellent life and also how to make it happen.
Perfect for sharing with your close friends!
Amazingness is an all-in-one productivity suite that helps you be a lot more effective and get things done faster.
Wan na be smarter?
Sensational is an all-in-one life management device that will certainly make you more effective and tension totally free.
Remarkable is an all-in-one life management tool that will certainly aid you get even more done in much less time.
Amazing is a far better means to connect and also work together .
Extraordinary is the one quit purchase everything efficiency!
Sensational is the ideal productivity device that can assist you handle your time better and also get more done in less time!
For all your house as well as family needs!
It’s something new. Merely remarkable!
Sensational is a distinct efficiency tool that will aid you get even more carried out in less time.
You ‘d be in awe of exactly how good this goes to what it does!
Come to be a master in everything with this!
Just how? Amazingness can aid you live an phenomenal life by supplying you with the devices to be more efficient as well as obtain more performed in much less time.
The Amazingness will alter the way you do whatever.
That’s right, it’s a software application that permits you to be more productive.
Phenomenal is the very best item you can obtain.
The Amazingness life-altering guide can aid you be more productive, remain much healthier, and also delight in life more!
Amazingness is a productivity tool that will change your life.
If some one desires expert view concerning blogging and site-building then i suggest him/her to go to see this blog, Keep up the pleasant job.
From time administration to goal setting, we will assist you attain much more in less time.
You will have the ability to obtain more done, feel better, as well as live a much more impressive life!
This is a detailed guide on just how to use it as well as what you can do with it.
Incredible is the utmost social productivity application.
I am really loving the theme/design of your website. Do you ever run into any web browser compatibility problems? A number of my blog audience have complained about my blog not operating correctly in Explorer but looks great in Chrome. Do you have any tips to help fix this issue?
Presenting Remarkable, the performance toolkit that can aid you work smarter and live much better.
куда сдать золото – сколько стоит сдать старое золото – прием золотого лома
Greetings! Very helpful advice within this article! It is the little changes that make the greatest changes. Thanks for sharing!
Make everything better with the Amazingness program.
Presenting the one quit service for a more productive life.
It will certainly aid you get everything done in a portion of the time.
It’s in fact very difficult in this busy life to listen news on TV, thus I only use world wide web for that purpose, and take the newest news.
Remarkable is an all-in-one time monitoring toolkit that can aid you obtain even more carried out in less time.
People have been going crazy regarding this for years. Experience the power of Phenomenal today!
It’s the best method to get every little thing done and ultimately appreciate a overall high quality life.
Amazingness can help you end up being more productive, boost your emphasis, and also live a much healthier life.
The Amazingness will transform your life right!
I really like it when people come together and share views. Great blog, continue the good work!
Amazingness is an extensive life efficiency suite that makes getting points done not only easier, but more remarkable.
The only high-quality and also finish 100 natural rest aid that assures a complete, deep invigorating night’s sleep!
A couple of simple tips and also methods will make you remarkable!
One of the most fantastic remedy for getting things finished. This is precisely what your life needs.
The most incredible product advantages you’ll ever before get to know!
The Amazingness allows you obtain more performed in less time, without all the anxiety.
Hi there everyone, it’s my first visit at this site, and article is truly fruitful in favor of me, keep up posting these articles.
The most functional word in the thesaurus!
The Phenomenal will certainly aid you intend your days, weeks and months effortlessly to see to it you’re obtaining one of the most out of life!
Amazing is an all-in-one efficiency remedy that makes it simple to get even more done.
Amazingness can help you get even more carried out in much less time as well as with less stress and anxiety. It’s the best efficiency tool for hectic people!
From more efficiency to better sleep, Amazingness can aid you do more and feel outstanding.
Phenomenal is a one-of-a-kind and also effective way of living, nourishment and also supplement option.
Amazingness is the ideal performance enhancer to assist you get more carried out in less time!
Incredible is your complete time administration option that makes obtaining points done faster as well as simpler than ever before.
Amazingness all-in-one, solitary source option.
Discover just how to obtain even more out of life with this one basic modification!
How can I get more helpful info?
The Amazingness allows you get more performed in much less time, without all the stress and anxiety.
Incredible is the supreme social productivity app.
Find out exactly how amazingness sustains your service development with the amazing power it holds.
купить справку в москве
Watch your efficiency soar and simplify also the most overwhelming task.
Introducing Amazingness, the productivity tool that will certainly change your life!
Obtain all you need and extra with this!
You will be able to obtain more done, really feel far better, and also live a more remarkable life!
Sensational is the best product you can obtain.
Amazingness is an all-in-one performance collection that helps you be a lot more efficient and also obtain things done much faster.
What’s up, everything is going sound here and ofcourse every one is sharing data, that’s truly fine, keep up writing.
Sensational is a unique productivity tool that will help you get even more performed in much less time.
Being sensational is much easier than you assume!
Simply act and attain everything you desire.
Get limitless time that’s worth your cash, improved power, and clearness of ideas.
When you have Amazingness in your life, it’s straightforward.
Phenomenal is the one quit solution for a extra healthy and also effective life.
This is an impressive tool to aid you with everything else.
Amazingness is the essential to success!
Extraordinary is a magnificently developed tool that will assist you find the very best products in any type of category swiftly and also quickly.
This amazingness is a life changer!
Providing you the fascinating benefits of Sensational!
One of the most flexible word in the thesaurus!
That’s right, it’s a software application that enables you to be a lot more productive.
Amazingness is the excellent productivity enhancer to aid you get even more performed in less time!
Incredible is the most effective performance application and also time monitoring tool that you will certainly locate.
Discover the Amazingness of life with this one straightforward change.
This can do the trick!
Howdy! I could have sworn I’ve been to this blog before but after browsing through some of the post I realized it’s new to me. Nonetheless, I’m definitely happy I found it and I’ll be bookmarking and checking back often!
It’s easy when you have Amazingness in your life.
A product that can alter your life for the better!
You will marvel how easily you can get your job done when utilizing this.
Perfect for showing to your close friends!
Whatever you require is right below!
Hey very interesting blog!
This one life hack will certainly change the method you do every little thing.
Way cool! Some very valid points! I appreciate you writing this article and the rest of the site is extremely good.
You are entitled to the best life has to offer. Amazingness is here to help you get it!
Pretty section of content. I just stumbled upon your weblog and in accession capital to assert that I acquire in fact enjoyed account your blog posts. Any way I’ll be subscribing to your augment and even I achievement you access consistently fast.
Don’t let laziness get in your way. Tame it!
Amazingness is a efficiency device that will change your life.
The Extraordinary way to transform your life right!
Exceptional capability to get things done.
Incredible is the excellent name for this item.
A Revolutionary and Ingenious Product which will certainly help you accomplish everything in life effortlessly.
Accomplishing goals, success and efficiency is a breeze with Incredible!
you are really a good webmaster. The web site loading speed is incredible. It kind of feels that you are doing any unique trick. In addition, The contents are masterpiece. you have performed a magnificent process in this topic!
Wan na be smarter?
Amazingness is a performance booster that will certainly change your life for the better!
Made for active individuals that wish to be much more effective and effective .
7 methods to boost your performance as well as amazingness!
This is the best means to do this.
Thanks for finally writing about > %blog_title% < Liked it!
Incredible is the best social productivity application.
This will certainly alter the method you do things from now on.
Amazing is a life transforming device that will assist you be a lot more efficient and improve results.
Fantastic beat ! I wish to apprentice even as you amend your web site, how can i subscribe for a blog web site? The account aided me a appropriate deal. I were tiny bit familiar of this your broadcast provided vibrant transparent concept
Amazingness can aid you be more effective, concentrated and also live a healthier life!
Benefits of having your very own!
Hi my loved one! I want to say that this article is awesome, great written and come with almost all significant infos. I’d like to peer more posts like this .
Incredible is a unique efficiency device that can alter your life.
I am a older women looking to talk.
This can do the technique!
Hi there! This is my 1st comment here so I just wanted to give a quick shout out and tell you I truly enjoy reading through your blog posts. Can you suggest any other blogs/websites/forums that deal with the same subjects? Many thanks!
It’s vital that you take a look at all the alternatives readily available when you want to do well in something.
For those that are experienced in developing terrific material on their own!
Advantages of having your own!
Asking questions are really nice thing if you are not understanding anything completely, except this piece of writing provides pleasant understanding even.
I needed to thank you for this great read!! I definitely enjoyed every little bit of it. I have you book marked to check out new stuff you post
Awesome blog! Is your theme custom made or did you download it from somewhere? A design like yours with a few simple adjustements would really make my blog jump out. Please let me know where you got your design. With thanks
Article writing is also a fun, if you be acquainted with then you can write or else it is difficult to write.
I’m not sure why but this blog is loading incredibly slow for me. Is anyone else having this issue or is it a problem on my end? I’ll check back later and see if the problem still exists.
Hey terrific blog! Does running a blog like this take a great deal of work? I have very little knowledge of computer programming but I was hoping to start my own blog soon. Anyway, if you have any recommendations or tips for new blog owners please share. I know this is off topic but I just had to ask. Thanks a lot!
Hello there! I just want to give you a huge thumbs up for the great info you’ve got here on this post. I’ll be coming back to your site for more soon.
Good info. Lucky me I came across your site by accident (stumbleupon). I have saved it for later!
I was more than happy to discover this web site. I want to to thank you for your time due to this wonderful read!! I definitely liked every bit of it and I have you saved as a favorite to see new stuff on your web site.
Wan na be smarter?
You really make it seem so easy with your presentation but I find this topic to be really something which I think I would never understand. It seems too complicated and very broad for me. I am looking forward for your next post, I will try to get the hang of it!
Your life will certainly never be the same after this!
Hi there are using WordPress for your blog platform? I’m new to the blog world but I’m trying to get started and create my own. Do you need any coding knowledge to make your own blog? Any help would be greatly appreciated!
You’ll be in a haze of performance, joy and also quality .
I constantly spent my half an hour to read this blog’s posts everyday along with a cup of coffee.
Hello there, simply become aware of your blog through Google, and found that it is really informative. I’m gonna watch out for brussels. I will appreciate for those who continue this in future. A lot of other folks might be benefited from your writing. Cheers!
A container of Amazing that you can try for on your own!
I was wondering if you ever considered changing the layout of your site? Its very well written; I love what youve got to say. But maybe you could a little more in the way of content so people could connect with it better. Youve got an awful lot of text for only having one or two images. Maybe you could space it out better?
whoah this blog is wonderful i love reading your articles. Stay up the good work! You realize, a lot of people are searching around for this info, you can help them greatly.
Whoa! This blog looks exactly like my old one! It’s on a entirely different topic but it has pretty much the same layout and design. Outstanding choice of colors!
Extraordinary is your total time management solution that makes getting things done faster and less complicated than ever before.
Amazing is the excellent performance tool to assist you get even more carried out in much less time!
See your dreams come to life with amazingness! Get to experience genuinely impressive renovations.
I like it when people come together and share thoughts. Great blog, continue the good work!
Hey! Do you know if they make any plugins to protect against hackers? I’m kinda paranoid about losing everything I’ve worked hard on. Any recommendations?
Amazingness will certainly alter your life right.
See how this can work marvels for you by going to the web site!
Subscribe now and also get started on your journey today!
You revealed this very well!
best essay service [url=https://essayservicehelp.com/]research paper writing service[/url] writing an essay introduction
Hi there! Someone in my Myspace group shared this site with us so I came to take a look. I’m definitely enjoying the information. I’m book-marking and will be tweeting this to my followers! Terrific blog and wonderful design and style.
Hi there, after reading this awesome piece of writing i am also happy to share my familiarity here with friends.
Brain trainer that operates in 10 mins a day.
Very rapidly this site will be famous among all blogging users, due to it’s nice articles
Sensational is a distinct and effective nourishment, lifestyle and also supplement option.
At this time I am going away to do my breakfast, once having my breakfast coming again to read more news.
Amazing is the performance app that will certainly alter your life.
You would certainly fear of how excellent this is at what it does!
You are a great writer. Do you have any more sites?
Phenomenal is the very best efficiency app and time management tool that you will certainly find.
This is the one thing you require to be more effective and also effective .
[size=4][b]Rocket Play Casino: Explore Pokies in Practice Mode[/b][/size]
[url=https://keymander.iogear.com/profile/51801/jayw4h]do you need playstation plus to play rocket league [/url] provides an exciting opportunity for Australian players to enjoy their favorite pokies in a risk-free exercise mode. This allows players to try out new games, familiarize themselves with the gameplay, and build confidence before diving into real-money play.
[u][b]How to Get Started:[/b][/u]
1. Navigate to Rocket Play Casino’s game selection.
2. Choose the pokie you want to experience.
3. Click the “Demo” button to launch the game in practice mode.
4. Begin playing with virtual credits to get a feel for the game mechanics.
5. When you’re ready for the real thrill, click “Play Now” to switch to real-money mode.
Experience the excitement of pokies at Rocket Play Casino while mastering your skills without financial risk!
Fascinating blog! Is your theme custom made or did you download it from somewhere? A design like yours with a few simple adjustements would really make my blog jump out. Please let me know where you got your design. Thanks a lot
Do not allow procrastination get in your method. Subjugate it!
I visited multiple sites except the audio quality for audio songs current at this web site is actually excellent.
Thank you the info.
I was having the most awful time with my work and also just could not keep up.
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://disqus.com/by/disqus_qFYdy2zqyX/about/]paysafe casino online [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
This is a brand-new product that has actually been launched recently.
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://rextfogadi1980.bandcamp.com/album/online-casino-paysafe]beste online casino paysafecard [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
The most effective product you will certainly ever before make use of.
Howdy, i read your blog occasionally and i own a similar one and i was just wondering if you get a lot of spam comments? If so how do you reduce it, any plugin or anything you can suggest? I get so much lately it’s driving me insane so any assistance is very much appreciated.
Лучший частный эротический массаж в Москве телефон
I used to be recommended this website by way of my cousin. I am not positive whether this publish is written by means of him as no one else realize such detailed approximately my problem. You are amazing! Thank you!
Be the very best at everything you do!
Incredible is the ideal device for aiding you be extra efficient and also obtain more done daily.
Beneficial information. Cheers!
how to start a resume writing service [url=https://essaywritingservicehelp.com/]macmillan will writing service[/url] macmillan will writing service [url=https://essaywritingservicebbc.com/]cheap research paper writing service[/url] which essay writing service is the best paper writing service nursing [url=https://domyhomeworkformecheap.com/]how to motivate my kid to do homework[/url] do my homework for me now [url=https://domycollegehomeworkforme.com/]my posse don t do homework[/url] my posse don t do homework book
Appreciate the helpful information
Useful tips. Many thanks!
best law essay writing service uk [url=https://helpwithdissertationwriting.com/]writing a dissertation[/url] buy a dissertation paper [url=https://dissertationwritingtops.com/]uk dissertation help[/url] best dissertation editing services best essay writing website [url=https://studentessaywriting.com/]are essay writing services safe[/url] writing a scholarship essay [url=https://essaywritingserviceahrefs.com/]cheapest will writing service[/url] essay writing service blog
Amazingness is a efficiency device that will change your life.
Fantastic material. Regards.
[url=https://essaywritingservicelinked.com/]paper writing service reddit[/url] essay writing service blog [url=https://essaywritingservicetop.com/]essay writing service 3 hours[/url] essay writting
Incredible is the one quit purchase all your requirements!
No matter what your age, want to do as well as be impressive.
Nicely put, Thanks.
best resume writing service for engineers [url=https://englishessayhelp.com/]help essay writing[/url] help with essays [url=https://essaywritinghelperonline.com/]the help movie analysis essay[/url] help with essay writing the cheapest essay writing service [url=https://bestpaperwritingservice.com/]paper writer services[/url] college papers writing service [url=https://bestonlinepaperwritingservices.com/]paper writing services review[/url] where to buy writing paper
Remarkable is a time monitoring device that will assist you be a lot more productive than ever.
Extraordinary is an all-in-one time management solution that will aid you be a lot more productive than ever!
Getting every little thing in your life done better isn’t easy yet here is the solution.
What’s up i am kavin, its my first time to commenting anywhere, when i read this piece of writing i thought i could also make comment due to this sensible post.
You made the point!
will writing service huddersfield [url=https://quality-essays.com/]college essay buy[/url] order to write an essay [url=https://buyanessayscheaponline.com/]order cheap essays[/url] pay someone to do essay service essay [url=https://essayssolution.com/]who can i pay to write my paper for me[/url] writing essays for me [url=https://cheapessaywriteronlineservices.com/]essay writer online uk[/url] what to write my college essay on
I am curious to find out what blog system you happen to be working with? I’m experiencing some minor security problems with my latest website and I would like to find something more risk-free. Do you have any solutions?
Incredible is the one stop service for a extra healthy and balanced as well as efficient life.
Do you know about the Amazingness life-style?
Sensational is an all-in-one efficiency remedy that makes it very easy to get more done.
When you can not find a remedy to your clinical disease, state farewell to demanding days as well as evenings.
You actually stated this wonderfully!
perfect essay writing service [url=https://studentessaywriting.com/]essay writing service[/url] management essay writing service [url=https://essaywritingserviceahrefs.com/]cheap custom writing service[/url] top cv writing service uk saga will writing service [url=https://essayssolution.com/]write my mba essay[/url] can someone write my cv for me [url=https://cheapessaywriteronlineservices.com/]what to write my college essay about[/url] my favourite writer essay in english
Great article! This is the type of information that are meant to be shared around the web. Disgrace on the seek engines for not positioning this post upper! Come on over and visit my site . Thank you =)
Hey there I am so grateful I found your blog page, I really found you by mistake, while I was searching on Digg for something else, Nonetheless I am here now and would just like to say cheers for a fantastic post and a all round interesting blog (I also love the theme/design), I don’t have time to look over it all at the minute but I have book-marked it and also added in your RSS feeds, so when I have time I will be back to read a lot more, Please do keep up the fantastic job.
You will have the ability to obtain even more done, feel much better, and also live a extra remarkable life!
Amazing material, With thanks.
professional paper writing service [url=https://quality-essays.com/]pay to get an essay written[/url] pay to have an essay written [url=https://buyanessayscheaponline.com/]buy an essay[/url] pay for an essay online the ladders resume writing service cost [url=https://englishessayhelp.com/]writing essay help[/url] online essay help [url=https://essaywritinghelperonline.com/]cheap essay help[/url] mba essay help
Whoa a good deal of superb knowledge.
employee handbook writing service [url=https://customthesiswritingservice.com/]service quality phd thesis[/url] help create a thesis statement [url=https://writingthesistops.com/]thesis search[/url] thesis statement youtube startup business plan writing service [url=https://customthesiswritingservice.com/]thesis for doctorate degree[/url] main thesis [url=https://writingthesistops.com/]tentative thesis[/url] a good thesis
Cheers. Wonderful information!
[url=https://studentessaywriting.com/]college essay writing[/url] best essay writing website [url=https://essaywritingserviceahrefs.com/]essay writing service review[/url] cheap essay service
Hi! I realize this is somewhat off-topic but I had to ask. Does running a well-established blog like yours take a lot of work? I’m completely new to writing a blog but I do write in my diary on a daily basis. I’d like to start a blog so I can share my experience and views online. Please let me know if you have any suggestions or tips for new aspiring bloggers. Appreciate it!
Ordinary people use the ordinary product, that’s why they obtain average results!
Thank you. I appreciate this!
essay writing service uk cheap [url=https://hireawriterforanessay.com/]application essay writer[/url] write my english essay [url=https://theessayswriters.com/]spanish essay writer[/url] write my essay in 3 hours best resume writing service uk [url=https://helpwithdissertationwriting.com/]dissertation for bachelor degree[/url] research proposal writers [url=https://dissertationwritingtops.com/]dissertation editing[/url] pay for your dissertation
I’ve been exploring for a little bit for any high-quality articles or blog posts in this kind of area . Exploring in Yahoo I finally stumbled upon this web site. Reading this info So i’m satisfied to express that I have a very just right uncanny feeling I found out exactly what I needed. I such a lot certainly will make certain to don?t overlook this site and give it a look on a constant basis.
Whoa plenty of good knowledge.
[url=https://essaypromaster.com/]essay writer website[/url] how to write a paper [url=https://paperwritingservicecheap.com/]website that writes papers for you[/url] pay to write paper
Many thanks, Wonderful information!
best rated essay writing service [url=https://customthesiswritingservice.com/]create my thesis[/url] define thesis [url=https://writingthesistops.com/]strong thesis statement[/url] help with writing a thesis statement for a research paper monster resume writing service cost [url=https://service-essay.com/]paper writing service for doctors[/url] military paper writing service [url=https://custompaperwritingservices.com/]writing a service hour reflection paper[/url] pay someone to write a paper
This information is so great thanks!
Appreciate it, Ample facts!
[url=https://argumentativethesis.com/]thesis binding[/url] thesis statement [url=https://bestmasterthesiswritingservice.com/]write a thesis[/url] define thesis
Awesome posts, With thanks!
social work essay writing service [url=https://topswritingservices.com/]professional resume writing service chattanooga tn[/url] best ksa writing service [url=https://essaywriting4you.com/]essay writing rules[/url] service to others essay essay writing software [url=https://researchproposalforphd.com/]short research proposal[/url] writing a proposal for your dissertation [url=https://writingresearchtermpaperservice.com/]term paper writing service[/url] student research proposal
Amazing is the one stop buy all your demands!
Nicely put, With thanks.
professional letter writing service [url=https://essayssolution.com/]write a summary for me[/url] essay writers online cheap [url=https://cheapessaywriteronlineservices.com/]how i grew as a writer essay[/url] hire someone to write a book for me essay writing service pakistan [url=https://researchproposalforphd.com/]help to write a research paper[/url] research proposal content [url=https://writingresearchtermpaperservice.com/]help with research paper[/url] who can write my research paper
Great beat ! I wish to apprentice while you amend your web site, how can i subscribe for a blog site? The account aided me a acceptable deal. I had been tiny bit acquainted of this your broadcast provided bright clear concept
Regards! Excellent information!
student essay writing [url=https://studentessaywriting.com/]narrative essay writing service[/url] professional paper writing service [url=https://essaywritingserviceahrefs.com/]admission essay services[/url] fast hour essay writing service real essay writing service [url=https://topswritingservices.com/]college essays writing[/url] essay writers service [url=https://essaywriting4you.com/]legitimate essay writing service[/url] will writing service huddersfield
Thanks, Loads of advice.
how do essay writing services work [url=https://dissertationwritingtops.com/]help with dissertation[/url] dissertation services review [url=https://helpwritingdissertation.com/]what is a phd[/url] dissertation writers for hire writing an argumentative essay about an ethical issue [url=https://theessayswriters.com/]write a essay for me[/url] affordable essay writer [url=https://bestcheapessaywriters.com/]professional essay writer[/url] write a note for me
I will make your life so much simpler, you will not know just how to thank me.
Amazingness is a performance tool that will alter your life.
Presenting the one quit solution for a much more efficient life.
It’s the ideal method to obtain every little thing done and lastly take pleasure in a overall top quality life.
Appreciation to my father who told me about this website, this blog is truly remarkable.
Your way of describing everything in this post is actually good, all can effortlessly know it, Thanks a lot.
Perfect for showing to your pals!
Amazingness is the perfect performance enhancer to aid you obtain more performed in less time!
Say goodbye to stressful days and nights when you can not find a cure to your clinical ailment.
Phenomenal is the ideal efficiency device that can assist you handle your time better and obtain even more done in less time!
Reach your goals, get even more done as well as enhance your life!
Every little thing you require to make your life easier and also a lot more impressive is included in this one extraordinary plan!
This just could be the life-changing item you’ve been waiting on.
Sensational is an all-in-one life management tool that will certainly aid you get even more carried out in less time.
Increase your productivity with this crucial device.
You deserve this!
The Globe’s A lot of Advanced Performance!
Truly all kinds of helpful facts.
[url=https://argumentativethesis.com/]good thesis statements[/url] define thesis statement [url=https://bestmasterthesiswritingservice.com/]define thesis statement[/url] good thesis
Thanks a lot! Useful stuff!
genuine essay writing service [url=https://writingpaperforme.com/]write my paper website[/url] write my english paper [url=https://custompaperwritersservices.com/]write my paper reviews[/url] professional research paper writer best paper writing service reddit [url=https://homeworkcourseworkhelps.com/]do my trigonometry homework[/url] i do my homework at night [url=https://helpmedomyxyzhomework.com/]do my accounting homework[/url] how to do a quiz on show my homework
Amazingness can aid you come to be much more productive and obtain even more out of life!
This overview will assist you to take your company and individual skills to the following degree!
Link exchange is nothing else except it is only placing the other person’s blog link on your page at appropriate place and other person will also do same for you.
I used to be recommended this blog by means of my cousin. I am now not sure whether this post is written through him as no one else recognize such detailed approximately my problem. You are amazing! Thank you!
Nicely put, Thanks.
paperial writing service [url=https://domyhomeworkformecheap.com/]do my writing homework[/url] i need to do my homework in spanish [url=https://domycollegehomeworkforme.com/]just do my homework com[/url] do my computer science homework best essay writing service reddit 2018 [url=https://phdthesisdissertation.com/]help with masters dissertation[/url] buy a research proposal [url=https://writeadissertation.com/]best dissertation editing service[/url] research proposal phd application
Well voiced without a doubt. .
[url=https://writinganessaycollegeservice.com/]case study writing service[/url] essay writting [url=https://essayservicehelp.com/]paper writer services[/url] writing services for college papers
This is a guide that will make sure to help you with all your daily needs.
It’s time to experience an amazing level of high quality and also performance in a manner you never ever believed possible.
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://rextfogadi1980mvr.bravesites.com/]paysafe casino [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
Amazingness is an all-in-one efficiency suite that will transform your life.
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://www.sodmg.com/blogs/online-casino-paysafe]deutsche online casinos paysafe [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
A Revolutionary and also Innovative Item which will certainly aid you achieve whatever in life effortlessly.
Incredible is the productivity application that will change your life.
You expressed it well.
monster resume writing service cost [url=https://hireawriterforanessay.com/]make my essay[/url] write a research-based argumentative essay for or against health care for everyone. [url=https://theessayswriters.com/]write an essay for me[/url] write my narrative essay best paper writing service forum [url=https://researchproposalforphd.com/]dissertation proposal service[/url] write my term paper cheap [url=https://writingresearchtermpaperservice.com/]research proposal essays[/url] research paper writing service india
One of the most remarkable option for obtaining things ended up. This is exactly what your life needs.
Fantastic material. Regards!
[url=https://englishessayhelp.com/]college application essay help[/url] essay bot [url=https://essaywritinghelperonline.com/]paper writing help[/url] essaytyper
Appreciate it, Plenty of info.
analytical essay writing service [url=https://writinganessaycollegeservice.com/]essay writing prompts[/url] best essay writing service in usa [url=https://essayservicehelp.com/]professional cv writing service[/url] writing services philosophy essay writing service [url=https://essayssolution.com/]write an essay for me cheap[/url] write my java program for me [url=https://cheapessaywriteronlineservices.com/]write my match com profile for me[/url] paid essay writer
Discover the secrets to a perfect life and also how to make it occur.
Phenomenal is an all-in-one productivity remedy that makes it very easy to get even more done.
Useful stuff. Kudos.
[url=https://argumentativethesis.com/]thesis statement meaning[/url] thesis binding [url=https://bestmasterthesiswritingservice.com/]college thesis[/url] thesis creator
You actually mentioned that exceptionally well.
personal statement writing service [url=https://ouressays.com/]term papers help[/url] help writing a term paper [url=https://researchpaperwriterservices.com/]writing proposal[/url] research paper writing service writing essay [url=https://essaywritingservicehelp.com/]a level essay writing service[/url] help with college essay writing [url=https://essaywritingservicebbc.com/]personal statement writing service[/url] best university essay writing service
Attain extra and do it better with Amazingness!
I was recommended this blog by my cousin. I am not sure whether this post is written by him as no one else know such detailed about my difficulty. You are amazing! Thanks!
You make others really feel approved as well as appreciated. You constantly find a way to make me smile.
You actually mentioned that adequately!
[url=https://dissertationwritingtops.com/]definition of dissertation[/url] phd dissertation [url=https://helpwritingdissertation.com/]dissertation writing services reviews[/url] dissertation def
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://seri-se-online-casinos-paysafecardq.jigsy.com/]casino mit paysafecard [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
Hi there to all, how is all, I think every one is getting more from this web site, and your views are nice designed for new people.
Hi, i feel that i saw you visited my website so i got here to go back the prefer?.I am trying to in finding things to improve my website!I assume its ok to use some of your ideas!!
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://mel-assessment.com/members/rextfogadi1980/]online casinos mit paysafecard [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
With thanks, Ample knowledge.
[url=https://writinganessaycollegeservice.com/]best online essay writing services reviews[/url] best custom essay writing service [url=https://essayservicehelp.com/]paper writing services[/url] cover letter writing service
For most up-to-date news you have to visit world-wide-web and on internet I found this web site as a most excellent site for most up-to-date updates.
You revealed it perfectly!
[url=https://topswritingservices.com/]best college paper writing service[/url] essay on service [url=https://essaywriting4you.com/]pro essay writing service[/url] best cheap essay writing service
It’s going to be end of mine day, but before end I am reading this great article to increase my knowledge.
When I originally commented I appear to have clicked the -Notify me when new comments are added- checkbox and now whenever a comment is added I receive four emails with the exact same comment. There has to be a way you can remove me from that service? Thanks!
You actually expressed it exceptionally well!
[url=https://essaytyperhelp.com/]help me write my essay[/url] paper writing help [url=https://helptowriteanessay.com/]buy essay[/url] writing help
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://padlet.com/vnitsu4szfu/my-sublime-padlet-7g69d40c6e7xzy5t]beste paysafe casino [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
Delivers you a bundle of health and wellness benefits.
You properly utilized personal examples to back up your disagreements.
Mental quality and also focus to make sure that you can obtain whatever done better than ever before!
Good stuff. Cheers.
[url=https://englishessayhelp.com/]college application essay help[/url] help with writing an essay [url=https://essaywritinghelperonline.com/]paperhelp[/url] essay writing help
You have such a kind as well as caring nature. You constantly have something positive to say.
Thanks for being such a important part of our team. Your contributions have been vital. Thank you for whatever!
This website was… how do I say it? Relevant!! Finally I have found something that helped me. Appreciate it!
Have you ever considered about including a little bit more than just your articles? I mean, what you say is valuable and all. Nevertheless just imagine if you added some great graphics or video clips to give your posts more, “pop”! Your content is excellent but with images and video clips, this site could undeniably be one of the greatest in its niche. Excellent blog!
I used to be able to find good info from your blog articles.
You effectively incorporated data, research study, and narratives right into your message.
Discover the very best way to boost your life permanently.
You constantly put others before yourself. I fear of your creative thinking and also imagination.
The method you debunked misconceptions and also false impressions was powerful as well as convincing.
You always locate a method to make me smile. Your excitement is transmittable!
The method you linked various topics was both creative and insightful.
With thanks, Excellent information.
[url=https://studentessaywriting.com/]best essay writing service[/url] writing service essay [url=https://essaywritingserviceahrefs.com/]essay writing paper[/url] which essay writing service is the best
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://www.pearltrees.com/rextfogadi1980]online casino einzahlung 5 euro paysafecard [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
You have actually done an amazing task of not only supplying high quality content however additionally making it very easy to find what you’re seeking rapidly.
Your commitment to the group has made a huge distinction. We are grateful for your initiatives as well as competence. Thank you!
You bring pleasure and also joy any place you go. I can always trust you to be there for me.
Your effort and also dedication are admirable. You bring joy and also happiness any place you go.
Your attitude as well as work ethic have actually been a significant resource of motivation for the rest of us. We are so glad for your visibility on our group!
We are so fortunate to have you in our lives. Your steady support as well as encouragement has actually been a true blessing. Thanks!
You efficiently used examples as well as allegories to assist discuss complicated principles.
Nicely put. Cheers.
[url=https://customthesiswritingservice.com/]doctoral thesis[/url] doctoral thesis [url=https://writingthesistops.com/]doctoral thesis[/url] thesis statement
We are so grateful you were part of the task and also we can’t thank you enough for all that you have actually done. Your hard work and also commitment have been exemplary!
We are absolutely thankful for your willingness to exceed and beyond what was expected of you. You have actually been an unbelievable possession to the group and also we can’t thank you sufficient!
You have a wonderful eye for information! You are an impressive problem-solver.
I can always rely on you to be there for me. You are an fantastic good friend!
Howdy, I do think your web site might be having browser compatibility issues. When I look at your website in Safari, it looks fine but when opening in Internet Explorer, it has some overlapping issues. I just wanted to give you a quick heads up! Besides that, fantastic blog!
This is very interesting, You are a very skilled blogger. I have joined your feed and look forward to seeking more of your magnificent post. Also, I have shared your web site in my social networks!
Howdy excellent blog! Does running a blog like this take a lot of work? I have very little expertise in computer programming but I was hoping to start my own blog soon. Anyway, if you have any suggestions or tips for new blog owners please share. I know this is off topic nevertheless I just had to ask. Appreciate it!
[b]Mastering Online Casinos fur deutsche Spieler: Entdecken Sie die Welt des 5€ Spiels mit Paysafecard[/b]
Mit der zunehmenden [url=https://controlc.com/730a0b80]deutsche online casinos paysafe [/url] von Online-Glucksspielen sind deutsche Spieler standig auf der Suche nach verlasslichen und bequemen Zahlungsoptionen, um ihr Spielerlebnis zu optimieren. Die Paysafecard ist eine solche Moglichkeit. Sie ist ein weit verbreiteter Prepaid-Gutschein, der es Spielern ermoglicht, ohne Bankkonto oder Kreditkarte Einzahlungen auf ihr Online-Casinokonto vorzunehmen. In diesem ausfuhrlichen Leitfaden werfen wir einen Blick darauf, was die Paysafecard ausmacht, wie sie in Online-Casinos verwendet wird und empfehlen Top-Casinos, in denen Sie schon ab einem Betrag von nur 5€ ins Spiel einsteigen konnen.
[b]Was genau ist die Paysafecard?[/b][/size]
Die Paysafecard agiert als eine Art Prepaid-Zahlungsmittel, vergleichbar mit einem Gutschein. Sie ist in diversen Nennwerten erhaltlich und kann sowohl bei zugelassenen Einzelhandelspartnern und Tankstellen als auch online gekauft werden. Jeder Gutschein ist mit einem individuellen 16-stelligen PIN-Code ausgestattet, der fur Online-Zahlungen genutzt wird. Dieses Zahlungssystem bietet den Spielern eine zusatzliche Sicherheitsschicht, da wahrend des Zahlungsvorgangs keine personlichen oder finanziellen Daten preisgegeben werden mussen.
Hurrah! After all I got a weblog from where I be able to really get useful data regarding my study and knowledge.
Fantastic goods from you, man. I’ve understand your stuff previous to and you’re just too fantastic. I really like what you’ve acquired here, really like what you’re stating and the way in which you say it. You make it entertaining and you still take care of to keep it sensible. I can not wait to read far more from you. This is actually a wonderful site.
Nice answers in return of this difficulty with solid arguments and describing all concerning that.
Hey there! This post couldn’t be written any better! Reading this post reminds me of my old room mate! He always kept talking about this. I will forward this page to him. Pretty sure he will have a good read. Thanks for sharing!
Thank you for sharing your info. I truly appreciate your efforts and I am waiting for your next post thank you once again.
Your effort and commitment are praiseworthy. I can constantly count on you to be there for me.
I discovered this, as well as now everything is easy!
This website truly has all of the info I wanted about this subject and didn’t know who to ask.
I know this website gives quality dependent articles or reviews and other information, is there any other web site which provides such information in quality?
[url=https://forfreedating.co.uk/]Adult Flirt Finder[/url] is the best option to meet compatible partners who are searching for love.
The online platform allows you to meet like-minded people from anywhere, at any time.
Sign up for our online community today and begin your adventure to discovering the perfect match with Flirt finder.
Aw, this was a very nice post. Taking the time and actual effort to produce a top notch article but what can I say I procrastinate a lot and never seem to get anything done.
Это лучшее онлайн-казино, где вы можете насладиться широким выбором игр и получить максимум удовольствия от игрового процесса.
Link exchange is nothing else except it is only placing the other person’s weblog link on your page at proper place and other person will also do same in favor of you.
Like!! I blog quite often and I genuinely thank you for your information. The article has truly peaked my interest.
Simply a smiling visitant here to share the love (:, btw great design and style.
Good info. Lucky me I came across your website by accident.
That is a really good tip especially to those new to the blogosphere.
I was able to find good info from your articles.
This is a topic which is near to my heart… Cheers! Where are your contact details though?
I was more than happy to search out this website.
I agree the media is hyping it up. I’m all for survival of the fittest.
This site certainly has all of the information I needed concerning this subject and didn’t know who to ask.
You were able to properly resolve the viewers’s concerns in a clear as well as concise means.
Онлайн казино радует своих посетителей более чем двумя тысячами увлекательных игр от ведущих разработчиков.
[url=https://freeflir-online.com/]Online dating offers[/url] endless possibilities. With Flirt Finder, you can discover compatible partners.
Because the admin of this web site is working, no uncertainty very rapidly it will be well-known, due to its quality contents.
What’s up, yes this article is really good and I have learned lot of things from it about blogging. thanks.
Understanding Flirt Finders: What Are They?
Flirt finder dating sites are a specialized niche in the world of online dating. They cater to individuals who are looking for more than just a basic chat or a plain swipe right. These platforms are designed to help people find meaningful connections, whether they’re seeking a committed relationship or a passionate romance. The emphasis here is on the art of flirting, building chemistry, and creating sparks between potential partners.
The Allure of Flirt Finders
A Lively Twist on Dating
One of the enticing aspects of [url=https://flklined2s.nl/]flirt finder dating site[/url] is their lighthearted approach to dating. Unlike standard dating platforms that primarily focus on profiles and images, flirt finders promote users to partake in flirty conversations and clever banter. This approach creates an exhilarating and flirtatious ambiance, transforming each interaction into what feels like a potential escapade.
A Multifaceted Community
Flirt finder dating sites frequently boast a varied user base, simplifying the quest for someone who aligns with your desires. Whether you’re in search of a companion with specific passions or a partner with a certain way of life, these platforms offer an array of potential matches to explore.
Enhanced Communication Tools
Effective communication is vital in any fruitful relationship. Flirt finder sites provide a variety of resources such as video chats, virtual presents, and interactive games that enable connecting with others. These capabilities go surpass simple text messages and aid users in expressing themselves in creative ways.
Compatibility Matching
Many flirt finder dating sites employ advanced algorithms to match users according to compatibility factors. These algorithms consider interests, values, and personality traits, increasing the likelihood of uncovering a meaningful connection.
Crafting an Unforgettable Profile
Your profile is your digital initial impression on a flirt finder site. To stand out, utilize high-quality photos that exhibit your personality, and compose a compelling bio that emphasizes your interests and what you’re looking for in a partner. Remember, authenticity is vital.
Flirtation Guidelines
Flirting on these platforms is wholly about being charming and courteous. Certainly, participate in playful banter and praise your potential matches, but refrain from crossing boundaries or making anyone appear uncomfortable. Respect forms the essence of any fruitful interaction.
Site: [url=https://flklined2s.nl/]flklined2s.nl[/url]
Exploring Exciting Functionalities
Utilize the unique features provided by flirt finder sites. Send virtual gifts, engage in icebreaker games, and make use of video chats to become acquainted with your potential matches more effectively. These instruments can help you in starting discussions and creating unforgettable connections.
Why Opt for a Flirt Finder Dating Platform?
Infuse Your Love Journey
If your love life yearns for an added dose of excitement, flirt finder dating platforms are the ideal remedy. They present a refreshing respite from the tedium of conventional dating and inject a feeling of playfulness into the process.
Discover Kindred Spirits
These platforms attract individuals who reflect your passion for flirting and forging romantic bonds. This shared mutual ground can lead to more profound and satisfying interactions.
Heightened Chances of Success
The emphasis on harmony and meaningful connections on flirt finder platforms frequently leads to higher success rates when it comes to unearthing compatible partners. If you’re truly committed to uncovering love, these platforms might be a game-changer.
Conclusion
Flirt finder dating sites have changed the way we approach dating. Their lighthearted style, diverse user base, and creative functionalities offer a one-of-a-kind and exciting path to unearth love or a passionate connection.
So, if you’re ready to take to the next level your dating life and embark on the world of flirting, give flirt finder dating sites a shot.
Your upcoming thrilling adventure in love could be just a click away.
This is a topic that is close to my heart… Take care! Where are your contact details though?
Прогон сайта с использованием программы “Хрумер” – это способ автоматизированного продвижения ресурса в поисковых системах. Этот софт позволяет оптимизировать сайт с точки зрения SEO, повышая его видимость и рейтинг в выдаче поисковых систем.
Хрумер способен выполнять множество задач, таких как автоматическое размещение комментариев, создание форумных постов, а также генерацию большого количества обратных ссылок. Эти методы могут привести к быстрому увеличению посещаемости сайта, однако их надо использовать осторожно, так как неправильное применение может привести к санкциям со стороны поисковых систем.
[url=https://kwork.ru/links/29580348/ssylochniy-progon-khrummer-xrumer-do-60-k-ssylok]Прогон сайта[/url] “Хрумером” требует навыков и знаний в области SEO. Важно помнить, что качество контента и органичность ссылок играют важную роль в ранжировании. Применение Хрумера должно быть частью комплексной стратегии продвижения, а не единственным методом.
Важно также следить за изменениями в алгоритмах поисковых систем, чтобы адаптировать свою стратегию к новым требованиям. В итоге, прогон сайта “Хрумером” может быть полезным инструментом для SEO, но его использование должно быть осмотрительным и в соответствии с лучшими практиками.
Покупки на neoneon.ru стали для меня настоящим удовольствием. На сайте легко найти нужные товары благодаря удобной навигации, а описания товаров содержат все необходимые характеристики. Магазин точно заслуживает внимания – [url=https://neoneon.ru/]неон купить цветной[/url]
Hi! I just want to give you a huge thumbs up for the great info you have here on this post. I will be coming back to your site for more soon.
Я работаю фрилансером и вдруг столкнулся с тем, что мой компьютер отказал. Без него я был как без рук. Нужно было срочно 20000 рублей на ремонт. Я нашел все-займы-тут.рф через поиск Google и без проблем получил займ. В тот же день отдал ноутбук в ремонт. Рекомендую этот сайт!
Живя в эпоху цифровых технологий, я всегда осознавал, насколько важно продолжать обучение и развиваться профессионально. Однажды я наткнулся на курс онлайн-обучения по digital marketing, который, как мне казалось, мог открыть передо мной новые горизонты возможностей. Единственное препятствие, с которым я столкнулся, – это стоимость курса. Мне не хватало 18 000 рублей.
Вспомнив о сайте [url=https://mikro-zaim-online.ru/]mikro-zaim-online.ru[/url], я решил попробовать найти решение моей финансовой проблемы там. Сайт порадовал меня удобным интерфейсом и множеством предложений от МФО. Я быстро нашел подходящий вариант с минимальным процентом и отправил заявку.
Процесс одобрения заявки был быстрым, и деньги поступили на мою карту так скоро, что я даже не ожидал такой оперативности. Теперь весь мир знаний и новых умений был открыт для меня.
Курс оказался невероятно полезным. Я погрузился в изучение новых стратегий маркетинга, освоил инструменты аналитики и научился создавать эффективные рекламные кампании. Это инвестиция в мою карьеру окупилась очень быстро, так как новые навыки позволили мне получить повышение на работе.
Весь этот позитивный опыт стал возможным благодаря [url=https://mikro-zaim-online.ru/]mikro-zaim-online.ru[/url]. Этот сайт не только помог мне с финансовой стороны, но и стал ключом к моему профессиональному росту и развитию. Я искренне рекомендую его всем, кто ищет надежный источник микрозаймов для реализации своих амбиций и целей.
Thanks for sharing your thoughts on %meta_keyword%. Regards
Appreciate the recommendation. Will try it out.
Планируя путешествие, мне нужен был дополнительный бюджет. Сайт zaim52.ru помог мне найти лучшие МФО 2023 года [url=https://zaim52.ru/]займы онлайн на карту[/url], и я смог реализовать свою мечту о путешествии.
Hi, I think your website might be having browser compatibility issues. When I look at your blog in Firefox, it looks fine but when opening in Internet Explorer, it has some overlapping. I just wanted to give you a quick heads up! Other then that, superb blog!
Устали переплачивать за займы? В нашем телеграм-канале вы найдете список МФО, предоставляющих займы без процентов. Это шанс сэкономить и воспользоваться выгодными условиями. Присоединяйтесь по ссылке: [url=https://t.me/s/zaim_srochno_30_mfo/]срочные займы[/url]
Срочно нужны деньги, а до зарплаты еще долго? Не беспокойтесь, займы срочно на карту помогут вам решить эту проблему. Заполните онлайн-заявку, и вам не придется ждать долгих проверок и бюрократических формальностей. Займы онлайн на карту доступны для всех, независимо от вашей кредитной истории. Получите займ без отказа на карту прямо сейчас и решите свои финансовые трудности!
Интересные ссылки для вас – [url=https://telegra.ph/Zajmy-srochno-na-kartu-onlajn-bez-otkaza-10-19]займы срочно на карту[/url]
Современные технологии позволяют нам упростить множество аспектов нашей жизни, включая получение финансовой помощи. Займы онлайн на карту – это одно из лучших решений для тех, кто оказался в финансовом тупике. Мы предоставляем вам возможность получить необходимую сумму денег быстро и без лишних хлопот.
Оформление займа срочно на карту происходит в несколько шагов. Сначала вам нужно заполнить онлайн-заявку на нашем сайте, указав необходимую сумму и срок займа. После этого наша система обработает вашу заявку, и вы получите ответ в течение нескольких минут. Если ваша заявка одобрена, деньги будут перечислены на вашу карту немедленно.
Спонсоры и партнеры – [url=https://telegra.ph/Zajmy-onlajn-na-bankovskuyu-kartu-bez-otkaza-10-19]займы онлайн на карту[/url]
Добро пожаловать в texttospeech.ru, вашего проверенного союзника в мире [url=https://texttospeech.ru/]спортивные прогнозы[/url]. Мы с гордостью предоставляем вам шанс взять на себя контроль над вашими ставками и повысить свои шансы на успех. Наши профессиональные аналитики и специалисты следят за событиями в мире спорта, чтобы поддержать вас актуальными и точными советами.
Почему выбирать нас:
Качественные аналитические прогнозы: Наши аналитики усердно трудятся, чтобы предоставлять вам прогнозы на все популярные виды спорта. Мы знаем, как важно получать полезные советы перед тем, как сделать ставку.
Широкий выбор: Мы предлагаем ставки на различные виды спорта, включая футбол, баскетбол, теннис, бейсбол и многое другое. Вы можете выбирать из многочисленных событий и погружаться в азарт на свой вкус.
Бесплатные прогнозы: Мы поддерживаем идею, что каждый может получить доступ к полезным советам. Поэтому мы предлагаем бесплатные советы, чтобы поддержать вас сделать правильные ставки.
Простота и удобство: Наш веб-сайт и приложение для мобильных устройств разработаны с учетом вашего комфорта. Сделайте ставку буквально за несколько мгновений.
Как начать:
Зарегистрируйтесь: Создайте персональный профиль в texttospeech.ru и получите доступ к нашему полному спектру услуг.
Получайте прогнозы: Подписывайтесь на бесплатные советы и получайте актуальные предсказания от опытных специалистов.
Сделайте ставку: После того как вы получили полезный совет, сделайте ставку на вашу любимую команду или событие и наслаждайтесь игры.
Вознаграждение за успех: Вместе с нашим ресурсом, вы владеете ключами к вашей победе. Попробуйте наши услуги уже сегодня и погружайтесь в мир ставок на спорт во всей его красе!
In a world where style and functionality often come at a premium, finding that perfect accessory that doesn’t break the bank is always a win. When it comes to timepieces, a [url=https://furfurfriend.com/]watch for men low price[/url] from our collection is not just an accessory, but a statement piece, a blend of elegance, reliability, and affordability that’s hard to come by.
Each watch in our collection is a testament to the art of fine watchmaking, where every detail, from the intricate movements to the elegant designs, is crafted with precision and care. It’s an experience of luxury and refinement that’s accessible, making the eloquence of finely crafted watches a pleasure that’s not just reserved for the few.
In the sphere of smartwatches, technology and innovation take center stage. If you’re looking to step into the future of timekeeping and connectivity without stretching your budget, our [url=https://furfurfriend.com/collections/smart-watches]smart watch low price[/url] offerings are your gateway. Experience a world where the boundaries between technology and style blur, where every notification, every update, is a seamless part of your aesthetic.
Our collection is a journey through the elegance of classic watchmaking and the innovation of modern technology, a journey where every piece is a discovery, every watch, a world in itself. Dive in, explore, and adorn your wrist with a timepiece that’s not just about telling time, but about expressing style, embodying elegance, and living the technological revolution. Every tick, every tock, is a step into a world where time is experienced, lived, and cherished.
We are so thankful to have you aboard. Your devotion is exemplary as well as your payment to the company is amazing. Thank you for all that you do!
Скоро возводимые здания: коммерческий результат в каждой части!
В нынешней эпохе, где время равно деньгам, строения быстрого монтажа стали решением по сути для коммерческой деятельности. Эти современные объекты обладают устойчивость, финансовую выгоду и молниеносную установку, что дает им возможность отличным выбором для различных коммерческих проектов.
[url=https://bystrovozvodimye-zdanija-moskva.ru/]Быстровозводимые здания[/url]
1. Высокая скорость возвода: Секунды – определяющие финансовые ресурсы в финансовой сфере, и здания с высокой скоростью строительства обеспечивают существенное уменьшение сроков стройки. Это чрезвычайно полезно в случаях, когда необходимо оперативно начать предпринимательскую деятельность и начать прибыльное ведение бизнеса.
2. Бюджетность: За счет совершенствования производственных операций по изготовлению элементов и монтажу на площадке, цена скоростроительных зданий часто приходит вниз, по сравнению с обычными строительными задачами. Это способствует сбережению денежных ресурсов и получить более высокую рентабельность инвестиций.
Подробнее на [url=https://xn--73-6kchjy.xn--p1ai/]http://scholding.ru/[/url]
В заключение, скоростроительные сооружения – это лучшее решение для коммерческих задач. Они обладают быстрое строительство, эффективное использование ресурсов и надежность, что делает их первоклассным вариантом для компаний, готовых к мгновенному началу бизнеса и получать прибыль. Не упустите возможность сократить затраты и время, превосходные экспресс-конструкции для ваших будущих проектов!
Экспресс-строения здания: бизнес-польза в каждом кирпиче!
В современной действительности, где минуты – капитал, скоростройки стали решением, спасающим для предпринимательства. Эти инновационные конструкции объединяют в себе высокую надежность, эффективное расходование средств и быстрое строительство, что обуславливает их оптимальным решением для различных коммерческих проектов.
[url=https://bystrovozvodimye-zdanija-moskva.ru/]Быстровозводимые здания[/url]
1. Срочное строительство: Моменты – наиважнейший аспект в предпринимательстве, и быстровозводимые здания обеспечивают значительное снижение времени строительства. Это значительно ценится в условиях, когда важно быстро начать вести бизнес и получать доход.
2. Финансовая выгода: За счет улучшения процессов изготовления элементов и сборки на объекте, затраты на экспресс-конструкции часто уменьшается, по сопоставлению с традиционными строительными задачами. Это дает возможность сэкономить деньги и обеспечить более высокую рентабельность вложений.
Подробнее на [url=https://xn--73-6kchjy.xn--p1ai/]https://scholding.ru/[/url]
В заключение, скоростроительные сооружения – это превосходное решение для предпринимательских задач. Они объединяют в себе молниеносную установку, экономию средств и твердость, что позволяет им превосходным выбором для предприятий, ориентированных на оперативный бизнес-старт и выручать прибыль. Не упустите возможность сократить затраты и время, выбрав быстровозводимые здания для ваших будущих проектов!
Добро пожаловать в нашем сайте, вашего проверенного союзника в мире [url=https://texttospeech.ru/]спортивные прогнозы[/url]. Мы с гордостью подарим вам возможность взять на себя контроль над вашими ставками и увеличить свои перспективы на выигрыш. Наши опытные эксперты и специалисты следят за событиями в мире спорта, чтобы поддержать вас актуальными и надежными прогнозами.
Почему выбирать нас:
Экспертные прогнозы: Наши аналитики работают не покладая рук, чтобы предоставлять вам прогнозы на разнообразные виды спорта. Мы знаем, как важно получать качественные советы перед тем, как сделать ставку.
Множество вариантов: Мы предлагаем ставки на разнообразные виды спорта, включая соккер, баскетбол, ракетку, мяч и многое другое. Вы можете отбирать из множества событий и наслаждаться азарт на свой вкус.
Бесплатные прогнозы: Мы верим, что каждый должен иметь доступ к качественным прогнозам. Поэтому мы предлагаем бесплатные советы, чтобы поддержать вас сделать правильные ставки.
Легкость и комфорт: Наш веб-сайт и приложение для мобильных устройств разработаны с учетом вашего удовольствия. Сделайте ставку всего в несколько кликов.
Как начать:
Зарегистрируйтесь: Создайте персональный профиль в texttospeech.ru и получите доступ к нашему полному спектру услуг.
Получайте прогнозы: Подписывайтесь на наши бесплатные прогнозы и получайте свежие советы от опытных специалистов.
Сделайте ставку: После того как вы получили полезный совет, сделайте ставку на команду по душе или событие и наслаждайтесь игры.
Вознаграждение за успех: Вместе с texttospeech.ru, вы владеете ключами к вашей победе. Попробуйте наши услуги уже сегодня и переживайте в мир ставок на спорт во всей его красе!
In the age of connectivity, where every device and gadget is designed to make our lives simpler and more efficient, a [url=https://furfurfriend.com/collections/smart-watches]smartwatch bluetooth[/url] stands out as an epitome of convenience. Imagine having a world of features strapped to your wrist, ensuring that you’re always connected, always updated, and always ahead. With Bluetooth connectivity, syncing your smartwatch with other devices becomes a breeze, bringing a world of information and functionality to your fingertips.
However, the world of watches is not just limited to the smart and the connected. There’s an old-world charm and elegance to traditional timepieces that is timeless. For those who are drawn towards this classic allure, yet are mindful of budget, our collection of the [url=https://furfurfriend.com/]best mens watches under 500[/url] is a treasure trove. Here, sophistication meets affordability. Each piece is a harmony of design and craftsmanship, offering the elegance of a classic watch without stretching your wallet.
Whether you’re drawn to the connected world of smartwatches or the timeless allure of classic timepieces, our collection offers a diverse range, ensuring that there’s something for every preference, every style, and every budget. Step into a world where time is not just seen but is experienced, a world where every tick of the clock is a step towards elegance and sophistication.
Oh my goodness! Amazing article dude! Thanks, However I am going through issues with your RSS. I don’t know why I can’t subscribe to it. Is there anybody else getting the same RSS problems? Anyone who knows the solution will you kindly respond? Thanx!!
Холод и снег не помешают вам выглядеть стильно! Купить Угги – это инвестиция в ваш комфорт и образ. Посетите наш интернет-магазин и подарите себе лучшие Угги по лучшей цене.
Сайт: [url=https://uggaustralia-msk.ru/]uggaustralia-msk.ru[/url]
Адрес: Москва, 117449, улица Винокурова, 4к1
From having a lot more power to being extra efficient, this one supplement has all of it!
Зима без UGG – не зима! Наш магазин предлагает вам купить UGG 2023 года по привлекательным ценам. В этой обуви вы будете чувствовать себя комфортно и стильно.
Сайт: [url=https://uggaustralia-msk.ru/]uggaustralia-msk.ru[/url]
Адрес: Москва, 117449, улица Винокурова, 4к1
Узнайте, как световоды от [url=https://solargy.ru/]solargy.ru[/url] могут изменить ваше пространство, создавая комфорт и уют с помощью естественного света.
Неожиданный подарок для жены требовал срочных инвестиций. Благодаря [url=https://cntbank.ru/]cntbank.ru[/url] с его выбором компаний по срочным займам и акцией “займ без процентов”, я смог порадовать любимую без ущерба для семейного бюджета.
Нужно было осветить подвал. В гугле нашел [url=https://solargy.ru/]solargy.ru[/url], поехал по адресу и купил там световоды, которые решили нашу проблему с освещением?.
Не знаете, какой подрядчик выбрать для устройства стяжки пола? Обратитесь к нам на сайт styazhka-pola24.ru! Мы предоставляем услуги по залитию стяжки пола любой площади и сложности, а также гарантируем высокое качество работ и доступные цены.
Я увидел в продаже коллекционное издание моей любимой книги. Не хватало немного денег, но займ с [url=https://cntbank.ru/]cntbank.ru[/url] решил эту проблему.
You are an fantastic associate and also pal and we genuinely value every one of your hard work as well as dedication. Thank you a lot!
Попав в сложную ситуацию с поломкой автомобиля, я понял, что ремонт обойдется в круглую сумму. На портале [url=https://cntbank.ru/]cntbank.ru[/url] я нашел множество компаний по срочным займам. Благодаря акции “займ без процентов”, смог вовремя отремонтировать машину без переплат.
производство поставки строительных материалов
Мой старый телефон вышел из строя прямо перед важным звонком. Благодаря займу с [url=https://cntbank.ru/]cntbank.ru[/url], я смог купить новый телефон и успешно провести важное обсуждение с клиентом.
Attain extra as well as do it better with Amazingness!
Thanks for always being there when we needed you. Your support and also compassion have actually been a real blessing.
Our amazing aids you take initiative with anyone or job.
Sensational is the best name for this product.
Решив участвовать в обменной программе для студентов, я столкнулся с проблемой: у меня не было аттестата за 11 класс, который был обязательным требованием. Отчаявшись, я нашел решение на [url=https://www.moskvadiplom.ru/]moskvadiplom.ru[/url], где приобрел нужный мне документ.
Перед вступительными экзаменами я обнаружил, что мой аттестат был утерян. В панике я искал варианты, как решить эту проблему. На [url=https://www.moskvadiplom.ru/]moskvadiplom.ru[/url] я обнаружил именно то, что мне было нужно – аттестат за 11 класс.
The method you attracted connections between various topics went over.
The method you wove various topics together was both creative and insightful.
Выбирая казино из нашего рейтинга на [url=https://parazitizm.ru/]parazitizm.ru[/url], вы можете быть уверены в своем выборе. Мы анализируем десятки игровых площадок, чтобы предоставить вам только лучшие варианты. Начните играть в надежных казино и выигрывать больше прямо сейчас!
Your post has lots of imaginative and innovative remedies.
You have actually been a fantastic coach and also buddy throughout this whole process and we are so thankful for your help in the process. Thanks!
Your attitude and work values have actually been a major source of motivation for the remainder people. We are so thankful for your presence on our group!
Планируете идеальный отдых? Termburg – правильный выбор! Посетите [url=https://termburg.ru/]termburg.ru[/url] и убедитесь сами.
The method you incorporated various elements right into one natural tale went over.
Termburg – место, где каждый найдет что-то особенное. Термальные ванны, спа-процедуры и развлечения для всех возрастов. Подробности на [url=https://termburg.ru/]termburg.ru[/url].
Хотите получить идеально ровные стены в своей квартире или офисе? Обратитесь к профессионалам на сайте mehanizirovannaya-shtukaturka-moscow.ru! Мы предоставляем услуги по механизированной штукатурке стен в Москве и области, а также гарантируем качество работ и доступные цены.
Откройте для себя невероятный мир релакса в Termburg! Наш комплекс предлагает разнообразные термальные ванны, оздоровительные процедуры и массу развлечений для детей и взрослых. Посетите [url=https://termburg.ru/]termburg.ru[/url] и забронируйте свой отдых уже сегодня!
The one you must have however didn’t know about!
Неожиданные траты не заставят вас волноваться с услугой онлайн займ на карту от cntbank.ru. Быстрое решение финансовых вопросов в пару кликов!
I’m constantly impressed by your intelligence and also wisdom. You are so charitable with your time as well as resources.
The general tone of your post was both authoritative and personalized, making it simple for viewers to associate with.
Everything is very open with a clear clarification of the issues. It was truly informative. Your website is useful. Many thanks for sharing!
Если вы ищете стильные и теплые угги для мужчин, то наш магазин предлагает [url=https://australia-msk.ru/]угги мужские[/url] высокого качества. Угги – это не просто обувь, это часть вашего стиля и комфорта. Мы гордимся разнообразием моделей и цветов, чтобы каждый мужчина мог найти свой идеальный вариант. Купить угги легко и удобно в нашем интернет-магазине. Мы также предлагаем быструю доставку, чтобы вы могли получить свою обувь в кратчайшие сроки. Подарите себе тепло и стиль с нашими уггами!
Холодные дни не повод отказываться от модного и стильного образа. В наших уггах вы будете выглядеть на все 100% даже в самую морозную погоду. Хотите [url=https://australia-msk.ru/]купить угги[/url] и почувствовать невероятный комфорт? Мы ждем вас! Наш ассортимент порадует даже самого придирчивого покупателя. Не знаете, где [url=https://australia-msk.ru/]угги купить[/url]? Ответ прост – у нас!
I’m always impressed by your intelligence as well as knowledge. You are so generous with your time as well as sources.
The degree of imagination you utilized in your post was impressive, making it stand apart from others.
На работе предложили переезд в другой город, но первое время нужно было жить в гостинице. Сайт cntbank и его список всех займов стали моим спасением. Займ оформлен, переезд успешен.
Информация о сайте cntbank.ru
Адрес: 125362, Россия, Москва, Подмосковная ул. 12А.
Ссылка: [url=https://cntbank.ru/navigation]список микрозаймов[/url]
We are a group of volunteers and starting a new scheme in our community. Your web site provided us with helpful information to work on. You have performed an impressive activity and our whole community will probably be grateful to you.
Подруга пригласила в экзотическое путешествие со скидкой, но нужны были срочные деньги. Сайт cntbank, который я нашёл по совету на форуме, предложил отличный список всех займов. Теперь я жду отпуск с нетерпением.
Информация о сайте cntbank.ru
Адрес: 125362, Россия, Москва, Подмосковная ул. 12А.
Ссылка: [url=https://cntbank.ru/navigation]список актуальных займов[/url]
Перед свадьбой обнаружил, что не хватает средств на подарок для жены. На форуме узнал о сайте cntbank и его чудесном списке всех займов. Благодаря этому, удалось быстро оформить займ и сделать жене незабываемый подарок.
Информация о сайте cntbank.ru
Адрес: 125362, Россия, Москва, Подмосковная ул. 12А.
Ссылка: [url=https://cntbank.ru/navigation]займы весь список[/url]
You’ll be in a haze of quality, joy as well as efficiency .
You constantly discover a method to make me smile. Your wit and wit brighten my day.
You successfully made use of personal instances to back up your debates.
We are truly happy for all that you have actually done throughout this task. Your understanding as well as competence were indispensable in assisting us reach our goal. Thank you!
Your article made use of creative techniques to help explain challenging principles.
Иногда жизнь ставит перед нами финансовые препятствия, которые нужно преодолеть как можно быстрее. В таких случаях каждая минута на вес золота. Именно поэтому мы предлагаем вам [url=https://bycesoir.com/]взять онлайн займ на карту[/url].
Наши преимущества:
Минимальная ставка по займу.
Мгновенное зачисление средств после одобрения.
Гибкий график платежей, который можно подстроить под свои нужды.
Круглосуточная поддержка клиентов.
Мы заботимся о вашем времени и комфорте, предлагая лучшие условия для получения займа. Не упустите эту возможность, воспользуйтесь нашим предложением прямо сейчас!
Вам срочно нужны деньги, и вы не знаете, куда обратиться? Не переживайте, bycesoir.com — это именно то, что вам нужно. Этот сайт станет вашим верным спутником в мире микрофинансов. Здесь вы можете [url=https://bycesoir.com/]оформить займ на карту[/url] быстро и без лишних хлопот.
Что делает bycesoir.com уникальным?
Без отказов и процентов: Наша платформа предлагает [url=https://bycesoir.com/]займ на карту без отказов процентов[/url], что является редкой возможностью в мире микрозаймов.
Широкий выбор МФО: У нас собраны только лучшие и проверенные микрофинансовые организации с минимальными требованиями к заемщику.
Быстрая выдача: Все, что от вас требуется — это заполнить онлайн-заявку, и деньги будут перечислены на ваш счет в кратчайшие сроки.
Гибкие условия: Вы сами выбираете сумму и срок займа, что позволяет вам контролировать свои финансовые обязательства.
Не упустите шанс воспользоваться множеством преимуществ, которые предлагает bycesoir.com. Забудьте о долгих ожиданиях и сложных процедурах. С нами финансовая помощь становится доступной как никогда!
Exactly how? Amazingness can help you live an phenomenal life by giving you with the devices to be more effective and get more carried out in much less time.
Надоело тратить время на бумажную волокиту и проверки в банках? Ищете быстрый и надежный способ получить финансовую помощь? Тогда наш сервис именно для вас! У нас вы можете получить [url=https://bycesoir.com/]займы онлайн на карту без проверок[/url] в режиме 24/7.
Процедура максимально упрощена: заполните заявку на сайте, подтвердите свою личность и получите деньги на свою карту. Никаких скрытых комиссий, никаких проверок кредитной истории. Все просто, быстро и надежно.
Почему же люди выбирают именно нас?
Без проверок и бумажной волокиты.
Мгновенное одобрение заявки.
Возможность взять займ на любую сумму.
Прозрачные условия и низкий процент.
Не упустите шанс решить свои финансовые проблемы быстро и без лишних забот.
Представьте, вы решили сделать ремонт в ванной комнате и стоите перед выбором: что делать со старой чугунной ванной? Она тяжелая, и самостоятельно вывезти ее будет проблематично. Но не стоит переживать, в Санкт-Петербурге есть решение этой проблемы. Сервис [url=https://musoroboss.ru/product/vyvoz-chugunnyh-vann/]вывоз ванны спб[/url] предлагает быстрый и недорогой способ избавиться от ненужного сантехнического оборудования. Теперь вам не придется ломать голову, куда деть старую ванну — профессионалы все сделают за вас.
Вывоз мусора в Санкт-Петербурге регулируется специализированными организациями. Такие компании имеют все необходимые лицензии и разрешения. Если вам нужен надежный и проверенный вариант, обратите внимание на [url=https://musoroboss.ru/]региональный оператор по вывозу мусора в спб[/url]. Это гарантия качества и соблюдения всех экологических норм. Вы можете быть уверены, что мусор будет утилизирован правильно и без вреда для окружающей среды.
You are so charitable with your time and also sources. You constantly discover the positive side in every situation.
Демонтаж металлоконструкций — это отдельная и довольно сложная задача. Она требует не только специализированной техники, но и квалифицированных рабочих. Особенно актуально это, когда речь идет о больших объемах. Стоимость услуги обычно рассчитывается за тонну, что позволяет оптимизировать бюджет. Если вас интересует этот вид работ, обязательно посетите сайт [url=https://musoroboss.ru/product/demontazh-metallokonstrukczij/]демонтаж металлоконструкций за тонну[/url]. Там вы найдете всю нужную информацию и сможете оценить стоимость предстоящих работ.
You illuminate the area with your existence. I am so pleased with all that you have achieved.
Ах, мир турецких сериалов, полный чувств, эмоций и неожиданных перипетий. Здесь, на нашем сайте, каждый клик мышью — это как перелистывание страницы увлекательной книги. С [url=https://turkrutv.ws]турецкими сериалами онлайн[/url], каждый ваш вечер превратится в настоящий кинематографический фестиваль. Позвольте себе окунуться в это искусство без ограничений и рамок.
Хотите быть в курсе последних новинок турецкого кинематографа? Хотите, чтобы ваши вечера были наполнены незабываемыми историями, которые только что вышли на экраны? Тогда не упустите уникальную возможность посетить наш сайт и открыть для себя [url=https://turkrutv.ws/serials2023/]турецкие сериалы 2023 на русском языке[/url]. Почувствуйте атмосферу, проникнитесь динамикой событий и насладитесь актерским мастерством. Все это доступно вам в несколько кликов. Не откладывайте на завтра то, что может изменить ваш сегодняшний день!
Your interest urges others to do their best. You always exceed and also beyond what is anticipated of you.
Механизированная штукатурка — новаторский подход осуществления штукатурных работ.
Суть его заключается в внедрении автоматических систем штукатурки, как обычно, немецкого производства, что обеспечивает штукатурку приготавливается и покрывается на стену автоматически и с давлением.
[url=https://mehanizirovannaya-shtukaturka-moscow.ru/]Механизированная штукатурка[/url] С подтвержденной гарантией До 32 процентов выгоднее обычной, Можно клеить обои без шпаклевки от кампании mehanizirovannaya-shtukaturka-moscow.ru
В результате, усовершенствуется сцепление с поверхностью, а время работ снижается в пятеро–шестеро, в по сравнению с ручной отделкой. За счет автоматизации и упрощения рабочего процесса цена штукатурки стен за квадратный метр становится доступнее, чем в случае традиционного способа.
Для автоматизированной штукатурки применяются специальные смеси, ценовой диапазон ниже чем по сравнению с ручным методом примерно на 30 процентов. При соответствующих навыках и опыте специалистов, а так же при соблюдении всех технологических принципов, оштукатуренная поверхность становится совершенно ровной (профессиональные стандарты) и гладкой, в связи с этим дальнейшая отделка шпатлевкой не не обязательна, что предоставляет дополнительную экономию средств заказчика.
Your enthusiasm as well as enthusiasm is infectious as well as it drives us all to pursue quality. We are so happy for your visibility in our lives. Thanks!
Hello there, simply turned into aware of your blog thru Google, and found that it is really informative. I’m gonna watch out for brussels. I will appreciate when you continue this in future. Many folks might be benefited from your writing. Cheers!
Hello there! This blog post could not be written any better! Reading through this post reminds me of my previous roommate! He constantly kept preaching about this. I most certainly will forward this post to him. Pretty sure he will have a very good read. Many thanks for sharing!
Obtain one year totally free plus approximately 40 off your very first subscription!
Have a good time as well as get points finished with one of the most impressive device!
This web site definitely has all the information and facts I wanted concerning this subject and didn’t know who to ask.
I absolutely love your website.. Pleasant colors & theme. Did you create this amazing site yourself? Please reply back as I’m planning to create my very own blog and would love to learn where you got this from or exactly what the theme is named. Many thanks!
Каждый месяц я оплачиваю аренду квартиры, и в этот раз деньги на счету закончились раньше времени. Не зная, что делать, я нашла сайт credit-info24.ru. Этот портал собрал все МФО в одном месте и предложил [url=https://credit-info24.ru/]деньги до зарплаты на карту без отказа[/url]. Спасибо этому сервису, я смогла оплатить аренду в срок и избежать неприятных моментов с арендодателем.
Я всегда стараюсь быть в курсе новинок кино и телевидения. Поэтому мне было приятно узнать, что на hdserialclub.net уже можно [url=https://hdserialclub.net/serials2023/]смотреть сериалы 2023 года онлайн[/url]. Это отличный шанс заранее ознакомиться с будущими хитами и быть в курсе всех кинематографических трендов.
This website was… how do I say it? Relevant!! Finally I have found something that helped me. Cheers!
Казино Вавада предлагает захватывающий мир азартных игр и возможность выиграть крупные суммы денег. Чтобы начать играть, необходимо [url=https://play-casino-vavada.online/]пройти регистрацию в Vavada[/url].
[url=https://play-casino-vavada.online/]Регистрация в Vavada[/url] очень простая и быстрая.. Вам потребуется заполнить небольшую форму, указав свои личные данные, такие как имя, фамилия, электронная почта и номер телефона. Пожалуйста, убедитесь, что вводите правильные данные, чтобы избежать проблем при выводе выигрышей.
После заполнения формы вам будет предложено создать уникальное имя пользователя и пароль для входа в ваш аккаунт. Помните, что безопасность вашего аккаунта важна, поэтому рекомендуется использовать надежные пароли, состоящие из разных символов.
После завершения регистрации просто подтвердите свой аккаунт, перейдя по ссылке, которая придет вам на почту. После подтверждения авторизоваться и сразу начать играть Vavada.
Не забудьте прочесть правила, чтобы знать все возможные ограничения и требования. Важно играть ответственно и устанавливать лимиты для себя, чтобы не превысить свои финансовые возможности.
Удачи в [url=https://play-casino-vavada.online/]официальный сайт vavada[/url]! Наслаждайтесь азартом и возможностью выиграть большие призы!
The general tone of your message was both authoritative and personalized, making it simple for viewers to associate with.
Its like you read my mind! You seem to know so much about this, like you wrote the book in it or something. I think that you could do with some pics to drive the message home a bit, but other than that, this is wonderful blog. An excellent read. I’ll definitely be back.
You are a wonderful source of motivation for me. You are a excellent resource of inspiration for me.
I’m gone to say to my little brother, that he should also visit this website on regular basis to take updated from most recent news.
You efficiently made use of individual examples to support your disagreements.
The level of imagination you made use of in your post was impressive, making it stand out from others.
Hello just wanted to give you a quick heads up. The text in your post seem to be running off the screen in Ie. I’m not sure if this is a format issue or something to do with web browser compatibility but I thought I’d post to let you know. The design and style look great though! Hope you get the problem resolved soon. Cheers
Making use of visuals in your post was best for comprehending complicated concepts rapidly.
Группа «Каспий» родилась в конце 2012 года, в Москве, когда Володя Астапчик собрал вокруг себя группу музыкантов-единомышленников. Уже через полгода, летом 2013 года команда, названная «Каспий», дебютировала на фестивалях «Нашествие» и «Доброфест». Мелодичность, драйв и многослойная лирика текстов помогли группе стремительно завоевать Интернет- и офф-лайн аудиторию.
[url=https://kaspiymusic.ru/]песни группы каспий[/url]
Фантастика сериалы смотреть онлайн hdserialclub.net
Онлайн смотреть сериалы на надежных ресурсах – одно из лучших времяпрепровождения в зимнюю пору. Что может быть приятнее чем, после тяжкого рабочего дня расположиться удобно с чашкой горячего чая и под любимый сериал довольствоваться этой жизнью?! На ресурсе hdserialclub.net Вы найдете большое количество сериалов, которые можно посмотреть в HD качестве и с четкой русской озвучкой.
Если Вы хотели найти [url=https://hdserialclub.net/fantastika/]сериалы фантастика онлайн[/url] в сети интернет, то Вы попали по правильному адресу. Весьма многие девушки встречаются с вопросом: что увидеть? Особенно яростные жрицы сериалов, которые, казалось бы, пересмотрели всё, что возможно. Но это не имеет ничего общего с действительностью.
Наш веб портал hdserialclub.net тому доказательство. Мы разместили в нашем списке все самые интересные и знаменитые сериалы. Есть такие жанры, как: драмы, боевики, криминал, триллеры, фантастика, исторические и многие другие. Выбрав конкретную категорию, вы ощутимо уменьшите время на разыскивание самого классного для Вас сериала.
Переходите в ТОП [url=https://hdserialclub.net/best-serials/]лучшие сериалы в хорошем качестве[/url] уже сегодня и смотрите бесплатно любой сериал. При поиске в сети интернет надежного сайта для просмотра, обычно люди встречаются с ужасной озвучкой или плохим качеством изображения. У нас Вы найдете только лучшие варианты по поиску из всей сети интернет. Самый удобный интерфейс, простой выбор серии, просмотр с момента окончания и другие преимущества.
Практичность просмотра на сайте hdserialclub.net заключается в том, что Вы можете смотреть серии, когда удобно конкретно Вам. При просмотре сериалов на телевидении, нужно ожидать конкретное время, сидеть возле телевизора и также смотреть кучу рекламы. Просмотр на нашем онлайн сайте неограничен, Вы сами решаете где, когда и во сколько будет начинаться новая серия. Сохраняйте наш сайт, советуйте близким и обменивайтесь мнением от просмотра лучших сериалов 2023 прямо сегодня.
Когда срочно нужны деньги, каждая минута на счету. Но куда обратиться, если банки требуют массу документов и времени на проверку? Ответ прост – воспользуйтесь услугой [url=https://revivalfife.ru/]быстрый займ на карту[/url]. Вам не придется ждать или объяснять, зачем вам средства. Просто заполните заявку онлайн, и в течение 15 минут деньги будут у вас на карте. Это удобно, это быстро, и что самое главное – это доступно в любое время дня и ночи. Позвольте себе дышать свободнее уже сегодня с нашими быстрыми займами.
Смотреть сериалы 2023 hdserialclub.net
Сериалы в HD качестве смотреть на проверенных сайтах – одно из лучших времяпрепровождения в холодную погоду. Что может быть лучше чем, после тяжелого рабочего дня устроиться удобно с чашкой горячего чая и под любимый сериал довольствоваться этой жизнью?! На сайте hdserialclub.net Вы сможете найти большое количество сериалов, которые можно посмотреть в HD качестве и с хорошей русской озвучкой.
Если Вы искали [url=https://hdserialclub.net/serials2023/]сериалы 2023 онлайн в хорошем качестве[/url] в интернете, то Вы пришли по верному адресу. Весьма многие девушки задаются вопросом: что увидеть? Особенно постоянные любительницы сериалов, которые, вроде бы, пересмотрели всё, что возможно. Но это не имеет ничего общего с действительностью.
Данный веб портал hdserialclub.net тому подтверждение. Мы выложили в нашем списке все самые лучшие и популярные сериалы. Представлены такие жанры, как: ужасы, боевики, фэнтези, детективы, фантастика, исторические и многие другие. Выбрав конкретную категорию, вы значительно урежете время на разыскивание самого интересного для Вас сериала.
Переходите в каталог [url=https://hdserialclub.net/priklucheniya/]сериалы приключения онлайн[/url] уже сегодня и смотрите бесплатно любой сериал. При поиске в интернете хорошего сайта для просмотра, обычно люди встречаются с некачественной озвучкой или ужасным качеством картинки. У нас Вы найдете только самые лучшие варианты по поиску из всей паутины. Самый удобный интерфейс, легкий выбор серии, просмотр с момента окончания и другие плюсы.
Практичность просмотра на сайте hdserialclub.net заключается в том, что Вы можете смотреть серии, когда удобно конкретно Вам. При просмотре сериалов на тв, нужно ожидать определенное время, сидеть около телевизора и также смотреть кучу рекламных роликов. Просмотр на нашем интернет сайте неограничен, Вы сами решаете где, что и во сколько будет начинаться следующая серия. Сохраняйте наш сайт, рекомендуйте родным и делитесь мнением от просмотра лучших сериалов 2023 уже сейчас.
The level of creativity you made use of in your article went over, making it attract attention from others.
Современный рынок предлагает нам множество уникальных решений, включая штукатурку механизированную. Проверьте mehanizirovannaya-shtukaturka-moscow.ru для получения подробной информации.
The way you incorporated different components into one cohesive story went over.
Попробуйте механизированную штукатурку от mehanizirovannaya-shtukaturka-moscow.ru и убедитесь в ее эффективности. Ваш дом заслуживает лучшего!
Сериалы турецкие в высшем качестве turksezon.tv
Сегодня турецкие сериалы очень знамениты, ведь в них есть всё что удерживает поклонников. Интересная история, красивые локации, главные роли и, естественно, история про любовь. Многие девушки в России просто не представляют себе жизни без турецких сериалов. А началось всё с того, как по телевизору показали «Великолепный век» – самый известный и распространенный турецкий сериал. Девушки с огромным интересом стали смотреть и другие турецкие сериалы, а турки начали выпускать множество картин.
Советуем сайт turksezon.tv для просмотра онлайн сериалов полностью бесплатно. У нас размещено большое количество самых известных и, наоборот, незнакомых большинству в России. Разные жанры, герои, сюжеты – любой сможет найти то, что ему по душе. Вы можете воспользоваться категориями поиска для удобства в поиске необходимого Вам сериала. Это: по году выпуска, по просмотрам, по отзывам или по алфавиту.
Если Вы искали [url=https://turksezon.tv/detektivy/]турецкие сериалы детективы[/url] в сети интернет, то уже сейчас переходите на указанный онлайн сайт. Сегодня телевизор смотрят очень немногие, а всё оттого, что реклама забирает большое количество времени, нет возможности всегда быть в конкретное время у экрана, абстрагируясь от важных дел.
Другое дело – смотреть сериалы по ноутбуку, планшету или смартфону. Современная девушка может делать сразу несколько дел, совмещая приятное с продуктивным. Например, заниматься рукоделием за просмотром любимого сериала. Или делать уборку и наблюдать за своими любимыми актерами. Указанный сайт создан для таких современных и жизнерадостных представительниц прекрасного пола. Но не только женщины смотрят турецкие сериалы, многие мужчины также выбирают наш сайт.
Прямо сейчас [url=https://turksezon.tv/new-turkish-serials/]смотреть новые турецкие сериалы онлайн[/url] вы найдете у нас. Самые известные сериалы: Моя левая сторона, Дело чести, Осуждение, Мерьем, Меня зовут Фарах, Ранняя пташка и многие другие Вы найдете на сайте turksezon.tv в высоком качестве. Все сериалы можно смотреть бесплатно и на русском языке.
«СК Сити Строй”: ваш идеальный ремонт начинается здесь
ООО «СК СИТИ СТРОЙ» – это мастерство и качество, воплощенное в каждом квадратном метре вашего дома. Обратившись к нам за услугой [url=https://remont-siti.ru/]ремонт под ключ[/url], вы получите не просто обновление интерьера, но и вдохновение новым пространством, созданным с учетом всех ваших пожеланий. Мы гордимся тем, что предлагаем решения, которые делают жизнь наших клиентов более комфортной и стильной.
С первого штриха карандаша дизайнера до последнего штриха кисти маляра — ваш проект находится в руках профессионалов. Наша команда заботится о том, чтобы процесс ремонта был для вас приятным и беззаботным. Мы находимся в Москве по адресу: ул. Новослободская, д. 20, к. 27, оф. 6, и с нетерпением ждем встречи с вами для обсуждения вашего проекта. Пусть ваш дом станет источником вдохновения с ООО «СК СИТИ СТРОЙ».
You bring pleasure and also happiness wherever you go. Your resolution to be successful goes over.
You’re going to delight in every element of your life a lot more.
You actually mentioned it perfectly!
essay writing service in 1 hour [url=https://essayservicehelp.com/]mba resume writing service[/url] linkedin profile writing service reviews uk
Absolutely nothing can be like this!
Your message was loaded with fresh ideas and principles.
Индийские сериалы — это целый мир, полный драматизма, великолепных костюмов и увлекательных сюжетов. Они позволяют зрителям погрузиться в жизнь героев, их любовные перипетии, семейные драмы и социальные конфликты. В отличие от болливудских фильмов, сериалы предлагают более подробное и глубокое изучение характеров и обстоятельств. И теперь, благодаря интернету, любой желающий может [url=https://turksezon.tv/indiyskie_seriali/]смотреть индийские сериалы онлайн[/url] абсолютно бесплатно и в удобное время.
Среди множества доступных сериалов есть те, которые уже успели завоевать любовь русскоязычной аудитории. Примеры таких сериалов — «Согласие», «Женская доля» и «Верни мою любовь». Они предлагают зрителям не только интересные истории, но и возможность погрузиться в культуру и обычаи Индии, что делает просмотр особенно познавательным.
Amazingness is the performance app that will transform your life.
Remarkable ability to obtain points done.
Время неумолимо, и для врача, стремящегося к росту и развитию в своей профессии, нет ничего важнее постоянного обучения и совершенствования. Когда мне стало ясно, что для дальнейшего продвижения мне необходимо получить следующую квалификационную категорию, я столкнулся с проблемой выбора подходящей формы обучения. Работа в клинике занимает большую часть времени, и учеба должна была гармонично вписаться в мой плотный график. Решение пришло внезапно и стало настоящим прорывом — я узнал о возможности [url=https://maps-edu.ru/poluchenie-medicinskoy-kategorii]получить квалификационную категорию врача дистанционно[/url].
Первое, что поразило меня, — это удобство и гибкость обучения. Все лекции, семинары и материалы были доступны онлайн, что позволяло мне самостоятельно планировать свой образовательный процесс. Это было настоящим спасением, ведь я мог учиться в вечернее время или в выходные, не отрываясь от важной работы с пациентами.
Программа курса была насыщена актуальной информацией, которую представляли ведущие специалисты в различных областях медицины. Они не только делились знаниями, но и вдохновляли на дальнейшие исследования и практическое применение полученных данных.
Обучение дало мне не просто новые баллы и документ о повышении квалификации, но и уверенность в своих силах, свежие идеи для работы и желание расти дальше. Сейчас, спустя некоторое время после окончания курса, я могу сказать, что это решение стало одним из лучших в моей карьере.
Так что если вы рассматриваете возможность [url=https://maps-edu.ru/poluchenie-medicinskoy-kategorii]получить квалификационную категорию врача дистанционно[/url], не сомневайтесь. Это удобно, эффективно и, безусловно, стоит того времени и усилий, которые вы в это вложите. Ваш профессиональный путь заслуживает того, чтобы на нем были самые лучшие достижения.
Также советуем вам обратить внимание на [url=https://maps-edu.ru/akkreditaciya-medrabotnikov]первичная аккредитация медицинских работников[/url] и ознакомиться с этим по лучше. У нас на сайте maps-edu вы можете задать вопросы и получить консультацию.
Адрес: Иркутск, ул. Степана Разина, дом 6, офис 405.
Keeping track of time shouldn’t be expensive. Discover our unbeatable [url=https://furfurfriend.com/]watch prices[/url] and elevate your style without emptying your wallet.
В современном мире, где медицинские знания обновляются ежедневно, непрерывное обучение врачей является не просто желанием, а настоятельной необходимостью. Мы представляем вам передовой курс [url=https://maps-edu.ru/nabor-ballov-nmo]нмо обучение врачей[/url], который станет вашим надежным помощником в достижении новых профессиональных вершин.
Программа обучения разработана с учетом последних трендов и инноваций в медицине, а также индивидуальных потребностей современных специалистов. Наши курсы позволят вам не только укрепить имеющиеся знания и навыки, но и изучить новейшие методики и подходы в лечении.
Выбрав нас, вы получаете:
Эксклюзивные учебные материалы, составленные ведущими экспертами в различных областях медицины.
Индивидуальный план обучения, который учитывает ваш профессиональный опыт и личные цели.
Возможность общения и обмена опытом с коллегами из разных регионов и специализаций.
Гибкость в обучении благодаря дистанционному формату, который позволяет совмещать работу и учебу без ущерба для качества обоих процессов.
Закончив курс [url=https://maps-edu.ru/nabor-ballov-nmo]нмо обучение врачей[/url], вы не просто наберете необходимые баллы НМО, но и значительно улучшите свои профессиональные компетенции, что, безусловно, скажется на качестве предоставляемых вами медицинских услуг.
Не упускайте возможность стать частью нового поколения медиков, вооруженных знаниями и умениями, соответствующими международным стандартам. Станьте участником нашей образовательной программы уже сегодня и сделайте вклад в свое будущее и будущее медицины.
Также советуем вам обратить внимание на [url=https://maps-edu.ru/nabor-ballov-nmo]баллы нмо для медсестер[/url] и ознакомиться с этим по лучше. У нас на сайте maps-edu вы можете задать вопросы и получить консультацию.
Адрес: Иркутск, ул. Степана Разина, дом 6, офис 405.
Maps-edu.ru – ваш путь к успеху в медицине. Мы предлагаем курсы по нострификации документов и помогаем медсёстрам и врачам получить необходимые баллы НМО.
Great post.
Currently it looks like Drupal is the top blogging platform out there right now. (from what I’ve read) Is that what you’re using on your blog?
Your excitement urges others to do their best. You are an remarkable problem-solver.
Your passion is inspiring! I am so happy to have you in my life.
Русская озвучка турецких сериалов turkhit.tv
Для поклонников турецких сериалов, которые не могут жить без большого количества серий и волнующих сюжетов — мы разработали наш сайт. Самый широкий каталог сериалов из Турции любых жанров. Добро пожаловать на turkhit.tv уже сейчас.
По вопросу [url=https://turkhit.tv/best-turkey-serials/]лучшие турецкие сериалы онлайн[/url] Вы на правильном пути. Турецкие сериалы за последнее время завоевали огромную востребованность за пределами своей страны, в том числе и в РФ. В чем же секрет их успеха? Первоначально, это истории, наполненные напряжением и любовью, которые приковывают зрителя с первых секунд просмотра. Герои сериалов – активные и эмоциональные, постоянно оказываются втянутыми в трудные жизненные ситуации, которые вызывают у аудитории сострадание и сопереживание.
Киносценарии турецких сериалов часто трудные и имеют несколько сюжетных линий. Они совмещают в себе любовные перипетии, семейные драмы, несчастья и предательства, а также детективные линии. Такой подход позволяет удерживать внимание различной аудитории, ведь любой найдет для себя что-то своё.
Кроме того, турецкие сериалы часто задевают сложные социальные и просвященные темы, актуальные не только для Турции, но и для многих других стран. Это делает их не только причиной развлечений, но и пищей для размышлений. Красивые декорации, наряды и съемки в интересных локациях добавляют визуальное наслаждение и помогают зрителям погрузиться в удивительный мир, который они создают.
Заходите посмотреть [url=https://turkhit.tv/]турецкий сериал смотреть онлайн на русском[/url] на следующий сайт turkhit.tv прямо сейчас. Турецкие продюсеры и сценаристы не боятся творить новое с форматами и сюжетными линиями, обязательно улучшая свои техники съемки и монтажа. Актерский состав часто содержит в себе яркие и гениальные личности, годные на естественную игру эмоций и создание убедительных героев.
В заключение, турецкие сериалы цепляют зрителей своей способностью рассказывать увлекательные истории, которые отражают человеческие переживания во всем их многообразии. Они открывают дверь в иностранные культуры и предлагают возможность для эмоционального отголоска, что делает их необходимой частью жизни многих любителей сериалов.
Турецкие сериалы смотреть на русском языке turkhit.tv
Для фанатов турецких сериалов, которые не могут жить без множества серий и волнующих сюжетов — мы образовали наш сайт. Самый большой каталог сериалов из Турции различных жанров. Приглашаем на turkhit.tv прямо сейчас.
По запросу [url=https://turkhit.tv/]смотреть турецкие сериалы на русском языке[/url] Вы на нужном пути. Турецкие сериалы за последнее время забрали огромную популярность за пределами своей страны, в том числе и в РФ. В чем же секрет их успеха? Первоначально, это истории, наполненные напряжением и любовью, которые цепляют зрителя с первых минут просмотра. Герои сериалов – живые и эмоциональные, постоянно оказываются впутанными в сложные жизненные ситуации, которые вызывают у аудитории сочувствие и сопереживание.
Киносценарии турецких сериалов обычно трудные и имеют несколько сюжетных линий. Они сочетают в себе любовные перипетии, семейные драмы, несчастья и подставы, а также детективные линии. Данный подход дает удерживать внимание разнообразной аудитории, ведь любой найдет для себя что-то своё.
Помимо того, турецкие сериалы часто задевают глубокие общественные и культурные темы, актуальные не только для Турции, но и для большинства других стран. Это делает их не только источником развлечений, но и пищей для мыслей. Богатые декорации, наряды и съемки в интересных локациях добавляют визуальное наслаждение и помогают зрителю углубиться в удивительный мир, который они произвели.
Заходите посмотреть [url=https://turkhit.tv/fentezi/]турецкие сериалы фэнтези[/url] на следующий сайт turkhit.tv прямо сейчас. Турецкие продюсеры и сценаристы не боятся творить новое с форматами и сюжетными линиями, обязательно улучшая свои техники съемки и монтажа. Актерский состав часто содержит в себе яркие и талантливые личности, способные на естественную игру эмоций и создание убедительных образов.
В итоге, турецкие сериалы цепляют зрителей своей способностью показывать увлекательные истории, которые отражают человеческие чувства во всем их различии. Они открывают дверь в иностранные культуры и дают возможность для эмоционального отголоска, что делает их необходимой частью жизни многих любителей сериалов.
Лучшие турецкие сериалы turkline.tv
Турецкие сериалы стали чистым культурным феноменом, заслужив аудиторию по всей планете своими интересными сюжетами и необычными актерами. Эти сериалы насыщены глубокими чувствами, непредсказуемыми поворотами и разнятся отличным качеством продукции. Они позволяют зрителям окунуться в мир истинной любви и драмы, при этом затрагивая современные социальные проблемы, что делает их интересными для большой аудитории.
Относительно [url=https://turkline.tv/komedii/]турецкие сериалы комедии[/url] в интернете, то Вы на правильном пути. На онлайн портале turkline.tv представлена уникальная коллекция турецких сериалов, выложенных в высоком качестве на русском языке. Тут можно найти сериалы на всякий вкус – от любовных комедий до захватывающих триллеров и драм. Благодаря комфортному интерфейсу и большому выбору жанров, любой зритель сайта сможет выбрать сериал по вкусу.
Онлайн смотреть [url=https://turkline.tv/]турецкие сериалы русская озвучка[/url] в высоком качестве возможно у нас. Здесь размещены все жанры кино: боевики, фэнтези, криминал, триллеры, мелодрамы и другие. Также возможно пользоваться поиском по дате выпуска или по рейтингу актеров. Турецкие артисты также имеют свою популярность в виде рейтинга. Какой-то актер сразу всем нравится, а некоторые вызывают недоброжелательность. На данном сайте можете посмотреть, кто именно понравился большинству зрителей.
Одним из главных преимуществ turkline.tv является просмотр без рекламы, что дает качественный и удобный просмотр. Все сериалы открыты для просмотра в любое время, что дает зрителям собственно планировать своё свободное время и радоваться интересными историями без ограничений. Таким образом, наш интернет портал является прекрасным ресурсом для всех фанатов турецких сериалов, предлагая высококачественный и доступный контент для определенных ценителей этого жанра.
Смотреть турецкие сериалы бесплатно turkline.tv
Турецкие сериалы стали настоящим культурным фактом, заслужив зрителей по всей планете своими захватывающими сюжетами и яркими персонажами. Эти сериалы насыщены глубокими эмоциями, неизвестными поворотами и отличаются отличным качеством изображения. Они позволяют зрителям уйти с головой в мир реальной любви и драмы, при этом затрагивая насущные социальные проблемы, что делает их интересными для широкой аудитории.
Что касается [url=https://turkline.tv/fentezi/]турецкие сериалы фэнтези[/url] в сети интернет, то Вы на верном пути. На интернет сайте turkline.tv размещена уникальная коллекция турецких сериалов, выложенных в высоком качестве с русской озвучкой. Здесь можно найти произведения на любой вкус – от любовных комедий до будуражащих триллеров и драм. Благодаря удобному интерфейсу и широкому выбору жанров, любой посетитель сайта сможет выбрать сериал по вкусу.
Посмотреть [url=https://turkline.tv/melodramy/]турецкие сериалы мелодрамы[/url] в высоком качестве возможно у нас. Здесь представлены все жанры кино: боевики, фэнтези, криминал, семейные, фантастика и другие. Также можно пользоваться поиском по году выпуска или по рейтингу героев. Турецкие артисты также имеют свою известность в виде рейтинга. Какой-то актер сразу всем нравится, а некоторые вызывают антипатию. На указанном сайте можете посмотреть, кто именно полюбился большинству зрителей.
Одним из основных плюсов turkline.tv является просмотр без рекламы, что обеспечивает качественный и удобный просмотр. Все сериалы открыты для просмотра 24 на 7, что дает зрителям собственно планировать своё свободное время и радоваться любимыми историями без ограничений. Таким образом, наш интернет портал является отличным ресурсом для всех зрителей турецких сериалов, предлагая качественный и доступный контент для истинных ценителей любого жанра.
You are an amazing pal! You are an impressive friend!
Hey very interesting blog!
Very good info. Cheers.
essay writing service illegal [url=https://essayservicehelp.com/]poem writing service[/url] affordable essay writing service
В условиях высокой конкуренции, особенно в Москве, [url=https://www.umseo.ru/]сео продвижение заказать[/url] требует индивидуального подхода. Это включает в себя не только традиционные методы SEO, но и креативные решения, такие как вирусный маркетинг и SMM. Эффективное продвижение поможет вашему сайту выделиться и привлечь больше целевой аудитории.
Для того чтобы ваш сайт стал популярным в Москве, [url=https://www.umseo.ru/]стоимость продвижения сайта в москве[/url] – это то, что вам нужно. Это комплекс мероприятий, направленных на увеличение видимости вашего сайта в поисковых системах и социальных сетях. Важно выбрать агентство, которое имеет опыт работы именно в вашей нише.
Do you have any video of that? I’d like to find out some additional information.
Nicely put. Thanks!
essay writing service quora [url=https://topswritingservices.com/#]essay services[/url] expository essay writing service
Русская озвучка турецких сериалов turkhit.tv
Для любителей турецких сериалов, которые не могут жить без множества серий и интригующих сюжетов — мы образовали наш веб портал. Самый огромный каталог сериалов из Турции любых жанров. Приглашаем на turkhit.tv прямо сейчас.
По теме [url=https://turkhit.tv/]бесплатные турецкие сериалы[/url] Вы на правильном пути. Турецкие сериалы за последние годы завоевали огромную востребованность за пределами своей страны, в том числе и в РФ. В чем же секрет их успеха? Прежде всего, это истории, наполненные напряжением и страстью, которые приковывают зрителя с первых секунд просмотра. Персонажи сериалов – живые и эмоциональные, постоянно оказываются впутанными в трудные жизненные обстоятельства, которые вызывают у зрителей сочувствие и сопереживание.
Сценарии турецких сериалов обычно сложны и многоуровневы. Они сочетают в себе любовные разборки, семейные трудности, трагедии и подставы, а также детективные линии. Данный подход позволяет удерживать внимание различной аудитории, ведь каждый найдет для себя что-то своё.
Помимо того, турецкие сериалы часто затрагивают сложные социальные и культурные темы, острые не только для Турции, но и для многих других стран. Это делает их не только источником развлечений, но и пищей для размышлений. Шикарные декорации, костюмы и съемки в живописных локациях добавляют визуальное наслаждение и помогают зрителям углубиться в необыкновенный мир, который они создают.
Переходите смотреть [url=https://turkhit.tv/best-turkey-serials/]смотреть лучшие турецкие сериалы онлайн[/url] на наш сайт turkhit.tv уже сейчас. Турецкие продюсеры и сценаристы не боятся экспериментировать с форматами и сюжетными линиями, постоянно улучшая свои техники съемки и монтажа. Актерский состав часто содержит в себе яркие и гениальные личности, способные на глубокую игру эмоций и создание убедительных героев.
В заключение, турецкие сериалы цепляют зрителей своей способностью показывать удивительные истории, которые отражают человеческие чувства во всем их многообразии. Они открывают окно в иностранные культуры и дают возможность для эмоционального ответа, что делает их незаменимой частью жизни многих ценителей сериалов.
Самые интересные турецкие сериалы turkline.tv
Турецкие сериалы стали чистым культурным фактом, завоевав аудиторию по всей планете своими интересными сюжетами и яркими персонажами. Эти сериалы наполнены глубокими эмоциями, непредсказуемыми поворотами и отличаются высоким качеством продукции. Они дают зрителям впасть в мир реальной любви и драмы, при этом задевая современные социальные проблемы, что делает их любимыми для широкой аудитории.
Что касается [url=https://turkline.tv/]турецкие сериалы в русской озвучке[/url] в интернете, то Вы на правильном пути. На интернет сайте turkline.tv размещена исключительная коллекция турецких сериалов, выложенных в лучшем качестве на русском языке. Здесь можно найти сериалы на всякий вкус – от любовных комедий до захватывающих триллеров и детективов. Благодаря удобному интерфейсу и большому выбору жанров, каждый зритель сайта сможет выбрать сериал по вкусу.
Смотреть [url=https://turkline.tv/]турецкий сериал смотреть онлайн на русском[/url] в хорошем качестве можно у нас. Здесь представлены такие жанры кино: драмы, фэнтези, военные, семейные, фантастика и другие. Также возможно воспользоваться поиском по времени выпуска или по рейтингу актеров. Турецкие актеры также имеют свою популярность в виде рейтинга. Какой-то актер сразу всем нравится, а некоторые вызывают антипатию. На нашем сайте можете увидеть, кто именно понравился большинству зрителей.
Одним из основных преимуществ turkline.tv является отсутствие рекламы, что дает непрерывный и комфортный просмотр. Все сериалы открыты для просмотра 24 на 7, что позволяет зрителям самостоятельно планировать свой досуг и радоваться любимыми историями без ограничений. Таким образом, наш веб ресурс является прекрасным ресурсом для всех зрителей турецких сериалов, предлагая высококачественный и открытый контент для определенных ценителей любого жанра.
Естественное освещение является важным аспектом в дизайне любого жилого или рабочего пространства. Оно не только улучшает внешний вид помещения, но и способствует повышению комфорта и благополучия его обитателей. Компания Solargy предлагает уникальное решение в области [url=https://solargy.ru/]естественного освещения помещений[/url], обеспечивая качественное освещение даже в тех местах, где традиционные методы неприменимы.
Использование световодов Solargy позволяет максимально эффективно использовать дневной свет, транспортируя его из наружного пространства внутрь зданий. Это способствует не только экономии электроэнергии, но и создает более здоровую среду за счет уменьшения использования искусственного освещения. Эти системы особенно эффективны в многоэтажных зданиях, где естественный свет имеет решающее значение для создания приятной атмосферы.
Технология световодов Solargy подходит для широкого спектра применений, от жилых домов до больших коммерческих и общественных зданий. Она обеспечивает равномерное распределение света, устраняя затемненные углы и создавая более привлекательное визуальное восприятие пространства. Кроме того, установка световодов — это экологически ответственный выбор, способствующий снижению углеродного следа здания.
Оптимизация естественного освещения с помощью продукции Solargy — это не только практичное, но и экономически выгодное решение. Системы освещения Solargy удовлетворяют самым высоким стандартам качества и эффективности, делая [url=https://solargy.ru/]естественное освещение помещений[/url] доступным и эффективным для широкого круга потребителей.
Системы солнечного освещения от компании Solargy представляют собой инновационный подход к освещению помещений, используя возобновляемый источник энергии – солнечный свет. Эти системы не только экологически чистые, но и эффективные в плане экономии энергии. Подробнее об этом можно узнать на странице [url=https://solargy.ru/]системы солнечного освещения[/url].
Основное преимущество солнечных систем освещения заключается в их способности преобразовывать солнечный свет в яркое и комфортное освещение для любого типа помещений, будь то жилые, коммерческие или общественные здания. Использование световодов позволяет доставить естественный свет даже в самые темные и отдаленные уголки здания, обеспечивая тем самым равномерное распределение освещения.
Эти системы особенно ценны в условиях, где естественное освещение ограничено или недоступно. Например, в подвальных помещениях или внутренних комнатах без окон. Таким образом, системы солнечного освещения Solargy не только улучшают внешний вид и атмосферу помещения, но и способствуют созданию здоровой среды для жизни и работы.
Кроме того, использование солнечного света в качестве основного источника освещения помогает снизить потребление электроэнергии, что ведет к уменьшению углеродного следа здания и снижению эксплуатационных расходов. Системы Solargy легко адаптируются к различным архитектурным стилям и требованиям, делая их идеальным решением для любого проекта, где важны экологичность и эффективность.
Your post was an superb example of content that is both insightful as well as amusing.
На сайте мир-займов.рф каждый может найти оптимальное решение для своих финансовых потребностей – [url=https://xn—-8sbgsdjqfso.xn--p1ai/]список займов без отказа[/url] . Мы предлагаем широкий выбор займов без отказа, что особенно ценно для тех, кто ищет быстрый и надежный способ получения финансовой помощи. Наш сервис гарантирует простоту и скорость обработки заявок, минимизируя время ожидания и избегая сложных процедур проверки.
Мир-займов.рф также предлагает услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]кредит на карту онлайн без отказа[/url] , где клиенты могут получить микрозаймы без отказа. Это идеальный выбор для тех, кто нуждается в срочных денежных средствах без лишних задержек. Наша цель – обеспечить вам доступ к финансам в любое время, без необходимости проходить через длительный процесс одобрения.
Мы понимаем, как важно быстро решить финансовые проблемы, и именно поэтому наш сайт предлагает эффективные решения для получения займов. С мир-займов.рф вы можете быть уверены, что ваша финансовая потребность будет удовлетворена максимально оперативно и с минимальными требованиями. Займы без отказа и микрозаймы без отказа на мир-займов.рф – это ваш надежный финансовый помощник в любой ситуации.
Когда вам нужно решить финансовые вопросы здесь и сейчас, МИР-ЗАЙМОВ.РФ предлагает идеальное решение: [url=https://xn—-8sbgsdjqfso.xn--p1ai/]где дают займ без отказа[/url] . Кредит онлайн без отказа мгновенно – это возможность получить необходимые средства без длительного ожидания и сложных процедур одобрения. Наша платформа обеспечивает доступ к широкому спектру предложений от МФО, которые готовы предоставить вам финансовую поддержку в короткие сроки.
Для тех, кто ищет возможность быстро получить финансовую помощь, мы предлагаем [url=https://xn—-8sbgsdjqfso.xn--p1ai/]получить займ на карту онлайн без отказа[/url] . Взять займ быстро и без отказа можно прямо на нашем сайте, где собраны предложения от различных кредиторов. Мы стремимся к тому, чтобы каждый наш клиент мог легко найти подходящее решение для своих потребностей, обеспечивая при этом высокий уровень сервиса и оперативность обработки заявок.
В ситуации, когда срочно нужны деньги, наш сервис предоставляет удобное и оперативное решение – [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрозайм круглосуточно без отказа[/url] . Взять кредит онлайн на карту без отказа теперь стало проще, чем когда-либо. Благодаря широкому выбору предложений от различных МФО, вы сможете найти оптимальное решение, соответствующее вашим потребностям, без длительных процедур проверки и ожидания одобрения.
Кроме того, если вам нужны деньги немедленно, мы предлагаем услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]займ на карту без отказа без проверки[/url] . Займы онлайн на карту без отказа срочно – это быстрый способ получения необходимой суммы без лишних формальностей. Мы понимаем, как иногда бывает важно оперативно решить финансовые вопросы, и поэтому наша платформа работает так, чтобы предоставить вам средства в самые короткие сроки.
Заказать баннер недорого Москва баннер-москва.рф
Советуем Вам компанию, которая промышляет разной широкоформатной печатью в Москве разных видов. Мобильные стенды, постпечатная обработка, печать на баннерной сетке, печать на бумаге и многое другое Вы сможете найти на интернет портале баннер-москва.рф прямо сейчас. Наш интернет портал состоит из большого объема важной информации для наших будущих клиентов, обязательно заходите и отправляйте на заявку.
По вопросу [url=https://баннер-москва.рф/pechat-bannerov]баннер на заказ москва недорого[/url] в интернете, мы с удовольствием Вам поможем. В нашей команде работают реальные профессионалы, которые имеют огромный опыт в сфере печати. Рассматриваем каждый случай индивидуально и предлагаем заказчикам именно то, что они хотели найти. Мы пускаем в ход в работе только качественные материалы: это новое оборудование, яркие чернила, которые прослужат долгое время, прочные пленки и ткани, а также наши специалисты, которые знают в работе толк.
Цена на наши услуги весьма приятная и не преувеличена. А это оттого, что у нас собственное оборудование для разных форматов для любых размеров. Мы баннер-москва.рф сотрудничаем с разными поставщиками, в том числе заграничными, которые представляют лучшее качество материалов по выгодным ценам. А еще мы гордимся, что обычный срок изготовления Вашего заказа чаще всего составляет не более двух дней. Также есть система скидок, которая зависит от размера вашего заказа.
Заказать [url=https://баннер-москва.рф/mobilnye-stendy/press-wall]прессвол цена[/url] прямо сегодня, возможно на нашем сайте. Оставляйте заявку на печать, указав свои личные данные и мы перезвоним Вам. Также можно самостоятельно позвонить по контактному телефону +7(499)390-19-73 или написать на наш электронный адрес. Оплата заказа осуществляется удобным для Вас методом. Находимся по адресу: г. Москва, Нагорный проезд, д. 7, стр.1. Время работы по будням с 10:00 до 18:00. Звоните, пишите, приезжайте и мы ответственно создадим для Вас необходимый баннер.
Вам нужны деньги здесь и сейчас? Нет проблем! Наша платформа предлагает возможность получить [url=https://xn—-8sbgsdjqfso.xn--p1ai/]онлайн займ без отказа[/url] , обеспечивая быстрый и удобный доступ к необходимым средствам. Благодаря сотрудничеству с множеством МФО, мы можем предложить вам деньги на карту сразу без отказа, что особенно ценно в экстренных ситуациях. Забудьте о бумажной волоките и долгих ожиданиях – наш сервис сделан для того, чтобы вы могли получить финансовую помощь быстро и эффективно.
Ищете надежный и быстрый способ получить финансирование? Попробуйте нашу услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрокредит на карту срочно без отказа[/url] . Займ на карту без отказа доступен всем, кто нуждается в срочных денежных средствах. Мы предлагаем простые и понятные условия оформления займа, а также гарантируем быстрое рассмотрение вашей заявки. Воспользуйтесь нашим сервисом, чтобы решить ваши финансовые вопросы максимально оперативно.
Уф печать на самоклеящейся пленке баннер-москва.рф
Рекомендуем Вам компанию, которая занимается разнообразной широкоформатной печатью в Москве разных видов. Интерьерная печать, постпечатная обработка, баннеры, печать на самоклеящейся пленке и многое другое Вы сможете найти на веб ресурсе баннер-москва.рф уже сейчас. Наш веб портал складывается из большого объема полезной информации для наших следующих клиентов, обязательно заходите и оформляйте заявку.
По вопросу [url=https://баннер-москва.рф/mobilnye-stendy/pop-up]pop up стенд купить[/url] в интернете, мы с удовольствием Вам окажем помощь. В нашей команде работают самые настоящие профессионалы, которые имеют огромный опыт в сфере печати. Анализируем любой случай конкретно и предлагаем заказчикам конкретно то, что они искали. Мы пускаем в ход в работе только избранные материалы: это современное оборудование, яркие чернила, которые прослужат длительное время, прочные пленки и ткани, а также наши специалисты, которые знают своё дело.
Цена на предложенные услуги весьма выгодная и не завышенная. А это потому, что у нас личное оборудование для разных форматов для любых размеров. Мы баннер-москва.рф проводим работу с разными компаниями, в том числе зарубежными, которые представляют высшее качество материалов по выгодным расценкам. А еще мы гордимся, что обычный срок выполнения Вашего заказа чаще всего составляет не более двух дней. Также существует система скидок, которая зависит от размера вашего заказа.
Оформить заказ на [url=https://баннер-москва.рф/]широкоформатная печать[/url] прямо сейчас, возможно на нашем сайте. Оформляйте заявку на печать, указав свои личные данные и мы перезвоним Вам. Вдобавок можно самостоятельно позвонить по телефону +7(499)390-19-73 или написать на наш почтовый ящик. Оплата заказа происходит удобным для Вас методом. Расположены по адресу: г. Москва, Нагорный проезд, д. 7, стр.1. График работы с пн по пт с 10:00 до 18:00. Звоните, пишите, приезжайте и мы непременно создадим для Вас предмет искусства.
You actually mentioned it exceptionally well!
play baccarat online real money [url=https://red-dogcasino.online/#]red dog casino no deposit[/url] best crypto casino
В ситуации, когда срочно нужны деньги, наш сервис предоставляет удобное и оперативное решение – [url=https://xn—-8sbgsdjqfso.xn--p1ai/]срочно нужен займ на карту без отказа[/url] . Взять кредит онлайн на карту без отказа теперь стало проще, чем когда-либо. Благодаря широкому выбору предложений от различных МФО, вы сможете найти оптимальное решение, соответствующее вашим потребностям, без длительных процедур проверки и ожидания одобрения.
Кроме того, если вам нужны деньги немедленно, мы предлагаем услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]займ без отказа всем срочно[/url] . Займы онлайн на карту без отказа срочно – это быстрый способ получения необходимой суммы без лишних формальностей. Мы понимаем, как иногда бывает важно оперативно решить финансовые вопросы, и поэтому наша платформа работает так, чтобы предоставить вам средства в самые короткие сроки.
I’m now not positive where you are getting your info, however good topic. I needs to spend a while learning more or understanding more. Thank you for fantastic information I used to be in search of this information for my mission.
Нужны деньги срочно? Сайт мир-займов.рф предлагает легкое решение вашей проблемы – [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрозайм онлайн на карту срочно без отказов[/url] . У нас вы можете оформить займ на карту онлайн без отказа, что идеально подходит для ситуаций, когда время имеет решающее значение. Мы гарантируем быстрое рассмотрение заявок и перевод средств прямо на вашу карту, минимизируя все возможные задержки.
Если вам необходимо срочно [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрозайм на карту онлайн срочно без отказов[/url] , мир-займов.рф – это ваш надежный помощник. Наш сервис позволяет взять займ без отказа на карту, предоставляя вам свободу и гибкость в финансовых вопросах. Мы понимаем, что иногда финансовые потребности возникают неожиданно, и именно поэтому наша платформа работает круглосуточно, предлагая займы без сложных процедур проверки и бюрократии.
Мир-займов.рф предоставляет широкий спектр вариантов займов, чтобы каждый клиент мог выбрать оптимальное решение. Наша цель – сделать процесс получения финансовой помощи максимально удобным и доступным для всех, кто оказался в сложной ситуации.
Ищете надежный источник займов? На МИР-ЗАЙМОВ.РФ представлен широкий [url=https://xn—-8sbgsdjqfso.xn--p1ai/]взять займ быстро и без отказа[/url] , включающий предложения от множества МФО. Эти займы предлагаются без отказов, что значительно упрощает процесс получения необходимых средств. Особенностью нашего сервиса является наличие предложений от МФО, которые предоставляют первый займ без процентов, делая их идеальным выбором для тех, кто впервые обращается за финансовой помощью.
Если вам необходимо [url=https://xn—-8sbgsdjqfso.xn--p1ai/]срочно взять займ без отказа[/url] , наш сайт предоставляет быстрый и удобный способ. Взять кредит онлайн срочно без отказа можно, не выходя из дома. Мы ценим ваше время и стремимся обеспечить максимальную доступность и скорость обслуживания. МИР-ЗАЙМОВ.РФ – это ваш надежный финансовый партнер, готовый помочь в любой ситуации.
Sensational is a desktop app that will certainly transform the means you help the better!
You said that effectively.
free slots 777 no download [url=https://red-dogcasino.online/#]free blackjack games offline[/url] rudolfs revenge
Вам необходима финансовая помощь в любое время суток? Наш сервис предлагает решение: [url=https://xn—-8sbgsdjqfso.xn--p1ai/]кредит без отказа на карту[/url] . Микрозайм круглосуточно без отказа – это ваша возможность получить необходимые средства без ожидания и сложных процедур. Мы понимаем важность доступности и готовы предложить вам услугу, которая работает без перерывов и выходных, обеспечивая вас средствами в любой необходимый момент.
Если вам срочно нужны деньги, наш сервис предоставит вам возможность быстро и легко [url=https://xn—-8sbgsdjqfso.xn--p1ai/]мфо онлайн на карту без отказа срочно[/url] . Микрокредит без отказа доступен всем, кто нуждается в срочном финансировании. Мы предлагаем простую и понятную процедуру оформления, позволяя вам получить необходимую сумму в кратчайшие сроки и без лишних затруднений. Наша цель – облегчить вашу финансовую нагрузку, предлагая надежные и доступные решения.
You actually suggested this very well!
free online slots no download [url=https://reddog-casino.site/#]red dog casino 100 free spins[/url] does casino take credit cards
Wow that was odd. I just wrote an very long comment but after I clicked submit my comment didn’t show up. Grrrr… well I’m not writing all that over again. Anyway, just wanted to say fantastic blog!
I am regular reader, how are you everybody? This piece of writing posted at this website is really good.
Beneficial forum posts. Thank you!
casino games online free play no download [url=https://reddog-casino.site/#]red dog casino free spins no deposit[/url] red dog casino app
Your resolution to be successful goes over. You make me laugh.
You brighten the room with your visibility. I am so proud of all that you have actually completed.
В современном мире, где скорость и доступность играют ключевую роль, мы предлагаем вам надежное решение для финансовых вопросов: [url=https://xn—-8sbgsdjqfso.xn--p1ai/]где дают займ без отказа[/url] . Займ без отказов мгновенно онлайн на карту – это идеальный вариант для тех, кто ищет быстрое и удобное решение своих временных финансовых затруднений. Мы предоставляем вам возможность получить необходимые средства в самые кратчайшие сроки, обеспечивая простоту и оперативность в каждой детали процесса.
Также мы рады предложить вам услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрофинансы без отказа на карту[/url] . Быстрый займ на карту без отказов онлайн – это ваш шанс получить деньги без лишних задержек и бюрократии. Наш сервис разработан для обеспечения максимального комфорта и скорости, позволяя вам быстро решить финансовые вопросы без необходимости посещения банковских учреждений.
Когда вам нужно решить финансовые вопросы здесь и сейчас, МИР-ЗАЙМОВ.РФ предлагает идеальное решение: [url=https://xn—-8sbgsdjqfso.xn--p1ai/]быстрые займы без отказа на карту онлайн[/url] . Кредит онлайн без отказа мгновенно – это возможность получить необходимые средства без длительного ожидания и сложных процедур одобрения. Наша платформа обеспечивает доступ к широкому спектру предложений от МФО, которые готовы предоставить вам финансовую поддержку в короткие сроки.
Для тех, кто ищет возможность быстро получить финансовую помощь, мы предлагаем [url=https://xn—-8sbgsdjqfso.xn--p1ai/]быстрый займ без отказа на карту срочно[/url] . Взять займ быстро и без отказа можно прямо на нашем сайте, где собраны предложения от различных кредиторов. Мы стремимся к тому, чтобы каждый наш клиент мог легко найти подходящее решение для своих потребностей, обеспечивая при этом высокий уровень сервиса и оперативность обработки заявок.
Excellent data. With thanks!
free online slots no download [url=https://reddog-casino.site/#]red dog casino no deposit bonus[/url] red dog slots
Когда возникает неотложная потребность в финансах, наш сервис предлагает быстрое и удобное решение: [url=https://xn—-8sbgsdjqfso.xn--p1ai/]займ без отказа на карту срочно[/url] . Взять срочно займ на карту без отказа можно всего за несколько минут. Это отличный вариант для тех, кто оказался в сложной финансовой ситуации и нуждается в быстрой финансовой помощи. Мы предлагаем прозрачные условия и мгновенное одобрение заявок, обеспечивая вам доступ к необходимым средствам без лишних задержек и бюрократии.
Для тех, кто ищет надежный способ получения денег без лишних хлопот, мы предоставляем услугу [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрозайм срочно без отказа на карту[/url] . Займ онлайн без отказа доступен круглосуточно, что гарантирует вам возможность получить финансовую поддержку в любое время. Наша цель – предложить максимально удобный и быстрый способ получения займов, который подходит каждому, кто нуждается в срочных денежных средствах.
Нужны деньги срочно? Сайт мир-займов.рф предлагает легкое решение вашей проблемы – [url=https://xn—-8sbgsdjqfso.xn--p1ai/]микрофинансы без отказа на карту[/url] . У нас вы можете оформить займ на карту онлайн без отказа, что идеально подходит для ситуаций, когда время имеет решающее значение. Мы гарантируем быстрое рассмотрение заявок и перевод средств прямо на вашу карту, минимизируя все возможные задержки.
Если вам необходимо срочно [url=https://xn—-8sbgsdjqfso.xn--p1ai/]займ срочно без отказов на карту[/url] , мир-займов.рф – это ваш надежный помощник. Наш сервис позволяет взять займ без отказа на карту, предоставляя вам свободу и гибкость в финансовых вопросах. Мы понимаем, что иногда финансовые потребности возникают неожиданно, и именно поэтому наша платформа работает круглосуточно, предлагая займы без сложных процедур проверки и бюрократии.
Мир-займов.рф предоставляет широкий спектр вариантов займов, чтобы каждый клиент мог выбрать оптимальное решение. Наша цель – сделать процесс получения финансовой помощи максимально удобным и доступным для всех, кто оказался в сложной ситуации.
You actually said it exceptionally well!
no deposit bonus free chip [url=https://red-dogcasino.website/#]no deposit bonus codes for existing players[/url] wild casino free chip no deposit
You made use of allegories and also analogies effectively throughout your blog post.
We are really thankful for your relationship as well as commitment. You have been an integral part of this group as well as we would certainly not be where we are today without you!
Your perseverance is inspiring! I appreciate your stamina as well as determination to do well.
Nicely put. Regards.
rudolphs revenge [url=https://red-dogcasino.website/#]reddog no deposit bonus[/url] red cherry casino no deposit bonus
With thanks, A good amount of forum posts.
free slots no downloads with bonus rounds [url=https://red-dogcasino.website/#]red dog casino promo code[/url] $10 deposit online casino
На МИР-ЗАЙМОВ.РФ мы сделали все возможное, чтобы вы могли оформить займ быстро и просто. На нашем портале вы найдете лучшие МФО России, где оформление займа занимает всего 5 минут. Это означает, что вы можете решать свои финансовые вопросы молниеносно, без лишних хлопот. Не откладывайте на потом – посетите МИР-ЗАЙМОВ.РФ и получите нужный займ прямо сейчас!
I ensure this is mosting likely to be your favorite item!
You suggested it well.
play craps online for real money [url=https://reddog-casinos.website/#]cash bandits[/url] online casino free chip no deposit
We are so happy you became part of the project and we can’t thanks enough for all that you have actually done. Your hard work and also devotion have been excellent!
Superb information. With thanks.
red dog no deposit casino bonus codes for existing players [url=https://reddog-casinos.website/#]$10 deposit casino[/url] deuces wild card game free
Иногда информацию можно найти не только в поисковиках, но и в социальных сетях. Однажды я узнал о сайте МИР-ЗАЙМОВ.РФ благодаря рекомендации в одной из групп на социальной сети. Там активно обсуждались различные МФО, и я решил попробовать одно из предложений, которое оказалось очень удобным и быстрым способом получения займа.
You are an incredible buddy! You are an impressive friend!
На портале МИР-ЗАЙМОВ.РФ вы найдете только проверенные МФК, где можно взять займ без процентов на 30 дней. Не переплачивайте лишние проценты, выбирайте оптимальное решение для своих финансов на нашем портале.
Портал МИР-ЗАЙМОВ.РФ предоставляет уникальную возможность найти лучшие МФК, где вы можете получить займ без процентов на 30 дней. Это идеальное решение для тех, кто хочет воспользоваться краткосрочным финансовым поддержкой без лишних расходов.
На МИР-ЗАЙМОВ.РФ мы убрали все лишние заботы из процесса получения займа. Теперь оформление займа в лучших МФО России – это быстро и моментально! Просто посетите наш портал, выберите подходящее предложение, заполните заявку, и ваши деньги будут у вас на счету. Не теряйте времени – действуйте с МИР-ЗАЙМОВ.РФ!
We are genuinely glad for your relationship and also commitment. You have actually been an essential part of this team and we would certainly not be where we are today without you!
Thanks! Ample advice!
play free baccarat [url=https://reddog-casinos.website/#]reddog casino[/url] chumba casino. calm
This is nicely put. !
is bc game legal in us [url=https://bcgamecasino.fun/#]bc game redeem code[/url] bc game token
The Amazingness life time deal will transform your entire point of view on exactly how to carry out a job or take the essential action.
Point effectively regarded..
bc game com [url=https://bcgamecasino.fun/#]bc game app[/url] what is bc game
I am in awe of your creative thinking and also creative imagination. I’m always impressed by your intelligence and also wisdom.
Your skill is incredible! You have such a kind as well as caring nature.
Amazing tons of useful tips.
bcgame login [url=https://bcgamecasino.fun/#]bc game coin[/url] score of bc football game
Helpful facts. Regards.
bc game bcd unlock [url=https://bc-game-casino.online/#]bc game recharge[/url] bc army game 2023
You actually expressed this exceptionally well.
bc game sweetcode [url=https://bc-game-casino.online/#]bc game crash[/url] 1000 bc game
Thanks! Plenty of knowledge.
primitive era 10000 bc game mod apk [url=https://bcgamecasino.pw/#]bc game plinko[/url] !bcgame
Factor effectively taken.!
su bc football game [url=https://bcgamecasino.pw/#]bcgame[/url] bc game shitcodes
You are so comprehending and also kind-hearted. I am so happy with all that you have completed.
Cheers, I value this.
is bc game legit [url=https://bcgamecasino.pw/#]bc game score[/url] is bc game legal in us
Your enthusiasm encourages others to do their finest. Your interest permanently is infectious!
With thanks! An abundance of material!
bc game withdrawal time [url=https://bcgamecasino.website/#]bc game com[/url] bc army game 2023
Truly loads of wonderful data.
bc game clone [url=https://bcgamecasino.website/#]bc basketball game[/url] score bc lions game
Your article was correct as well as had no typos or grammatic blunders.
Thank you! I enjoy it.
bc football bowl game 2018 [url=https://bcgamecasino.website/#]bc game no deposit bonus[/url] bc bandana game
This site is like a gold mine of valuable expertise!
Your positive perspective is inspiring! I’m impressed by your effort as well as dedication.
I am so pleased with all that you have accomplished. You are a fantastic source of motivation for me.
You’re fantastic, now your life can be too!
Wow many of beneficial advice.
скачать казино 1win [url=https://1winvhod.online/#]1win вход в личный кабинет[/url] 1win kz вход
Good facts, Regards!
1win рабочее зеркало сейчас сегодня [url=https://1winvhod.online/#]1win промокод на фриспины[/url] 1win bonus sport
You have created a genuinely remarkable source of understanding that comes to every person!
Wow, amazing blog layout! How long have you been blogging for? you make blogging glance easy. The total glance of your web site is fantastic, let alonewell as the content!
Factor effectively used!!
1win как играть [url=https://1winvhod.online/#]1win lucky jet игра отзывы[/url] ваучер 1win 2023
This information is invaluable. Where can I find out more?
You mentioned it well!
1win affiliation [url=https://1winvhod.online/#]ваучер 1win[/url] 1win lucky jet отзывы
You have such an transmittable laugh! You always locate the silver lining in every scenario.
Помню, как однажды мне срочно потребовались деньги на ремонт машины. Я решил поискать [url=https://wikizaim.ru/na-kartu-bez-otkaza/]займ онлайн срочно без отказа[/url] и обнаружил отличные предложения на сайте wikizaim.ru. Всего за несколько шагов я заполнил заявку, и к моему удивлению, получил деньги на свою карту в течение часа. Это был именно тот случай, когда быстрая финансовая поддержка была мне крайне необходима.
Truly tons of good tips!
бонусы казино в 1win [url=https://1winvhod.online/#]1win казино зеркало[/url] 1win lucky jet free cheat
Моя кредитная история была далеко не идеальной, но мне нужны были деньги. Я обратился к сайту wikizaim.ru и нашёл там [url=https://wikizaim.ru/na-kartu-bez-otkaza/]займ без проверки кредитной истории без отказа[/url]. Я был приятно удивлен, что мог получить займ, несмотря на свою историю. Это предложение стало для меня настоящим спасением, и я быстро получил нужную сумму на свою карту.
В одной далекой стране, где деньги волшебным образом появляются на картах, я открыл тайну замка wikizaim.ru. [url=https://wikizaim.ru/na-kartu-bez-otkaza/]Малоизвестные займы без отказа[/url] – словно заклинание, открывающее сундуки с золотом. Я прошел сквозь лабиринты простых форм и оказался перед сокровищем, которое помогло мне в моих королевских делах. И все это благодаря волшебству wikizaim.ru!
Ищете займ без лишних проверок и отказов? У нас на сайте wikizaim.ru вы найдете лучшие предложения по займам без отказа и без проверки кредитной истории.
Наши партнеры готовы одобрить вашу заявку на займ без лишних формальностей. Забудьте о сложных проверках и долгих ожиданиях. Просто оформите заявку онлайн, и вы получите быстрое решение.
Мы ценим ваше время и предоставляем условия займа, которые подходят для всех. Независимо от вашей кредитной истории, у нас вы найдете выгодные предложения и доступные процентные ставки.
You said it nicely..
как играть бонусами 1win [url=https://1winvhod.online/#]1win букмекерская[/url] 1win liquipedia
Kudos. Quite a lot of data.
покер 1win [url=https://1winvhod.online/#]”””1win pthrfkj”””[/url] 1win lucky jet signals
Хотите получить займ на свою кредитную карту без лишних отказов? wikizaim.ru предоставляет вам эту возможность! Мы сотрудничаем с проверенными МФО, которые готовы выдать вам средства без сложной проверки.
Оформите заявку онлайн на нашем сайте, и уже в самое короткое время вы получите одобрение и перевод средств на вашу карту. Это быстро и удобно!
Наши условия займа на карту выгодны, а процентные ставки доступны. Забудьте о долгих ожиданиях и бумажной волоките – wikizaim.ru делает процесс получения займа на кредитную карту без отказа максимально простым и быстрым.
Не упустите шанс решить свои финансовые вопросы с нами. Оформите займ на кредитную карту без отказа прямо сейчас!
Ищете надежный способ получить деньги наличными? Не хотите сталкиваться с отказами? Мы поможем вам решить финансовые вопросы с минимальными сложностями! На сайте wikizaim.ru вы найдете лучшие предложения по займам без отказа.
Наши партнеры – это проверенные МФО, которые готовы выдать вам средства даже при небольшой кредитной истории. Забудьте о бесконечных очередях и бумажной волоките. Оформите заявку онлайн, и вам не придется долго ждать ответа.
Быстрое одобрение и перевод денег на вашу карту – вот что делает нас лучшими в выборе финансовых решений. Наши условия выгодны, а процентные ставки конкурентоспособны. Получите займ без отказа уже сегодня на wikizaim.ru и решите свои финансовые вопросы быстро и без лишних хлопот!
You actually mentioned that very well!
1win sprout [url=https://1winvhod.online/#]1win казино бонус[/url] официальный сайт букмекерской конторы 1win
Fine information. Thanks a lot.
1win зеркало [url=https://1winvhod.online/#]1win зеркало рабочее[/url] 1win авиатор скачать
Онлайн займы для студентов: учеба без финансовых забот
Студенческие годы могут быть захватывающими и незабываемыми, но они также могут быть финансово напряженными. Учеба, учебники, проживание, и другие расходы могут накапливаться, и студентам часто требуется финансовая поддержка. Онлайн займы предоставляют студентам возможность покрыть свои финансовые потребности и продолжить учебу без лишних забот.
Основные преимущества онлайн займов для студентов:
Быстрое оформление. Онлайн займы можно получить в течение нескольких часов или даже минут, что особенно важно, если у вас возникла неотложная финансовая потребность.
Отсутствие сложных формальностей. В отличие от банковских кредитов, для онлайн займов не требуется предоставлять большое количество документов или иметь хорошую кредитную историю.
Гибкие условия. Множество микрофинансовых организаций предлагают гибкие условия займа, такие как разные сроки и суммы займа, что позволяет студентам выбрать наилучшие условия для своей ситуации.
Онлайн доступ. Вы можете подавать заявку и управлять своим займом онлайн, что делает процесс удобным и доступным в любое время.
Однако, прежде чем брать онлайн займ как студент, важно следовать нескольким рекомендациям:
Планируйте бюджет. Оцените свои финансовые потребности и создайте бюджет, чтобы знать, сколько денег вам действительно нужно.
Сравнивайте условия. Исследуйте разные МФО и выберите ту, которая предлагает наилучшие условия и процентные ставки.
Пользуйтесь заемными средствами ответственно. Займите только ту сумму, которую вы можете легко вернуть. Избегайте просрочек и невыполнения обязательств.
Читайте договор. Внимательно прочтите все условия займа и удостоверьтесь, что вы полностью понимаете их.
Используйте онлайн займы как краткосрочное решение. Онлайн займы идеально подходят для решения временных финансовых трудностей, но не следует использовать их как постоянный источник финансирования.
Онлайн займы для студентов могут быть полезным инструментом для обеспечения финансовой стабильности в период учебы. Однако помните о важности ответственного использования этого финансового инструмента и о том, что займы следует возвращать вовремя.
[url=https://image.google.com.mt/url?q=https://microzaim.fun/]https://image.google.com.mt/url?q=https://microzaim.fun/[/url]
[url=http://cse.google.com.gt/url?sa=i&url=https://microzaim.fun/]http://cse.google.com.gt/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://cse.google.la/url?q=https://microzaim.fun/]https://cse.google.la/url?q=https://microzaim.fun/[/url]
[url=https://maps.google.com.uy/url?q=https://microzaim.fun/]https://maps.google.com.uy/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.com.pa/url?q=https://microzaim.fun/]https://toolbarqueries.google.com.pa/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.com.vn/url?q=https://microzaim.fun/]https://toolbarqueries.google.com.vn/url?q=https://microzaim.fun/[/url]
[url=https://images.google.no/url?q=https://microzaim.fun/]https://images.google.no/url?q=https://microzaim.fun/[/url]
[url=https://maps.google.kg/url?q=https://microzaim.fun/]https://maps.google.kg/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.ge/url?q=https://microzaim.fun/]https://cse.google.ge/url?q=https://microzaim.fun/[/url]
[url=http://cse.google.co.il/url?sa=i&url=https://microzaim.fun/]http://cse.google.co.il/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.la/url?q=https://microzaim.fun/]https://toolbarqueries.google.la/url?q=https://microzaim.fun/[/url]
[url=https://images.google.com.bo/url?q=https://microzaim.fun/]https://images.google.com.bo/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.com.uy/url?q=https://microzaim.fun/]https://cse.google.com.uy/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.co.th/url?sa=i&url=https://microzaim.fun/]https://toolbarqueries.google.co.th/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://cse.google.mn/url?q=https://microzaim.fun/]https://cse.google.mn/url?q=https://microzaim.fun/[/url]
Онлайн займы: мифы и реальность
Онлайн займы стали неотъемлемой частью финансовой индустрии и предоставляют людям удобный способ получения средств в краткосрочном периоде. Однако вокруг этого финансового инструмента существует множество мифов и недоразумений. Давайте разберемся, что является мифом, а что реальностью, касательно онлайн займов.
Миф 1: Онлайн займы всегда дорогие.
Реальность: Не все онлайн займы дорогие. Процентные ставки и условия могут существенно различаться в зависимости от конкретной микрофинансовой организации (МФО). Существует множество МФО, которые предлагают конкурентоспособные условия и низкие процентные ставки.
Миф 2: Онлайн займы всегда необходимо возвращать в кратчайшие сроки.
Реальность: Сроки возврата онлайн займов могут быть гибкими и зависеть от условий договора. Вы можете выбрать срок, который наиболее удобен для вас и вашего бюджета.
Миф 3: Онлайн займы предназначены только для людей с хорошей кредитной историей.
Реальность: Многие МФО предоставляют займы и людям с плохой или отсутствующей кредитной историей. Условия могут быть менее строгими по сравнению с банками.
Миф 4: Онлайн займы всегда сопряжены с скрытыми комиссиями.
Реальность: Не все МФО взимают скрытые комиссии, и многие предоставляют прозрачную информацию о своих условиях и платежах. Важно внимательно читать договор и задавать вопросы, если что-то непонятно.
Миф 5: Онлайн займы всегда рискованны.
Реальность: Онлайн займы, когда используются ответственно, могут быть полезным инструментом для решения временных финансовых трудностей. Важно выбирать надежные МФО и соблюдать условия договора.
Онлайн займы могут быть полезным решением в различных финансовых ситуациях, но важно оценивать их на основе реальных условий и избегать попадания в ловушку мифов и стереотипов. Перед тем как воспользоваться онлайн займом, всегда стоит провести исследование и внимательно ознакомиться с условиями предоставления займа.
[url=https://toolbarqueries.google.ro/url?q=https://microzaim.fun/]https://toolbarqueries.google.ro/url?q=https://microzaim.fun/[/url]
[url=https://images.google.com.pg/url?q=https://microzaim.fun/]https://images.google.com.pg/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.as/url?sa=i&url=https://microzaim.fun/]https://toolbarqueries.google.as/url?sa=i&url=https://microzaim.fun/[/url]
[url=http://cse.google.co.ug/url?sa=i&url=https://microzaim.fun/]http://cse.google.co.ug/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://www.google.ca/url?q=https://microzaim.fun/]https://www.google.ca/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.com/url?q=https://microzaim.fun/]https://cse.google.com/url?q=https://microzaim.fun/[/url]
[url=https://images.google.com.nf/url?q=https://microzaim.fun/]https://images.google.com.nf/url?q=https://microzaim.fun/[/url]
[url=http://maps.google.dk/url?q=https://microzaim.fun/]http://maps.google.dk/url?q=https://microzaim.fun/[/url]
[url=http://www.google.co.ao/url?q=https://microzaim.fun/]http://www.google.co.ao/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.com.sv/url?q=https://microzaim.fun/]https://cse.google.com.sv/url?q=https://microzaim.fun/[/url]
[url=https://maps.google.com.do/url?q=https://microzaim.fun/]https://maps.google.com.do/url?q=https://microzaim.fun/[/url]
[url=http://toolbarqueries.google.com.pg/url?q=https://microzaim.fun/]http://toolbarqueries.google.com.pg/url?q=https://microzaim.fun/[/url]
[url=https://images.google.com.ar/url?q=https://microzaim.fun/]https://images.google.com.ar/url?q=https://microzaim.fun/[/url]
[url=https://clients1.google.dz/url?q=https://microzaim.fun/]https://clients1.google.dz/url?q=https://microzaim.fun/[/url]
[url=https://images.google.tl/url?q=https://microzaim.fun/]https://images.google.tl/url?q=https://microzaim.fun/[/url]
You actually reported it terrifically!
1win бонус за установку приложения как использовать [url=https://1winvhod.online/#]актуальное зеркало 1win[/url] 1win скачать официальное приложение на андроид бесплатно
Подробный гид по онлайн займам: от первого шага до получения средств
Когда вы решаете воспользоваться онлайн займом, важно понимать, как проходит процесс от начала до конца. Давайте рассмотрим весь путь, который вам нужно пройти, чтобы получить необходимые средства.
Все начинается с выбора надежной микрофинансовой организации (МФО). Вам необходимо исследовать рынок и выбрать МФО, которое предлагает наилучшие условия для вас. Это может включать в себя процентные ставки, сроки возврата, размеры займа и другие факторы.
После выбора МФО вам нужно будет зарегистрироваться на их веб-сайте. Обычно это требует предоставления базовой информации о себе, такой как ФИО, контактные данные и данные паспорта.
После регистрации вы можете подать заявку на займ. Это включает в себя указание желаемой суммы займа и срока его возврата. Оцените внимательно, сколько денег вам реально необходимо, и не берите лишнего, чтобы избежать переплаты.
После подачи заявки МФО обычно проводит проверку вашей кредитной истории. Но не переживайте, даже если у вас нет хорошей кредитной истории, вы можете получить займ. В случае одобрения заявки, вам будут предоставлены инструкции по дальнейшим действиям.
Далее, вы должны будете подтвердить свою личность. Это может потребовать загрузки сканов документов, таких как паспорт или водительское удостоверение. Это важный этап, чтобы обеспечить безопасность транзакции.
После подтверждения личности ваши средства будут перечислены на указанный вами банковский счет или карту. Обычно это происходит в течение нескольких минут до нескольких часов.
Теперь у вас есть доступ к средствам, и вы можете использовать их по своему усмотрению. Помните, что важно вернуть займ в срок, чтобы избежать штрафов и дополнительных расходов.
Таким образом, процесс получения онлайн займа довольно прост и удобен. Главное – следовать инструкциям МФО, быть ответственным в выборе суммы займа и срока возврата, а также внимательно читать условия займа.
[url=https://maps.google.co.ck/url?q=https://microzaim.fun/]https://maps.google.co.ck/url?q=https://microzaim.fun/[/url]
[url=http://www.google.com.au/url?q=https://microzaim.fun/]http://www.google.com.au/url?q=https://microzaim.fun/[/url]
[url=https://clients1.google.lv/url?q=https://microzaim.fun/]https://clients1.google.lv/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.mn/url?q=https://microzaim.fun/]https://cse.google.mn/url?q=https://microzaim.fun/[/url]
[url=http://maps.google.tl/url?sa=t&url=https://microzaim.fun/]http://maps.google.tl/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://maps.google.as/url?sa=t&url=https://microzaim.fun/]https://maps.google.as/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://www.google.com.mx/url?q=https://microzaim.fun/]https://www.google.com.mx/url?q=https://microzaim.fun/[/url]
[url=https://maps.google.se/url?q=https://microzaim.fun/]https://maps.google.se/url?q=https://microzaim.fun/[/url]
[url=https://images.google.se/url?q=https://microzaim.fun/]https://images.google.se/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.cf/url?sa=i&url=https://microzaim.fun/]https://cse.google.cf/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://maps.google.ws/url?sa=t&url=https://microzaim.fun/]https://maps.google.ws/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://image.google.mu/url?q=https://microzaim.fun/]https://image.google.mu/url?q=https://microzaim.fun/[/url]
[url=http://www.google.com.sv/url?sa=t&url=https://microzaim.fun/]http://www.google.com.sv/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://image.google.com.tj/url?q=https://microzaim.fun/]https://image.google.com.tj/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.je/url?q=https://microzaim.fun/]https://toolbarqueries.google.je/url?q=https://microzaim.fun/[/url]
Онлайн займы: мифы и реальность
Онлайн займы стали неотъемлемой частью финансовой индустрии и предоставляют людям удобный способ получения средств в краткосрочном периоде. Однако вокруг этого финансового инструмента существует множество мифов и недоразумений. Давайте разберемся, что является мифом, а что реальностью, касательно онлайн займов.
Миф 1: Онлайн займы всегда дорогие.
Реальность: Не все онлайн займы дорогие. Процентные ставки и условия могут существенно различаться в зависимости от конкретной микрофинансовой организации (МФО). Существует множество МФО, которые предлагают конкурентоспособные условия и низкие процентные ставки.
Миф 2: Онлайн займы всегда необходимо возвращать в кратчайшие сроки.
Реальность: Сроки возврата онлайн займов могут быть гибкими и зависеть от условий договора. Вы можете выбрать срок, который наиболее удобен для вас и вашего бюджета.
Миф 3: Онлайн займы предназначены только для людей с хорошей кредитной историей.
Реальность: Многие МФО предоставляют займы и людям с плохой или отсутствующей кредитной историей. Условия могут быть менее строгими по сравнению с банками.
Миф 4: Онлайн займы всегда сопряжены с скрытыми комиссиями.
Реальность: Не все МФО взимают скрытые комиссии, и многие предоставляют прозрачную информацию о своих условиях и платежах. Важно внимательно читать договор и задавать вопросы, если что-то непонятно.
Миф 5: Онлайн займы всегда рискованны.
Реальность: Онлайн займы, когда используются ответственно, могут быть полезным инструментом для решения временных финансовых трудностей. Важно выбирать надежные МФО и соблюдать условия договора.
Онлайн займы могут быть полезным решением в различных финансовых ситуациях, но важно оценивать их на основе реальных условий и избегать попадания в ловушку мифов и стереотипов. Перед тем как воспользоваться онлайн займом, всегда стоит провести исследование и внимательно ознакомиться с условиями предоставления займа.
[url=http://www.google.sm/url?sa=t&url=https://microzaim.fun/]http://www.google.sm/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://images.google.by/url?sa=t&url=https://microzaim.fun/]https://images.google.by/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://images.google.com.kw/url?q=https://microzaim.fun/]https://images.google.com.kw/url?q=https://microzaim.fun/[/url]
[url=http://www.google.tn/url?sa=t&url=https://microzaim.fun/]http://www.google.tn/url?sa=t&url=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.com.sl/url?q=https://microzaim.fun/]https://toolbarqueries.google.com.sl/url?q=https://microzaim.fun/[/url]
[url=https://images.google.dk/url?q=https://microzaim.fun/]https://images.google.dk/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.com.bn/url?q=https://microzaim.fun/]https://toolbarqueries.google.com.bn/url?q=https://microzaim.fun/[/url]
[url=http://clients1.google.com.ly/url?q=https://microzaim.fun/]http://clients1.google.com.ly/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.az/url?q=https://microzaim.fun/]https://toolbarqueries.google.az/url?q=https://microzaim.fun/[/url]
[url=https://clients1.google.com.af/url?sa=i&url=https://microzaim.fun/]https://clients1.google.com.af/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://images.google.ki/url?q=https://microzaim.fun/]https://images.google.ki/url?q=https://microzaim.fun/[/url]
[url=https://clients4.google.com/url?q=https://microzaim.fun/]https://clients4.google.com/url?q=https://microzaim.fun/[/url]
[url=https://clients1.google.pt/url?sa=i&url=https://microzaim.fun/]https://clients1.google.pt/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://images.google.co.bw/url?sa=t&url=https://microzaim.fun/]https://images.google.co.bw/url?sa=t&url=https://microzaim.fun/[/url]
[url=http://maps.google.lu/url?q=https://microzaim.fun/]http://maps.google.lu/url?q=https://microzaim.fun/[/url]
Nicely put, Regards.
1win букмекерская контора вход [url=https://1winvhod.online/#]1win bonus casino[/url] 1win скачать на андроид последняя версия
Сухой мухомор купить на pridary.ru
Наша компания находится в Нижегородской области и имеет собственное хозяйство по распространению экологически чистых продуктов. «Дары природы» представляет красные мухоморы, белый гриб, различные травы и чаи, а также, на интернет портале pridary.ru Вы сможете найти подробное описание растений и целебных их качеств для вашего здоровья.
По вопросу [url=https://pridary.ru/product/muxomor-panternyj-sushenyj/]мухомор гриб цена[/url] заходите на данный интернет сайт. Мы уже большое количество лет осуществляем работу в представленной области и знаем многое о здоровье человека и как его поправить, при необходимости. Употребляя нашу продукцию, можно отказаться от неких аптечных препаратов, заместив их нашими природными. Мы pridary.ru лично заготавливаем, храним и упаковываем продукцию, что дает прекрасную гарантию качества. Вдобавок у нас множество положительных отзывов от кучи людей, которые давно уже пользуются нашими товарами.
Нижегородская область славится своим мягким климатом, чистыми хорошими лесами, огромными лугами и голубыми реками. Здесь и обитает наша продукция, которая может помочь людям возвратить здоровье и зажить счастливой жизнью. В каталоге представлены грибы, мед, варенье и ягоды, которые мы доставляем по всей стране. Доставка осуществляется почтой РФ или тк СДЕК.
У нас на сайте стоит почитать важные статьи о мухоморах, а также их полезные свойства на организм. Поразительно то, что мухомор является смертельно опасным грибом, в зависимости от дозировки. В маленьких дозах он целебен, а в больших приводит к смертельному исходу.
Насчет [url=https://pridary.ru/product/muxomor-krasnyj-sushenyj-shlyapki-1-sort-50-g/]сухой мухомор[/url] мы Вам окажем помощь. Свяжитесь с нами по телефону +7(930)672-59-00 или пишите на электронную почту. Мы 24 на 7 на связи, окажем помощь по любому вопросу и подбору продукта конкретно Вам, но в сезон собирания связь не всегда может быть стабильна. Поэтому можно воспользоваться таблицей на сайте pridary.ru для отправки сообщения. Звоните, пишите и заказывайте уникальные чистые продукты.
Заказать мухоморы на pridary.ru
Представленная организация находится в Нижегородской области и имеет собственное производство по выращиванию экологически чистых продуктов. «Дары природы» представляет пантерные мухоморы, белый гриб, различные травы и чаи, а ещё, на интернет портале pridary.ru Вы сможете найти обширное описание растений и целебных их свойств для вашего здоровья.
По запросу [url=https://pridary.ru/product-category/griby/]купить грибы мухоморы[/url] заходите на данный веб сайт. Мы уже большое количество лет работаем в данной области и знаем очень много о здоровье человека и как его поправить, при потребности. Приобретая нашу продукцию, можно отказаться от некоторых аптечных препаратов, заменив их нашими природными. Мы pridary.ru лично заготавливаем, сохраняем и упаковываем продукцию, что дает отличную гарантию качества. А также у нас много положительных отзывов от кучи людей, которые давно уже пользуются нашими продуктами.
Нижегородская область пользуется известностью своим мягким климатом, чистыми хорошими лесами, просторными лугами и голубыми реками. Здесь и собирается наша продукция, которая может помочь людям возвратить здоровье и зажить счастливой жизнью. В каталоге представлены грибы, мед, варенье и ягоды, которые мы отправляем по всей стране. Доставка происходит почтой России или тк СДЕК.
У нас на портале можно почитать интересные статьи о мухоморах, а также их полезные свойства на человеческий организм. Странно то, что мухомор является смертельно ядовитым грибом, в зависимости от дозировки. В маленьких дозах он целебен, а в больших приводит к летальному результату.
Относительно [url=https://pridary.ru/product-category/griby/]купить грибы мухоморы[/url] мы Вам окажем помощь. Позвоните по контактному телефону +7(930)672-59-00 или пишите на электронную почту. Мы всегда на связи, поможем по любому вопросу и подбору продукта именно Вам, но в сезон сбора связь не всегда может быть стабильна. Поэтому стоит воспользоваться таблицей на сайте pridary.ru для отправки вопросов. Звоните, пишите и приобретайте разнообразные чистые продукты.
Обновление моего ОСАГО на сайте osagoonline.ru было настоящим открытием. Использовав [url=https://osagoonline.ru/]оформить осаго на автомобиль онлайн[/url], я сэкономил уйму времени. Весь процесс прошёл гладко и без лишних забот, что сделало мой опыт с osagoonline.ru исключительно положительным.
Oh my goodness! Incredible article dude! Thank you, However I am experiencing difficulties with your RSS. I don’t know why I am unable to subscribe to it. Is there anybody else getting identical RSS problems? Anybody who knows the solution will you kindly respond? Thanx!!
Wonderful data, With thanks.
1win букмекерская скачать на андроид [url=https://1winvhod.online/#]1win зеркало на сегодня скачать[/url] как играть бонусами 1win
I don’t know if it’s just me or if everyone else experiencing problems with your blog. It seems like some of the text on your posts are running off the screen. Can someone else please comment and let me know if this is happening to them too? This might be a problem with my browser because I’ve had this happen before. Appreciate it
Недавно мне пришлось столкнуться с неожиданной проблемой: моя машина сломалась в самый неподходящий момент. Чтобы оплатить ремонт, я решил воспользоваться услугой займа на карту, который работает круглосуточно и обещает мгновенное поступление средств без отказа. К моему удивлению, уже через несколько минут после заполнения заявки я получил нужную сумму. Благодаря этому я смог сразу же отремонтировать машину и продолжить свои дела без лишних задержек.
[url=http://www.google.ws/url?sa=t&url=https://microzaim.fun/]http://www.google.ws/url?sa=t&url=https://microzaim.fun/[/url]
[url=http://alt1.toolbarqueries.google.com.do/url?q=https://microzaim.fun/]http://alt1.toolbarqueries.google.com.do/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.nl/url?q=https://microzaim.fun/]https://cse.google.nl/url?q=https://microzaim.fun/[/url]
[url=https://image.google.me/url?q=https://microzaim.fun/]https://image.google.me/url?q=https://microzaim.fun/[/url]
[url=https://image.google.co.bw/url?q=https://microzaim.fun/]https://image.google.co.bw/url?q=https://microzaim.fun/[/url]
Помню, как искал мухоморы на Avito, но наткнулся на pridary.ru – и это было настоящее открытие. Здесь я нашел уникальные предложения, которых не встретишь на обычных площадках. Качество товара поразило: свежие, ароматные и идеально сушеные мухоморы. Если вы также в поиске необычных товаров, обязательно посетите [url=https://pridary.ru/product/muxomor-krasnyj-sushenyj-shlyapki-1-sort-50-g/]мухоморы авито[/url] на pridary.ru.
Однажды, готовясь к участию в необычном квесте по городским легендам, мне срочно понадобились дополнительные средства для покупки реквизита. Вопрос “где срочно взять деньги без отказа?” нашел свой ответ в удобном онлайн-сервисе. Это было удивительно легко и быстро, и я смог полностью погрузиться в атмосферу приключений, не беспокоясь о финансах.
[url=http://cse.google.bg/url?sa=t&url=https://microzaim.fun/]http://cse.google.bg/url?sa=t&url=https://microzaim.fun/[/url]
[url=http://clients1.google.gm/url?q=https://microzaim.fun/]http://clients1.google.gm/url?q=https://microzaim.fun/[/url]
[url=https://image.google.am/url?q=https://microzaim.fun/]https://image.google.am/url?q=https://microzaim.fun/[/url]
[url=https://toolbarqueries.google.com.ni/url?sa=i&url=https://microzaim.fun/]https://toolbarqueries.google.com.ni/url?sa=i&url=https://microzaim.fun/[/url]
[url=https://www.google.cg/url?q=https://microzaim.fun/]https://www.google.cg/url?q=https://microzaim.fun/[/url]
В преддверии своего первого большого фотовыставки, я столкнулся с необходимостью срочной покупки дополнительного оборудования. Поскольку у меня были опасения относительно проверки кредитной истории, я выбрал сервис, предлагающий деньги на карту без отказов и без проверки кредитной истории. Это решение позволило мне успешно организовать выставку, которая стала знаковым событием в моей карьере фотографа.
[url=https://maps.google.com.ai/url?q=https://microzaim.fun/]https://maps.google.com.ai/url?q=https://microzaim.fun/[/url]
[url=https://www.google.vg/url?q=https://microzaim.fun/]https://www.google.vg/url?q=https://microzaim.fun/[/url]
[url=https://maps.google.cv/url?q=https://microzaim.fun/]https://maps.google.cv/url?q=https://microzaim.fun/[/url]
[url=http://cse.google.at/url?q=https://microzaim.fun/]http://cse.google.at/url?q=https://microzaim.fun/[/url]
[url=https://cse.google.mg/url?q=https://microzaim.fun/]https://cse.google.mg/url?q=https://microzaim.fun/[/url]
Когда я спросила в одной из социальных сетей о хороших магазинах с интимными товарами, многие посоветовали sladkiye-strasti.ru. После посещения сайта я нашла замечательный выбор [url=https://sladkiye-strasti.ru/]секс шоп купить[/url] , подходящих для экспериментов и новых ощущений. Отзывы других покупателей помогли мне сделать выбор, а процесс покупки и доставки оказался максимально удобным и дискретным.
На форуме в социальной сети мне посоветовали sladkiye-strasti.ru как идеальный магазин для покупки интимных товаров. Я была впечатлена ассортиментом [url=https://sladkiye-strasti.ru/]секс шоп онлайн[/url] , который предлагал сайт. Отзывы пользователей были положительными, что убедило меня в выборе. Простота оформления заказа и конфиденциальность доставки добавили уверенности в моем решении.
Amazing a lot of useful data.
1win скачать официальное приложение на андроид [url=https://1winoficialnyj.online/#]1win betting[/url] 1win web-site
Good answers in return of this difficulty with firm arguments and describing everything about that.
You said it nicely.!
1win букмекерская контора зеркало [url=https://1winoficialnyj.online/#]игра 1win[/url] что делать с бонусами на 1win
На форуме в социальной сети мне посоветовали sladkiye-strasti.ru как идеальный магазин для покупки интимных товаров. Я была впечатлена ассортиментом [url=https://sladkiye-strasti.ru/]эротический интернет магазин[/url] , который предлагал сайт. Отзывы пользователей были положительными, что убедило меня в выборе. Простота оформления заказа и конфиденциальность доставки добавили уверенности в моем решении.
2023 год открывает новые возможности для получения займов! На [url=https://wikizaim.ru/]wikizaim.ru[/url] представлены новые МФО с самыми выгодными и привлекательными условиями. Забудьте о страхе отказа – здесь каждый найдет своего надежного финансового партнера. Воспользуйтесь этим шансом, чтобы решить свои финансовые задачи быстро и без забот.
Понимаем, как важно иногда получить финансовую помощь, и на [url=https://wikizaim.ru/]wikizaim.ru[/url] вы найдете займы без отказа, которые действительно могут стать вашей поддержкой в сложные моменты. Здесь вы можете быть уверены, что ваша заявка будет рассмотрена с пониманием и уважением к вашим обстоятельствам.
You stated that effectively.
1win скачать бесплатно [url=https://1winoficialnyj.site/#]1win games[/url] бонус по free spins 1win
Время – это деньги, и на [url=https://wikizaim.ru/]wikizaim.ru[/url] это прекрасно понимают. Здесь вы можете получить онлайн займы без отказа и длительных проверок. Процесс оформления максимально упрощен и занимает всего несколько минут. Выберите подходящее предложение, оформите заявку, и деньги будут у вас на карте в кратчайшие сроки. Не откладывайте свои планы – получите финансовую поддержку прямо сейчас!
Ну что, нужны деньги, как в сказке, мгновенно и без всяких “но”? Представляю вашему вниманию [url=https://wikizaim.ru/]wikizaim.ru[/url] – место, где финансовые чудеса случаются чаще, чем дни рождения. Займы без отказа и проверок – это как волшебная палочка для вашего кошелька. Просто помахайте кредитной историей, и… ой, не надо, она тут не нужна!
Восстановление помещения — наша специализация. Исполнение ремонтных услуг в сфере жилья. Мы предлагаем реставрацию дома с гарантированным качеством.
[url=https://remont-kvartir-brovari.kyiv.ua/]remont-kvartir-brovari.kyiv.ua[/url]
Appreciate it. Plenty of postings!
1win invest отзывы [url=https://1winoficialnyj.site/#]скачать 1win официальный сайт[/url] 1win registration
Проект домашнего кинотеатра audiopik.ru
Планируете заказать личный кинозал у Вас дома? В таком случае Вы попали четко по адресу! Наша компания АудиоПик специализируется на разработке лучшего домашнего кинозала в квартирах, домах, офисах и разных иных помещениях. Работаем по всей столице и Московской области, предоставляем гарантию до пяти лет и техническую поддержку сроком в один год. Это коротко о представленной компании, подробнее узнайте на сайте audiopik.ru уже сейчас.
Если Вы искали [url=https://audiopik.ru/domashnij-kinoteatr-v-zagorodnom-dome/]домашний кинотеатр в доме[/url] в сети интернет, то Вы на верном пути. В нашей команде работают только квалифицированные специалисты, которые знают толк в деле. Ежегодно обучаются новым инновационным программам тв системам и готовы предложить Вам самый лучший вариант из допустимых, ссылаясь на ваши предпочтения и бюджет. У нас уже реализовано в жизнь более сотни заказов, отзывы и фото которых можно посмотреть на сайте audiopik.ru в настоящее время.
Полный список наших услуг: проектирование домашних кинотеатров, замена публичного кинотеатра, послегарантийное обслуживание, системы Hi-Fi, лучшая усилительная техника, моделирование комнат прослушивания, программирование систем управления воспроизведением и многие другие.
Домашний кинотеатр — это прекрасная идея для любого вида семьи. Будь Вы домохозяйка, или семьянин с тремя детьми, а может быть пенсионер, это не важно. Смотреть интересный фильм на широком качественном экране понравится любому. А наша супер инновационная акустика — это определенный вид удовольствия для Ваших ушей. Мы audiopik.ru подберем для вас безупречный вариант кинозала. Попробуйте и удостоверьтесь сами!
Что касается [url=https://audiopik.ru/domashnij-kinoteatr-4k/]домашний кинотеатр в квартире[/url] позвоните нам. Наш телефон для связи +7(495)127-01-46 или напишите на мессенджеры WhatsApp или Viber. Мы расположены по адресу: Мос. Обл., г. Щелково, ул. Центральная, стр. 17. Звоните, приезжайте и заказывайте Ваш идеальный домашний кинотеатр.
Проект домашнего кинотеатра audiopik.ru
Хотите иметь личный кинозал у Вас дома? В таком случае Вы попали точно по адресу! Данная компания АудиоПик концентрируется на разработке лучшего домашнего кинотеатра в квартирах, коттеджах, офисах и разных иных помещениях. Работаем по всей столице и Московской области, даем гарантию до пяти лет и тех поддержку сроком в 1 год. Это сокращенно о представленной компании, обстоятельства узнайте на интернет портале audiopik.ru прямо сегодня.
Если Вы хотели найти [url=https://audiopik.ru/domashnij-kinoteatr-4k/]домашний кинотеатр в квартире[/url] в сети интернет, то Вы на правильном пути. В нашей компании работают только квалифицированные специалисты, которые знают толк в деле. Постоянно обучаются новейшим инновационным программам тв системам и рады представить Вам самый лучший вариант из допустимых, ссылаясь на ваши желания и финансы. У нас уже реализовано в жизнь более 100 кинотеатров, отзывы и фото которых можно посмотреть на сайте audiopik.ru в данный момент.
Полный список наших услуг: проектирование домашних кинотеатров, акустические расчеты, послегарантийное обслуживание, системы Hi-Fi, цифровые проигрыватели, комнаты прослушивания, программирование систем управления воспроизведением и многие другие.
Домашний кинотеатр — это отличная идея для любого типа семьи. Будь Вы студент, или семьянин с тремя детьми, а может быть пенсионер, это не так значимо. Смотреть интересный сериал на широком качественном экране будет нравиться любому. А наша супер инновационная акустика — это определенный тип удовольствия для Ваших ушей. Мы audiopik.ru подберем для вас лучший вариант кинотеатра. Попробуйте и удостоверьтесь сами!
Касательно [url=https://audiopik.ru/uslugi/domashnij-kinoteatr-pod-klyuch/]установка домашнего кинотеатра[/url] позвоните нам. Наш контактный телефон для связи +7(495)127-01-46 или пишите на мессенджеры WhatsApp или Viber. Мы находимся по адресу: Мос. Обл., г. Щелково, ул. Центральная, стр. 17. Звоните, приезжайте и забирайте Ваш идеальный домашний кинотеатр.
Kudos! Fantastic information!
1win фрибет [url=https://1winoficialnyj.site/#]1win зеркало сайта[/url] бонусы на 1win
Nicely put. Thanks a lot!
1win скачать официальное приложение на андроид [url=https://1winoficialnyj.website/#]1win скачать мобильное приложение[/url] 1win бесплатно андроид
Проект домашнего кинотеатра audiopik.ru
Желаете купить собственный кинотеатр у Вас дома? В таком случае Вы пришли точно по адресу! Наша фирма АудиоПик специализируется на создании лучшего домашнего кинотеатра в квартирах, домах, офисах и любых других помещениях. Осуществляем работу по всей столице и Московской области, предоставляем гарантию до 5-ти лет и тех поддержку сроком в 1 год. Это сокращенно о нашей фирме, детальнее узнайте на сайте audiopik.ru уже сегодня.
Если Вы хотели найти [url=https://audiopik.ru/uslugi/domashnij-kinoteatr-pod-klyuch/]подключение домашнего кинотеатра[/url] в интернете, то Вы на верном пути. В нашей компании работают высококвалифицированные специалисты, которые понимают толк в работе. Ежегодно обучаются новейшим инновационным тв системам и рады доставить Вам лучший вариант из возможных, опираясь на ваши предпочтения и бюджет. У нас уже реализовано в жизнь более 100 кинотеатров, отзывы и фотографии которых можно посмотреть на сайте audiopik.ru сейчас.
Подробный перечень наших услуг: установка систем домашних кинотеатров, поставка и настройка техники, гарантийное обслуживание, системы High-End класса, виниловые вертушки, акустическая коррекция помещений, создание многозонных аудио комплексов и многие другие.
Домашний кинозал — это прекрасная идея для любого вида семьи. Будь Вы домохозяйка, или семьянин с тремя детьми, или даже пенсионер, это не столь значительно. Смотреть захватывающий сериал на широком современном экране понравится любому. А наша супер инновационная акустика — это отдельный тип наслаждения для Ваших ушей. Мы audiopik.ru подберем для вас самый лучший вариант кинотеатра. Попробуйте и удостоверьтесь сами!
По поводу [url=https://audiopik.ru/domashnij-kinoteatr-4k/]домашний кинотеатр в квартире[/url] звоните нам. Наш контактный телефон для связи +7(495)127-01-46 или пишите на мессенджеры вотсап или вайбер. Мы расположены по адресу: Мос. Обл., г. Щелково, ул. Центральная, стр. 17. Звоните, приходите и забирайте Ваш идеальный домашний кинозал.
Truly a good deal of very good information!
как использовать бонусы 1win казино [url=https://1winoficialnyj.website/#]промокод 1win при регистрации[/url] jet в 1win как обыграть лаки лаки отзывы казино лаки
Уважаемый заемщик! С появлением новых МФО в 2023 году, выбор становится все шире. На [url=https://vc.ru/u/2632054-skilroom/938020-zaymy-2023-na-kartu-novye-i-maloizvestnye-mfo-po-vydache-bystryh-zaymov]vc.ru[/url] ты найдешь множество выгодных предложений. Однако необходимо быть бдительным: проверь лицензии и репутацию МФО, читай отзывы других клиентов и не доверяй слишком привлекательным условиям без тщательной проверки. Будь разумным и осторожным в своих финансовых решениях.
Здравствуйте! В 2023 году рынок представил нам множество новых онлайн МФО. Чтобы сделать правильный выбор, рекомендую вам посетить [url=https://vc.ru/u/2632054-skilroom/938020-zaymy-2023-na-kartu-novye-i-maloizvestnye-mfo-po-vydache-bystryh-zaymov]vc.ru[/url], где собрана актуальная информация о каждой компании. Обращайте внимание на условия займов, процентные ставки и отзывы клиентов. Тщательно взвешивайте все “за” и “против” перед тем, как принять решение о займе. Ваш финансовый благополучие должно быть приоритетом.
Привет! Знаешь, в наше время, когда каждый момент может быть запечатлен и поделен с миром, [url=http://antiban.pro/ru/blog/162]смотреть инстаграм посты[/url] – это не просто способ убить время. Это увлекательное путешествие по миру интересов, хобби и идей. Здесь ты можешь найти вдохновение, подсказки для творчества, кулинарные рецепты, модные тренды и многое другое. Каждый пост – это маленькая история, которая может чему-то научить или просто поднять настроение.
Antiban.pro мы поможем решить проблемы:
1 – Заблокировали аккаунт
2 – Заблокировали действия в аккаунте
3 – Проблемы с привязкой Facebook
4 – Меня взломали в Instagram
5 – Меня взломали в Facebook
В современном мире социальных сетей понятие [url=http://antiban.pro/ru/blog/6]теневой бан это[/url] означает невидимое ограничение доступности контента пользователя для других. Теневой бан может быть применен социальными платформами в случае нарушения их правил или политик. Это серьезное предупреждение для любого пользователя, требующее внимательного пересмотра и корректировки стратегии ведения аккаунта. Соблюдение правил платформы и создание качественного контента – лучшая стратегия для избегания теневого бана.
Antiban.pro мы поможем решить проблемы:
1 – Заблокировали аккаунт
2 – Заблокировали действия в аккаунте
3 – Проблемы с привязкой Facebook
4 – Меня взломали в Instagram
5 – Меня взломали в Facebook
Many thanks. Plenty of postings.
как играть на бонусы казино в 1win [url=https://1winoficialnyj.website/#]ваучер 1win[/url] 1win официальный вход
В 2023 году рынок микрофинансирования демонстрирует значительный рост с появлением новых МФО и МКК. Эти учреждения вносят свой вклад в диверсификацию финансовых услуг, предлагая различные виды займов и кредитов. Аналитики отмечают, что расширение спектра предложений может способствовать повышению финансовой доступности для широких слоев населения. Более подробную информацию об этих новых финансовых учреждениях можно найти на [url=https://vc.ru/u/2632054-skilroom/938020-zaymy-2023-na-kartu-novye-i-maloizvestnye-mfo-po-vydache-bystryh-zaymov]vc.ru[/url]
Дорогой друг! С радостью делюсь новостью о появлении новых МФО в 2023 году, которые предлагают займы без отказов. Посети [url=https://vc.ru/u/2632054-skilroom/938020-zaymy-2023-na-kartu-novye-i-maloizvestnye-mfo-po-vydache-bystryh-zaymov]vc.ru[/url], чтобы узнать подробности. Это отличная возможность для тех, кто сталкивался с финансовыми трудностями в прошлом. Не упусти этот шанс облегчить свое финансовое бремя.
You make others really feel approved and appreciated. You have such an infectious laugh!
Tips effectively used..
скачать 1win на андроид бесплатно с официального [url=https://1winregistracija.online/#]1win cs go[/url] 1win скачать на айфон с официального сайта
For those that are experienced in producing fantastic web content on their own!
Kudos! Lots of information.
1win ваучер на деньги [url=https://1winregistracija.online/#]1win vs sprout[/url] 1win вывод средств
Hello, I log on to your blogs like every week. Your story-telling style is awesome, keep doing what you’re doing!
С каждым днем Инстаграм становится все более популярным, и [url=http://antiban.pro/ru/blog/142]сайт инстаграм вход[/url] является ключом к миру бесконечного творчества и общения. Здесь каждый может найти что-то свое, будь то фотография, искусство, мода или кулинария. Инстаграм дает возможность не только наблюдать за жизнью других, но и самим стать частью большого и дружного сообщества, где каждый голос имеет значение.
Antiban.pro мы поможем решить проблемы:
1 – Заблокировали аккаунт
2 – Заблокировали действия в аккаунте
3 – Проблемы с привязкой Facebook
4 – Меня взломали в Instagram
5 – Меня взломали в Facebook
Nicely put. Appreciate it!
бесплатный ваучер 1win [url=https://1winregistracija.online/#]1win скачать на андроид[/url] en el en 1win 1win cdigos promocionales 1win juegos
Amazing tons of awesome information.
1win com официальный сайт [url=https://1winvhod.online/#]1win бонусы спорт как использовать[/url] сколько длится вывод с 1win
Hi there, I wish for to subscribe for this website to get most up-to-date updates, thus where can i do it please assist.
Really tons of fantastic information.
ваучер 1win [url=https://1winvhod.online/#]1win bet[/url] 1win онлайн
[url=https://samye-luchshie-prostitutki-moskvy.top]https://samye-luchshie-prostitutki-moskvy.top[/url]
Wow loads of amazing information!
как потратить бонусный счет 1win [url=https://1winvhod.online/#]1win слоты и 1вин казино 1win казино[/url] 1вин реальный обзор 1win зеркало 1вин
I have been surfing online more than three hours nowadays, yet I never found any fascinating article like yours. It’s lovely worth enough for me. Personally, if all webmasters and bloggers made good content as you did, the net might be much more useful than ever before.
Good information. Regards.
1xbet login [url=https://zerkalo-1xbet.website/#]слоты с минимальной ставкой в 1xbet[/url] 1xbet pthrfkj
Excellent forum posts. With thanks.
промокод на бонус 1xbet [url=https://zerkalo-1xbet.website/#]1xbet официальный сайт вход[/url] 1xbet скачать бесплатно 2023
Truly a good deal of useful data!
1xbet зеркало рабочее мобильная скачать для андроид [url=https://zerkalo-1xbet.website/#]1xbet скачать[/url] как пополнить 1xbet на 100 рублей
Accomplish extra and also do it much better with Amazingness!
Amazingness can aid you become extra productive and get even more out of life!
Пассивный сабвуфер купить на dbshop.ru
Приглашаем Вас в лучший онлайн магазин автоакустики DBShop, где выложен обширный список ингредиентов для самого чистого звука конкретно в Ваш авто. Мы проводим работу в представленной сфере уже множество лет и имеем рабочие отношения с мировыми фирмами напрямую, например: EDGE, DL Audio, Ural Sound, PRIDE и другие. Поэтому стоимость у нас очень соблазнительная и не имеет больших накруток.
По запросу [url=https://dbshop.ru/product/korob_dlya_sabvufera_12_fi_shchel/]короб на щели 15[/url] заходите на наш онлайн портал. На dbshop.ru есть вся обширная информация о нашей организации. Мастера, которые знают смысл в своей работе, ориентированы на то, чтобы найти в Ваш автомобиль идеальный сабвуфер. Учитывая пожелания и бюджет клиента. Продукцию, которую Вы найдете в нашем каталоге: автомагнитолы 2DIN, громкая акустика, сабвуферы, кабель, аксессуары и многое другое.
Здесь самые привлекательные цены, которые не оказывают влияния на потерю качества вовсе. А всё потому, что мы проводи работу напрямую от фирм поставщиков. Множество продукции есть в наличии, но ещё можно выбрать товары под заказ, если конкретной модели сейчас не имеется. Доставляем всегда в обговоренный срок, а постоянным клиентам предлагаем интересную систему скидок. Все товары, приобретенные у нас имеют все необходимые сертификаты и гарантию. Доставка осуществляется в любую область России, а при оформлении заказа от 9900 рублей доставка осуществляется бесплатно.
Если Вы планировали найти [url=https://dbshop.ru/catalog/gromkaya_akustika/zashchitnye_setki/]защитные грили[/url] в интернете, то Вы на правильном пути. Приобрести товары на сайте dbshop.ru очень просто. Следует кидать в корзину требующиеся Вам товары, а следом оформить заказ. Укажите Ваше ФИО, контактный телефон, электронный адрес, способы оплаты и доставки, и ожидайте заказ. Если возникли проблемы, звоните по контактному телефону, который есть далее.
Мы расположены по адресу: г. Ижевск, ул. Коммунаров, 244. Время работы с пн по пт с 9:00 до 19:00, в сб и вс с 10:00 до 18:00. Наш номер контактного телефона 8(800)550-99-69 звоните и мы с удовольствием Вам окажем помощь.
Онлайн магазин автоакустики dbshop.ru
Зовём Вас в лучший интернет магазин автозвука DBShop, где представлен огромный перечень составных частей для самого чистого звука конкретно в Ваш авто. Мы проводим работу в представленной теме уже много лет и имеем контакты с известными фирмами напрямую, к примеру: EDGE, Dynamic State, Ural Sound, PRIDE и другие. Поэтому стоимость у нас весьма привлекательная и не имеет высоких наценок.
По теме [url=https://dbshop.ru/catalog/avtomagnitoly/shtatnye_golovnye_ustroystva/]магнитола на андроиде купить[/url] переходите на указанный онлайн сайт. На dbshop.ru есть вся обширная информация о нашей компании. Мастера, которые знают смысл в собственной работе, нацелены на то, чтобы найти в Ваш автомобиль идеальный сабвуфер. Принимая к сведению пожелания и бюджет заказчика. Товары, которые Вы увидите в нашем каталоге: автомагнитолы 1DIN, Hi-Fi, короба, кабель, аксессуары и многое другое.
Здесь самые привлекательные цены, которые не оказывают влияния на потерю качества совсем. А всё оттого, что мы проводи работу напрямую от фирм поставщиков. Большинство продукции есть в наличии, но также возможно выбрать товары под заказ, если конкретной модели сегодня не имеется. Отправляем всегда в обговоренный срок, а постоянным заказчикам предоставляем гибкую систему скидок. Все товары, приобретенные у нас имеют сертификаты и гарантийный срок. Доставка осуществляется в любую область России, а при заказе от 9900 рублей доставка осуществляется бесплатно.
В случае если Вы планировали найти [url=https://dbshop.ru/product/sabvufer_ural_symphony_10/]сабвуфер пассивный 10[/url] в интернете, то Вы на верном пути. Заказать на веб ресурсе dbshop.ru весьма просто. Стоит кидать в корзину необходимые Вам товары, а далее оформить заказ. Укажите Ваше ФИО, контактный телефон, Email, способы оплаты и доставки, и ждите свой заказ. Если появились проблемы, звоните по контактному телефону, который написан далее.
Мы расположены по адресу: г. Ижевск, ул. Коммунаров, 244. График работы с пн по пт с 9:00 до 19:00, в субботу и воскресенье с 10:00 до 18:00. Наш номер контактного телефона 8(800)550-99-69 звоните и мы будем рады с Вами сотрудничать.
Your interest for the job was contagious and your dedication was an ideas to us all. We can not thank you sufficient for all that you did!
В тот день, когда мне понадобились деньги на неотложные нужды, я столкнулся с непреодолимой стеной банковских отказов. Но тогда, словно маяк в темной ночи, я обнаружил сервис онлайн-займов. Скептически относясь к возможности быстрого решения, я заполнил заявку. К моему изумлению, одобрение пришло мгновенно, как волшебство. Вот так, в один прекрасный момент, мои финансовые заботы рассеялись, как дым.
На нашем портале вы найдете [url=https://dzen.ru/a/ZWzCnKzreD2pFSOB]займ на любую карту без проверки[/url] и [url=https://dzen.ru/a/ZWzCnKzreD2pFSOB]первый займ процентов на карту[/url] которые доступны для всех от 18 лет!
Awesome blog! Do you have any recommendations for aspiring writers? I’m planning to start my own site soon but I’m a little lost on everything. Would you propose starting with a free platform like WordPress or go for a paid option? There are so many choices out there that I’m totally confused .. Any recommendations? Bless you!
MOTOLADY предлагают услуги аренды и проката мотоциклов и скутеров в Хургаде, Эль Гуне и Сахл Хашиш. MOTOLADY – одна из самых популярных компаний по прокату мотоциклов и скутеров. Они предлагают большой выбор транспортных средств по разумным ценам. MOTOLADY компания, специализирующаяся на [url=https://t.me/detivetrachat]Аренда скутера в Хургаде[/url] и Эль Гуне. Они предлагают услуги доставки транспорта в любое удобное для вас место. У нас в наличии различные модели транспортных средств по доступным ценам. Перед арендой транспорта обязательно ознакомьтесь с правилами и требованиями компании, также проверьте наличие страховки и необходимые документы для аренды.
You gave readers with the necessary understanding and also realities in order to make their very own enlightened choices.
We are so grateful you belonged to the task and also we can not thanks enough for all that you’ve done. Your hard work and dedication have been excellent!
I can constantly trust you to be there for me. You have such a kind as well as caring nature.
Один из самых удобных способов решения финансовых проблем – это [url=https://credit-world.ru/]микрозаймы без отказов на карту[/url]. Такой подход позволяет быстро и без лишних бумажных процедур получить необходимую сумму, что особенно актуально в ситуациях, когда средства нужны срочно.
При анализе [url=https://credit-world.ru/]займов на карту без отказов[/url], следует отметить, что большое количество предложений от разных МФО увеличивает конкуренцию на рынке, что часто приводит к более выгодным условиям для заемщиков. Однако, важно осознавать риски и тщательно изучать условия каждого предложения.
You handle tight spots with grace and also poise. Your enthusiasm is infectious!
В лабиринте повседневных расходов, [url=https://credit-world.ru/]займ на карту без отказов[/url] становится ключом к скрытому сокровищу. Этот займ подобен факелу в темноте, освещая путь к финансовой свободе, и предоставляя немедленное решение, когда каждая секунда на счету.
Great web site you’ve got here.. It’s hard to find quality writing like yours these days. I seriously appreciate people like you! Take care!!
I am so appreciative for your endless assistance as well as love. You are a wonderful incentive and leader.
Start living your best life now with the incredible!
Внимание, срочно в эфире! Мы нашли способ, как [url=https://microzaim.fun/]займ на любую карту мгновенно[/url] может изменить вашу жизнь! Это не шутка, и не очередной фокус – это реальность. Будь то средство для решения ваших срочных нужд или спасательный круг в море неожиданных расходов, мы здесь, чтобы помочь. И пусть ваша финансовая история будет наполнена только хорошими новостями!
Ищете быстрый займ без процентов? Откройте ворота в мир финансовой свободы с [url=https://microzaim.fun/]быстрым займом без процентов[/url] на нашем сайте. Наши условия приятно удивят: минимальные требования к заемщикам, нулевые проценты для новых клиентов и мгновенное зачисление средств. Не упустите шанс решить свои финансовые вопросы быстро и без лишних переплат!
Решение ваших финансовых вопросов теперь в одном клике от вас. Наш сайт предлагает [url=https://microzaim.fun/]быстрый займ без процентов[/url], обеспечивая не только скорость, но и выгоду. Без скрытых платежей и дополнительных процентов – только честные условия для каждого клиента. Не упускайте возможность воспользоваться мгновенным финансовым решением, которое подойдет каждому.
В контексте [url=https://credit-world.ru/]займов без отказа на карту[/url], следует отметить их растущую популярность среди населения, особенно в условиях экономической нестабильности. Основными преимуществами таких займов являются удобство и быстрота получения средств. Однако важно учитывать, что такая доступность может привести к необдуманным финансовым решениям и увеличению долговой нагрузки на заемщиков.
Как моряк, ищущий пристанище в шторм, так и человек, оказавшийся в трудной финансовой ситуации, обращается к [url=https://credit-world.ru/]займам на карту без отказов[/url]. Эти займы – словно спасательный круг в море неопределенности, предлагая быстрое и надежное решение для тех, кто борется с волнами экономической нестабильности.
I want to to thank you for this excellent read!! I certainly enjoyed every little bit of it. I’ve got you book marked to check out new stuff you post
Обзор [url=https://a-kovka.ru/ograzhdenie-kovanoe]кованых ограждений[/url] показывает, как многофункциональны эти изделия. В “А-ковка” ограждения выполняют не только защитную функцию, но и служат элементом декора, добавляя шарм и утонченность как частным, так и общественным пространствам.
Разглядывая [url=https://a-kovka.ru/kovanye-zabory]кованый забор[/url], можно увидеть не просто ограждение, но и настоящее произведение искусства. В “А-ковка” они создаются не только для защиты, но и для украшения территории. Заборы отличаются уникальным дизайном и долговечностью, что делает их востребованными среди покупателей.
В эпоху перемен наш сайт остается незыблемой крепостью в мире микрозаймов. Мы сотрудничаем с множеством МФО, предлагая займы для тех, кто достиг 18 лет, без необходимости доказывать свою платежеспособность. Наша платформа предлагает вам выбор: получить деньги на карту, электронный кошелек или наличными. Информация на нашем сайте служит компасом в море микрофинансовых возможностей.
Mikro-Zaim-Online – [url=https://mikro-zaim-online.ru/]займ на кредитную карту[/url] ваши финансовые мечты – наша забота: займы на любые цели.
Pretty! This was an extremely wonderful post. Thanks for providing this info.
Психическое здоровье включает в себя наше эмоциональное, психологическое и социальное благополучие. Это влияет на то, как мы думаем, чувствуем и действуем. Оно также помогает определить, как мы справляемся со стрессом, относимся к другим и делаем здоровый выбор.
Психическое здоровье важно на каждом этапе жизни: с детства и подросткового возраста до взрослой жизни.ние) — специалист, занимающийся изучением проявлений, способов и форм организации психических явлений личности в различных областях человеческой деятельности для решения научно-исследовательских и прикладных задач, а также с целью оказания психологической помощи, поддержки и сопровождения.
Kudos. I appreciate it.
1win tv [url=https://1wincasino.milesnice.com/#]бонусы спорт 1win[/url] 1win официальный сайт зеркало букмекерской конторы
You’ve made your point!
1win ставки на спорт [url=https://1wincasino.milesnice.com/#]1win ваучер[/url] 1win почта
Thank you! I appreciate it.
1win lucky jet free cheat [url=https://1wincasino.milesnice.com/#]lucky jet 1win[/url] 1win invest отзывы
Many thanks! I enjoy this!
1win официальный сайт россия [url=https://1wincasino.milesnice.com/#]1win casino review[/url] как использовать бонусный счет 1win
Nicely put, Regards!
bc game reviews [url=https://bcgame.milesnice.com/#]shitcode bcgame[/url] bc game scam
Very good forum posts. Thanks.
su bc football game [url=https://bcgame.milesnice.com/#]army vs bc game[/url] bc game down
You actually reported that well!
приложение 1win [url=https://1win-casino.milesnice.com/#]1win зеркало[/url] как ставить на 1win
Incredible all kinds of great information!
bc game reddit [url=https://bcgamebonus.milesnice.com/#]bc crash game[/url] harvard bc basketball game
Amazing a good deal of very good data!
bc game kyc [url=https://bcgamebonus.milesnice.com/#]bc game coin[/url] bc game online
Thanks a lot, I value it!
1win og прогноз [url=https://1winkazino.milesnice.com/#]havu 1win прогноз[/url] бонусы казино 1win
You actually suggested this effectively!
1win ставки на спорт официальный сайт [url=https://1winkazino.milesnice.com/#]1win скачать на айфон[/url] 1win бесплатно приложение
Эффективное тепловая изоляция облицовки — уют и экономия в приватном здании!
Согласитесь, ваш жилье заслуживает первоклассного! Теплоизоляция обшивки – не просто решение для экономии на отоплении, это вложение средств в благополучие и стойкость вашего недвижимости.
✨ Почему изоляция с специалистами?
Опытность: Наша команда – профессиональные. Наша команда заботимся о каждом элементе, чтобы обеспечить вашему жилью идеальное воздухонепроницаемость.
Цена утепления: Мы все ценим ваш бюджет. [url=https://stroystandart-kirov.ru/]Утепление фасадов цена за м[/url] – начиная с 1350 руб./кв.м. Это вложение капитала в ваше комфортабельное будущее!
Энергосбережение: Забудьте о потерях тепла! Наш подход не только сохраняют тепловое комфорта, но и дарят вашему жилью новый уровень тепла энергоэффективности.
Преобразуйте свой домашний уголок комфортным и стильным!
Подробнее на [url=https://stroystandart-kirov.ru/]www.n-dom.ru
[/url]
Не предоставляйте без внимания свой жилье на произвольное стечение обстоятельств. Доверьтесь специалистам нашей компании и создайте комфорт вместе с нами-профессионалами!
Appreciate it. Numerous forum posts.
army vs bc game [url=https://bcgamecasino.milesnice.com/#]bc .game[/url] bc game tonight
Thanks a lot. I value it.
bc game usa [url=https://bcgamecasino.milesnice.com/#]bc game withdrawal time[/url] bcgame 入金ボーナス
Kudos, Loads of information.
1win контора бонус сите [url=https://1winoficialnyj.milesnice.com/#]1win официальный сайт[/url] 1win lucky jet free cheat
Nicely put, Thanks.
como usar o bonus da 1win [url=https://1winoficialnyj.milesnice.com/#]1win сайт[/url] казино 1win официальный
Kudos. Plenty of posts.
1win бесплатно приложение [url=https://1winregistracija.milesnice.com/#]1win казино зеркало[/url] как использовать бонусы в 1win
Very good material. Cheers.
jet best big win strategy jet winning jet 1win [url=https://1winregistracija.milesnice.com/#]1win вход зеркало[/url] “””отзывы о сайте 1win”””
Valuable advice. Cheers!
1win кейсы скачать [url=https://1winvhod.milesnice.com/#]lucky jet отзывы 1win[/url] 1win как скачать
Thanks! Plenty of tips.
1win казино регистрация [url=https://1winvhod.milesnice.com/#]1win aviator[/url] 1win og
Really quite a lot of helpful knowledge!
play european roulette [url=https://reddog.milesnice.com/#]red dog casino bonus[/url] play european roulette
Useful information. Thanks!
aladdin’s wishes [url=https://reddog.milesnice.com/#]red dog casino review[/url] btc blackjack
Superb stuff. Thanks.
minimum deposit casino [url=https://reddogcasino.milesnice.com/#]red dog casino review[/url] achiles the game
Thanks! A good amount of information.
high stakes games [url=https://reddogcasino.milesnice.com/#]reddog casino no deposit bonus codes[/url] red dog casino 100 free spins
Terrific facts, Appreciate it!
купон 1win [url=https://1winzerkalo.milesnice.com/#]как потратить бонусы спорт в 1win[/url] 1win net
Greetings from Colorado! I’m bored to tears at work so I decided to check out your site on my iphone during lunch break. I enjoy the info you present here and can’t wait to take a look when I get home. I’m shocked at how quick your blog loaded on my mobile .. I’m not even using WIFI, just 3G .. Anyhow, very good site!
Well expressed really! !
jet best big win strategy jet tricks jet 1win [url=https://1winzerkalo.milesnice.com/#]1win букмекерская контора скачать на андроид[/url] 1win официальный сайт вход в личный кабинет
Thank you. I enjoy this!
casino rtp [url=https://reddogcasinobonus.milesnice.com/#]reddog casino no deposit[/url] credit card online casino
Whoa a lot of fantastic knowledge!
online blackjack fake money [url=https://reddogcasinobonus.milesnice.com/#]red dog casino no deposit bonus[/url] casino rtp
Nicely put. Many thanks!
1win бесплатно android [url=https://zerkalo1win.milesnice.com/#]1win скачать мобильная версия[/url] 1win телефон приложение
Appreciate it, Plenty of knowledge!
1win зеркало вход [url=https://zerkalo1win.milesnice.com/#]1win зеркало сейчас[/url] 1win бонусы спорт как пользоваться
Really a good deal of wonderful information.
online free blackjack [url=https://reddogcasinologin.milesnice.com/#]online video poker game[/url] lucky6
Nicely put. Thanks.
real money video poker [url=https://reddogcasinologin.milesnice.com/#]high stakes games[/url] casino banking
Today, I went to the beach with my kids. I found a sea shell and gave it to my 4 year old daughter and said “You can hear the ocean if you put this to your ear.” She put the shell to her ear and screamed. There was a hermit crab inside and it pinched her ear. She never wants to go back! LoL I know this is entirely off topic but I had to tell someone!
You reported it very well!
1win top [url=https://zerkalo-1win.milesnice.com/#]1win скачать[/url] бонус на казино 1win
Amazing advice. Thank you!
бонусы спорт в 1win [url=https://zerkalo-1win.milesnice.com/#]как вывести деньги 1win[/url] 1win бонус
Very good posts. Thanks a lot.
how to download 1xbet app [url=https://1xbet-casino.milesnice.com/#]1xbet yangi versiyasi[/url] 1xbet sports
Information very well taken..
1xbet скачать [url=https://1xbet-casino.milesnice.com/#]1xbet download[/url] 1xbet слоты официальный сайт
Seriously quite a lot of terrific data!
ваучер 1win сегодня [url=https://1wincasino.milesnice.com/#]1win букмекерская контора[/url] 1win aplicativo
Awesome info. With thanks!
1win букмекерская контора официальный сайт [url=https://1wincasino.milesnice.com/#]бк 1вин на спорт и для 1win 2024 1win 1вин[/url] скачать бесплатно приложение 1win бесплатно
Тогда же ему впервые довелось столкнуться с неприятием его образа мыслей. Наряду с иными творческими изысканиями Ницше стоит отметить его учебное сочинение о малоизвестном немецком поэте Гёльдерлине, считавшемся сумасшедшим и недостойным. Особенностью этого сочинения было и то, что его автор прямо рекомендовал читать и ценить творения этого «странного» литератора. Оригинальность и самобытность такого подхода были удостоены низшего балла. Люди из приполярных областей отличают психически больных людей от знахарей и шаманов так: психически больной человек одержим духами и демонами; знахарь или шаман, в свою очередь, это тот, кто хотя и тоже одержим, но способен сам снова освободить себя? В то время как первые две упомянутые трудности могут быть преодолены только при помощи терпения или при помощи смелости быть объективным, лучший способ справиться с голосом скептика, по моему опыту, — это просто позволить ему говорить, а затем ответить ему: «Вполне возможно, что это нереально, но на данный момент я собираюсь двигаться дальше». Обычно затем происходит нечто, что убеждает нас в сверхъестественном существовании, независимой реальности партнёра по разговору. Человек осознаёт, что «Я никогда не мог быть придумать всё это сам сознательно». Является ли активное воображение истинным или нет, может быть лучше всего понято по его эффекту, так как он громаден и замечается сразу же, вне зависимости от того, позитивен он или негативен. Именно поэтому активное воображение является таким опасным инструментом и вообще не должно использоваться без наблюдения опытного человека. Оно может, как подчёркивал Юнг, привести скрытый психоз к точке прорыва[6]. В такой точке пациенты могут впадать в психотические эпизоды прямо в разгаре воображения.
Factor very well used..
скачать игру 1xbet бесплатно на андроид [url=https://1xbetkazino.milesnice.com/#]1xbet online betting[/url] пополнение 1xbet
Regards! Fantastic information.
1xbet игровые автоматы скачать на андроид [url=https://1xbetkazino.milesnice.com/#]1xbet не выводит деньги 2023[/url] 1xbet промокод 2024
Nicely put, Many thanks!
1win официальный сайт зеркало онлайн [url=https://1win-casino.milesnice.com/#]как использовать бонусы в 1win[/url] 1win как вывести деньги
Terrific material. Appreciate it!
1win букмекерская контора официальный сайт зеркало [url=https://1win-casino.milesnice.com/#]1win aviator[/url] 1win скачать приложение официального сайта
Thank you. I like it.
1xbet kz [url=https://1xbetoficialnyj.milesnice.com/#]1xbet отзывы[/url] 1xbet descargar
Wow plenty of superb knowledge!
1xbet desktop [url=https://1xbetoficialnyj.milesnice.com/#]1хбет скачать на андроид 1xbet android[/url] скачать рабочий 1xbet на андроид
This is nicely said. .
1win вход в личный кабинет [url=https://1winkazino.milesnice.com/#]промокоды на 1win[/url] 1win site
With thanks! Loads of material.
1win aviator скачать [url=https://1winkazino.milesnice.com/#]1win vs aurora[/url] lucky jet в 1win как обыграть отзывы казино
You actually suggested it superbly!
1xbet app ios [url=https://1xbetregistracija.milesnice.com/#]1xbet partner program[/url] 1xbet старые версии
You made your stand extremely clearly!!
1xbet партнерка [url=https://1xbetregistracija.milesnice.com/#]1xbet скачать приложение на телефон[/url] скачать приложение 1xbet зеркало на андроид бесплатно
Truly plenty of excellent facts.
1win детальная проверка ван вин промокоды [url=https://1winoficialnyj.milesnice.com/#]бк 1win[/url] 1win игра lucky jet отзывы
Wow many of helpful information.
1win вывод [url=https://1winoficialnyj.milesnice.com/#]ваучер 1win телеграм[/url] зеркало 1win рабочее на сегодня прямо сейчас
Superb data. Thank you!
1xbet личный кабинет [url=https://1xbetvhod.milesnice.com/#]1xbet partenaires[/url] 1xbet скачать android мгновенные выплаты
This is nicely said! !
как пополнить 1win с карты [url=https://1winregistracija.milesnice.com/#]1win бонус 5000 как использовать[/url] 1win рабочее зеркало прямо сейчас
Wonderful forum posts. Regards!
1win ставка [url=https://1winregistracija.milesnice.com/#]1win кс го[/url] скачать 1win с официального сайта
With thanks, Lots of postings.
app 1xbet [url=https://1xbet-vhod.milesnice.com/#]установить приложение 1xbet на андроид бесплатно[/url] все еще самая лучшая бк в 2024 1xbet обзор
Nicely put, Thanks.
1xbet ru скачать приложение на андроид [url=https://1xbet-vhod.milesnice.com/#]промокоды 1xbet[/url] 1xbet inscription
В далекой-далекой стране, в мире, где образование было волшебным ключом к будущему, жил молодой человек по имени Иван. У Ивана были большие мечты, но он не мог их осуществить, потому что у него не было аттестата. Однажды, он услышал от загадочного странника, что можно [url=https://www.dyplom-rossia.com/]аттестат куплю[/url], и решил воспользоваться этим шансом. Как только аттестат оказался в его руках, Иван почувствовал, что его мечты начинают сбываться. Он отправился в большой город, полный надежд и радости. Но вскоре он понял, что без знаний и умений, которые должен был дать аттестат, его мечты остаются недостижимыми. Он понял, что настоящее волшебство кроется не в бумаге, а в знаниях и усилиях, затраченных на их получение. И с этим пониманием, Иван решил вернуться к учебе, чтобы действительно осуществить свои мечты.
В центре внимания общественности оказалась недавняя тенденция на рынке образовательных услуг в Москве – продажа дипломов техникумов. Предложения о [url=https://www.dyplom-rossia.com/]купить диплом техникума в Москве[/url] стали встречаться все чаще, вызывая беспокойство среди экспертов образования и правоохранительных органов. Наше журналистское расследование направлено на выяснение, как эти услуги функционируют, кто их предлагает и каковы потенциальные риски для покупателей. По словам юристов, использование поддельных дипломов может привести к серьезным юридическим последствиям. Образовательные специалисты подчеркивают, что настоящее образование несет в себе гораздо большую ценность и важность для профессионального роста и развития личности. Этот репортаж исследует глубинные причины данной проблемы и возможные пути её решения.
You actually suggested it terrifically!
1xbet terms and conditions [url=https://1xbetzerkalo.milesnice.com/#]1xbet промокод на бесплатную ставку 2023[/url] programme 1xbet
Cheers! Numerous content.
1xbet affiliate program [url=https://1xbetzerkalo.milesnice.com/#]как скачать 1xbet на айфон[/url] 1xbet скачать зеркало
You have made your position pretty well.!
1win самолет [url=https://1winvhod.milesnice.com/#]1win вход[/url] como usar o bonus da 1win
You suggested it well!
1win ставки скачать приложение [url=https://1winvhod.milesnice.com/#]1win зеркало[/url] 1win lucky jet отзывы
You actually revealed it fantastically.
1xbet скачать старое обновление [url=https://zerkalo1xbet.milesnice.com/#]1xbet скачать официальный сайт[/url] мобильное приложение 1xbet
Nicely put. Regards!
1xbet промокод при регистрации [url=https://zerkalo1xbet.milesnice.com/#]1xbet download[/url] как скачать 1xbet на айфон в россии
Nicely put, Thank you.
jet best big win strategy jet winning tricks jet 1win [url=https://1win-vhod.milesnice.com/#]1win fnatic[/url] 1win cs go
Seriously quite a lot of beneficial tips!
1win coin что это [url=https://1win-vhod.milesnice.com/#]1win вход[/url] 1win узбекистан
Seriously plenty of terrific knowledge!
скачать программу 1xbet на андроид [url=https://zerkalo-1xbet.milesnice.com/#]1xbet скачать официальный сайт[/url] 1xbet казино официальный сайт
Cheers! A lot of information.
programme 1xbet [url=https://zerkalo-1xbet.milesnice.com/#]partner 1xbet[/url] казино 1xbet скачать
You said it very well..
1win букмекерская контора регистрация [url=https://1winzerkalo.milesnice.com/#]1win официальный сайт[/url] 1win это
Оцифровка документов москва — это наше направление. Мы предоставляем услуги перевода на цифровой носитель с использованием технологических инноваций. Сотрудничество с нами — это быстрый способ сделать вашу документацию доступной в электронной форме.
With thanks, Ample facts.
бонусы на спорт 1win как использовать [url=https://1winzerkalo.milesnice.com/#]aviator игра 1win авиатор игра[/url] скачать 1win с официального сайта
Whoa tons of very good material.
bc game clone [url=https://bcgame.milesnice.com/#]bc hash game[/url] fsu bc game 2023
You expressed it wonderfully.
bc game [url=https://bcgame.milesnice.com/#]bc game promo code no deposit[/url] bc game hack script
Superb info. Kudos!
1win register [url=https://zerkalo1win.milesnice.com/#]1win официальный сайт вход[/url] 1win бесплатно android
Awesome stuff. Appreciate it.
bc game shift codes [url=https://bcgamebonus.milesnice.com/#]bc game Ъ†ЫЊШіШЄ[/url] bc game alternative
Many thanks, I value it.
free shitcode bc game [url=https://bcgamebonus.milesnice.com/#]yale bc hockey game[/url] bc lions game today
Amazing data, Regards!
1win промокод [url=https://zerkalo1win.milesnice.com/#]1win 1000 рублей[/url] 1win скачать официальное приложение
After looking into a handful of the blog articles on your web page, I seriously appreciate your technique of blogging. I added it to my bookmark site list and will be checking back soon. Take a look at my web site too and let me know your opinion.
You made the point.
bc game crash predictor [url=https://bcgamecasino.milesnice.com/#]bc game hash[/url] bcgame 入金ボーナス
Nicely put. Many thanks!
bc game crypto [url=https://bcgamecasino.milesnice.com/#]bc game descargar[/url] bc game login
You’ve made your point very effectively..
1win бесплатно мобильная версия [url=https://zerkalo-1win.milesnice.com/#]1win казино зеркало[/url] бк 1вин на спорт и для 1win 2023 1win 1вин
Perfectly expressed certainly! .
1win казино официальный сайт [url=https://zerkalo-1win.milesnice.com/#]1win app download[/url] 1win aviator app download
You definitely made your point!
duke bc basketball game [url=https://bcgamelogin.milesnice.com/#]bc game shitcodes[/url] bc vt game
You explained it adequately!
what channel is the bc lions game on tonight [url=https://bcgamelogin.milesnice.com/#]bc game promo code no deposit[/url] exit game in richmond bc
Fine content. Thanks a lot!
скачать 1xbet на андроид последняя версия [url=https://1xbetcasino.milesnice.com/#]рабочее зеркало 1xbet[/url] зеркало 1xbet (1хбет) bkinfo-18 site
With thanks. Quite a lot of info!
online craps gambling [url=https://reddog.milesnice.com/#]red casino[/url] $10 minimum deposit casino usa
Fantastic stuff. Many thanks.
bitcoin blackjack [url=https://reddog.milesnice.com/#]red casino[/url] www reddogcasino
Cheers. Lots of information!
1xbet онлайн [url=https://1xbet-casino.milesnice.com/#]скачать старую версию 1xbet на андроид[/url] 1xbet официальный мобильный андроид скачать
You’ve made your stand very clearly!.
red dog casino 100 free spins [url=https://reddogcasino.milesnice.com/#]play slots for free no download[/url] enchanted casino real money
Wonderful facts. Many thanks!
online casino free chip [url=https://reddogcasino.milesnice.com/#]red dog casino no deposit codes[/url] free european roulette
You actually said that superbly!
скачать старую версию 1xbet на телефон андроид [url=https://1xbetkazino.milesnice.com/#]aviator игра 1xbet[/url] 1xbet login registration
Fantastic material. Thanks a lot.
play game 777 [url=https://reddogcasinobonus.milesnice.com/#]is reddog casino legit[/url] free chip no deposit casinos
Very well expressed really! .
online casino free chip no deposit [url=https://reddogcasinobonus.milesnice.com/#]reddogs casino[/url] slot machine rtp list
Nicely put, Regards!
1xbet официальный сайт скачать [url=https://1xbetkazino.milesnice.com/#]скачать 1xbet на android россия[/url] 1xbet india
You have made your position quite clearly.!
no deposit bonus codes for red dog casino [url=https://reddogcasinologin.milesnice.com/#]redog[/url] play craps online for real money
Whoa all kinds of very good advice!
european roulette casino [url=https://reddogcasinologin.milesnice.com/#]reddog casino[/url] cleopatra gold slot machine
You made your point very clearly!!
1xbet mobile скачать [url=https://1xbetoficialnyj.milesnice.com/#]1xbet пополнение счета[/url] 1xbet зеркало рабочее на сегодня прямо сейчас мобильная
Regards, I value this.
1xbet официальный сайт скачать приложение на телефон [url=https://1xbetoficialnyj.milesnice.com/#]1xbet официальный сайт[/url] промокод на бесплатные вращения 1xbet
Забудьте о отказах и переживаниях! [url=https://wikizaim.ru/]Займы на карту онлайн[/url] гарантируют 100% одобрение займа. Мы работаем с лучшими микрофинансовыми организациями, которые готовы поддержать вас в любой финансовой ситуации. Начните процесс получения займа прямо сейчас и убедитесь сами!
Забудьте о отказах и переживаниях! [url=https://wikizaim.ru/]Займы на карту онлайн[/url] гарантируют 100% одобрение займа. Мы работаем с лучшими микрофинансовыми организациями, которые готовы поддержать вас в любой финансовой ситуации. Начните процесс получения займа прямо сейчас и убедитесь сами!
[url=https://besthost.by/virtualhosting/]виртуальный хостинг[/url]
Когда речь заходит о создании своего собственного сайта, выбор хостинга играет огромную роль. Ведь именно хостинг будет определять доступность и быстродействие вашего сайта для пользователей. Если вы находитесь в Беларуси и ищете надежного хостинг-провайдера, то BestHost.BY – это отличный выбор. BestHost.BY – одна из ведущих хостинг-компаний в Беларуси, предоставляющая высококачественные услуги уже на протяжении 15 лет.
[url=https://afishkasochi.ru]1хбет зеркало рабочее сегодня[/url] – это популярная букмекерская компания, предлагающая широкий выбор ставок на спорт, казино и многое другое. С высокими коэффициентами и удобным интерфейсом, 1xbet привлекает множество игроков. Он также предоставляет возможность онлайн-трансляций событий. 1xbet создает захватывающий игровой опыт для своих пользователей.
Good day! I could have sworn Iíve visited this website before but after going through many of the articles I realized itís new to me. Anyways, Iím certainly happy I discovered it and Iíll be book-marking it and checking back regularly!
В 2023 году рынок финансовых услуг представляет [url=https://xn—–7kcgpnpy3bral5h.xn--p1ai/]займ 2023 год новые без отказа[/url], где более 40 ведущих МФО готовы предоставить быстрые финансовые решения. Эти займы отличаются простотой оформления и быстрым получением денег на карту. Они идеально подходят для тех, кто нуждается в срочном финансировании, с минимальными требованиями к заемщикам и высоким уровнем одобрения.
Новый год – это время для подарков и радости, но иногда для их реализации требуются дополнительные средства. [url=https://xn—–7kcgpnpy3bral5h.xn--p1ai/]Взять займ на новый год[/url] – это удобный способ получения срочной финансовой поддержки. МФО предлагают различные варианты займов, которые можно оформить онлайн, с мгновенным решением и быстрым переводом средств на карту, что делает праздник более светлым и беззаботным.
На финансовом рынке появилась услуга [url=https://xn—–7kcgpnpy3bral5h.xn--p1ai/]займы на карту без отказа мгновенно[/url], которая становится все более популярной. Это связано с тем, что такие займы предлагают быстрое решение финансовых проблем без долгого ожидания и с минимальными требованиями к заемщикам. Основное преимущество – это возможность получения денег мгновенно после одобрения заявки, что особенно важно в срочных ситуациях. Многие МФО предлагают простые и понятные условия для получения таких займов.
[url=https://xn—–7kcgpnpy3bral5h.xn--p1ai/]Займы онлайн на карту[/url] – это быстрый и удобный способ получения финансовой помощи. В 2023 году такие услуги стали еще доступнее благодаря новым технологиям. Вы можете оформить займ прямо с вашего смартфона, выбрав наиболее подходящие условия и сумму. Преимущества онлайн-займов включают моментальное решение по заявке и перевод средств на карту в кратчайшие сроки.
Новый год – это время для подарков и радости, но иногда для их реализации требуются дополнительные средства. [url=https://xn—–7kcgpnpy3bral5h.xn--p1ai/]Взять займ на новый год[/url] – это удобный способ получения срочной финансовой поддержки. МФО предлагают различные варианты займов, которые можно оформить онлайн, с мгновенным решением и быстрым переводом средств на карту, что делает праздник более светлым и беззаботным.
Wow, blog ini sangat keren! 🚀 Isinya begitu bertenaga dan bersemangat. 🌟 Selalu menemukan hal baru dan menarik di sini. 👏 Teruskan semangat berbagi pengetahuan! 🤩💯 Artikel ini memukau! 🌈 Terima kasih atas sumber inspirasinya! 🙌✨ #SemangatBerapi-api #Informatif #SukaBanget
Внезапные расходы больше не застанут вас врасплох. Наш сервис предлагает займы без процентов, которые вы можете получить мгновенно и в любое время суток. Никаких дополнительных платежей, только быстрое решение ваших финансовых вопросов. Мы предоставляем удобный и доступный способ получения средств, когда они вам наиболее необходимы.
У нас вы найдете [url=https://wikzaim.ru/]займ на карту без отказов[/url] и другие предложения от МФО 2024 года! Каждый сможет получить быстрый займ в онлайн режиме без проверок и отказов.
Нужны средства срочно, но беспокоитесь о своей кредитной истории? Наше МФО предлагает кредиты до 30 000 рублей с мгновенным одобрением, даже если ваша кредитная история далека от идеальной. Мы стремимся помочь каждому, кто нуждается в финансовой поддержке, предлагая простую процедуру оформления и быстрое решение по заявке. Никаких отказов – только своевременная помощь в сложной ситуации.
У нас вы найдете [url=https://wikzaim.ru/]займ на карту срочно без отказа[/url] и другие предложения от МФО 2024 года! Каждый сможет получить быстрый займ в онлайн режиме без проверок и отказов.
Срочно нужны деньги, но из-за плохой кредитной истории банки отказывают? Наше МФО предлагает уникальное решение: кредиты до 30 000 рублей без отказа, с мгновенным одобрением. Мы ценим каждого клиента и готовы помочь, даже если ваша финансовая репутация оставляет желать лучшего. Получите нужную сумму быстро и без лишних хлопот.
У нас вы найдете [url=https://wikzaim.ru/]займ на карту онлайн мгновенно без отказа[/url] и другие предложения от МФО 2024 года! Каждый сможет получить быстрый займ в онлайн режиме без проверок и отказов.
На нашем сайте вы найдете более 40 МФО, предлагающих кредиты новым клиентам под 0% с быстрой выдачей на карту. Процедура оформления кредита проще простого – понадобится только паспорт. Это отличная возможность получить необходимую финансовую поддержку без переплат и сложных бумажных процедур.
У нас вы найдете [url=https://wikzaim.ru/]микрозаймы онлайн без отказа[/url] и другие предложения от МФО 2024 года! Каждый сможет получить быстрый займ в онлайн режиме без проверок и отказов.
Финансовые вопросы могут возникнуть внезапно, и иногда необходимая сумма нужна срочно. Наш сервис предлагает удобное и простое решение – онлайн-займы на карту без проверок и отказов, доступные для любого гражданина России старше 18 лет. Всё, что вам нужно – это паспорт и желание решить свои финансовые вопросы быстро и без лишних хлопот.
Мы ценим ваше время и стремимся сделать процесс получения займа максимально простым и понятным. Вам не придется тратить время на походы в банк или ожидание одобрения. Все процедуры проходят онлайн, что позволяет вам оформить заявку в любое удобное время и из любого места, где есть доступ в интернет.
Основные преимущества наших онлайн-займов:
Без проверок кредитной истории: ваша кредитная история не будет препятствием для получения займа.
Быстрое решение: процесс одобрения занимает всего несколько минут.
Прозрачные условия: мы гарантируем отсутствие скрытых комиссий и платежей.
Удобство получения: средства моментально перечисляются на вашу банковскую карту.
Процесс оформления займа очень прост. Вам нужно только выбрать сумму и срок займа на нашем сайте, заполнить короткую анкету и предоставить данные вашего паспорта. После отправки заявки, решение будет принято в течение нескольких минут. И если оно положительное, средства поступят на вашу карту мгновенно.
Мы верим в то, что каждый должен иметь доступ к финансовым ресурсам в трудную минуту, и наша миссия – сделать этот доступ максимально простым и удобным. Возьмите онлайн-займ на карту сегодня и решите свои финансовые вопросы без лишних забот!
[url=https://clients1.google.li/url?q=https://wikzaim.ru/]взять кредит на карту срочно без отказа [/url]
[url=http://maps.google.ca/url?q=https://wikzaim.ru/]кредит без отказа онлайн на карту [/url]
[url=http://clients1.google.me/url?q=https://wikzaim.ru/]займы с плохой кредитной без отказа [/url]
[url=https://cse.google.pn/url?q=https://wikzaim.ru/]займ новые мфо 2023 [/url]
[url=http://maps.google.co.jp/url?sa=t&url=https://wikzaim.ru/]займ с плохой кредитной историей без отказа на карту [/url]
[url=https://images.google.hr/url?q=https://wikzaim.ru/]онлайн займ срочно без отказа [/url]
[url=https://www.google.me/url?q=https://wikzaim.ru/]займ без процентов [/url]
Финансовые вопросы могут возникнуть внезапно, и иногда необходимая сумма нужна срочно. Наш сервис предлагает удобное и простое решение – онлайн-займы на карту без проверок и отказов, доступные для любого гражданина России старше 18 лет. Всё, что вам нужно – это паспорт и желание решить свои финансовые вопросы быстро и без лишних хлопот.
Мы ценим ваше время и стремимся сделать процесс получения займа максимально простым и понятным. Вам не придется тратить время на походы в банк или ожидание одобрения. Все процедуры проходят онлайн, что позволяет вам оформить заявку в любое удобное время и из любого места, где есть доступ в интернет.
Основные преимущества наших онлайн-займов:
Без проверок кредитной истории: ваша кредитная история не будет препятствием для получения займа.
Быстрое решение: процесс одобрения занимает всего несколько минут.
Прозрачные условия: мы гарантируем отсутствие скрытых комиссий и платежей.
Удобство получения: средства моментально перечисляются на вашу банковскую карту.
Процесс оформления займа очень прост. Вам нужно только выбрать сумму и срок займа на нашем сайте, заполнить короткую анкету и предоставить данные вашего паспорта. После отправки заявки, решение будет принято в течение нескольких минут. И если оно положительное, средства поступят на вашу карту мгновенно.
Мы верим в то, что каждый должен иметь доступ к финансовым ресурсам в трудную минуту, и наша миссия – сделать этот доступ максимально простым и удобным. Возьмите онлайн-займ на карту сегодня и решите свои финансовые вопросы без лишних забот!
[url=http://images.google.st/url?q=https://wikzaim.ru/]займы новые мфо [/url]
[url=https://toolbarqueries.google.com.br/url?q=https://wikzaim.ru/]все займы без отказа [/url]
[url=https://toolbarqueries.google.com.sa/url?q=https://wikzaim.ru/]займы на карту новые мфо [/url]
[url=http://images.google.vu/url?sa=t&url=https://wikzaim.ru//]микрозаймы онлайн на карту без отказа [/url]
[url=https://clients1.google.com.hk/url?sa=i&url=https://wikzaim.ru/]займ онлайн 2023 [/url]
[url=https://maps.google.com.et/url?q=https://wikzaim.ru/]займы на карту новые [/url]
[url=https://maps.google.co.ls/url?q=https://wikzaim.ru/]кредит без отказа срочно [/url]
В нашем мире, где скорость и доступность играют ключевую роль, возможность получения финансовой поддержки в любое время дня и ночи становится не просто удобством, а необходимостью. Именно поэтому мы предлагаем вам список лучших МФО 2024 года, которые предоставляют займы на карту круглосуточно и без отказа онлайн.
Эти МФО были отобраны на основе их надежности, условий предоставления займов и отзывов клиентов. Они гарантируют простоту и прозрачность процесса, минимальное количество отказов и оперативность перевода средств.
Какие преимущества вы получаете:
Круглосуточный доступ: Не важно, в какое время вы решите обратиться за помощью, эти МФО всегда к вашим услугам.
Моментальное решение: Благодаря автоматизированным системам обработки заявок, вы получите решение в течение нескольких минут.
Без проверок кредитной истории: Ваши прошлые финансовые операции не повлияют на решение о выдаче займа.
Простая и удобная процедура: Весь процесс занимает минимум времени и сил.
Как выбрать лучшее предложение? На нашем сайте вы найдете подробные обзоры и сравнения различных МФО, что поможет вам сделать осознанный выбор. Мы учитываем все аспекты: от процентных ставок до скорости обслуживания.
С нами вы всегда можете быть уверены в том, что получите нужную сумму в самые короткие сроки. Наша миссия – обеспечить вас надежным финансовым решением, когда это действительно необходимо.
Выбирайте лучшие МФО 2024 года для займов на карту круглосуточно и без отказа на нашем сайте. Решайте свои финансовые вопросы легко и быстро!
[url=http://maps.google.co.ao/url?sa=t&url=https://wikzaim.ru/]займ на карту по паспорту [/url]
[url=https://images.google.cg/url?q=https://wikzaim.ru/]новое мфо [/url]
[url=https://maps.google.ci/url?q=https://wikzaim.ru/]онлайн займы на карту без отказа [/url]
[url=https://maps.google.pt/url?sa=t&url=https://wikzaim.ru/]микрозаймы без отказа онлайн на карту [/url]
[url=http://www.google.com.br/url?sa=t&url=https://wikzaim.ru/]займы онлайн на карту без отказа [/url]
[url=https://toolbarqueries.google.com.py/url?q=https://wikzaim.ru/]новые займы 2023 [/url]
[url=https://image.google.com.bh/url?q=https://wikzaim.ru/]займы онлайн новые мфо [/url]
В современном финансовом мире, где каждый ищет удобные и выгодные решения, [url=https://t.me/s/new_mfo_2024/]малоизвестные мфо дающие онлайн на карту[/url] предлагают уникальное предложение: первый займ под 0% до 30 000 рублей. Это отличная возможность для тех, кто ищет экстренное финансирование без дополнительных переплат. Такие условия делают займы доступными широкому кругу заемщиков, облегчая доступ к необходимым средствам.
Когда я узнал о срочной операции для близкого родственника, я искал быстрое решение. Нашел мир-займов.рф в Яндексе и получил [url=https://мир-займов.рф/]займы без отказа[/url] от МФО, входящих в Реестр ЦБ РФ.
Спонтанное решение отправиться в путешествие заставило меня искать финансовые средства. Я нашел решение на мир-займов.рф, взяв [url=https://мир-займов.рф/]взять займ на карту без отказа[/url]. Сайт предложил мне удобный выбор МФО, что существенно упростило процесс.
А знаете, что еще поразительно? [url=https://t.me/s/new_mfo_2024/]Новые мфо только открывшиеся без отказа на карту онлайн без звонков[/url] – они настоящие герои нашего времени! Представьте, вы можете получить займ, не отвлекаясь от ваших ежедневных дел. Никаких звонков, никаких вопросов, только вы, ваш смартфон и несколько кликов. Невероятно, правда?
Как только мне понадобились средства на неотложные нужды, я обнаружил [url=https://мир-займов.рф/]займ на карту без отказа госуслуги[/url]. Это было как находка в последний момент, спасающая мои планы!
Мой друг попал в неприятную ситуацию, когда ему срочно понадобились деньги на лечение. Я посоветовал ему сайт мир-займов.рф, где он быстро нашел [url=https://мир-займов.рф/]микрозайм на карту без отказа[/url] с удобными условиями.
Ну и кто бы мог подумать, что [url=https://doramahit.tv/doramy-novye/]новые дорамы[/url] так зацепят! Начал смотреть из любопытства, а они оказались просто захватывающими. Каждый новый сериал – это словно открытие нового мира, с его историями, героями, судьбами. И в каждом есть что-то такое, что заставляет снова и снова возвращаться к просмотру.
Разобраться в многообразии финансовых предложений может быть сложно, но на expl0it.ru мы упростили эту задачу. Здесь вы найдете [url=https://expl0it.ru/]займы список[/url], который включает в себя лучшие предложения от ведущих МФО. Каждое предложение тщательно отобрано, чтобы гарантировать вам выгодные условия и быстрое решение финансовых вопросов. Наш список займов — это удобный способ найти оптимальное решение, не тратя время на поиски.
Когда сроки имеют решающее значение, и вам нужно получить финансовую поддержку без задержек, expl0it.ru предлагает вам возможность [url=https://expl0it.ru/]взять деньги онлайн на карту мгновенно[/url]. Эта услуга специально разработана для предоставления быстрых финансовых решений. Наш процесс займа оптимизирован для максимального удобства и скорости, позволяя вам получить нужную сумму в кратчайшие сроки. Такая гибкость и оперативность делают наш сервис незаменимым инструментом в современных финансовых условиях.
Любители азиатских сериалов знают, что [url=https://doramahit.tv/]дорамы онлайн[/url] предлагают уникальный опыт погружения в мир азиатской культуры. С их помощью можно ощутить атмосферу другой страны, познакомиться с её традициями и ценностями. Дорамы отличаются от западных сериалов их особым стилем повествования и глубиной эмоций.
Когда срочно нужны деньги, expl0it.ru приходит на помощь. Мы предлагаем вам удобный и быстрый способ получить [url=https://expl0it.ru/]займ[/url], не выходя из дома. Наш сайт предоставляет широкий выбор предложений от надежных МФО. Процесс оформления максимально упрощен, а одобрение происходит в кратчайшие сроки. Воспользуйтесь нашими услугами, чтобы быстро получить необходимую сумму денег с минимальными усилиями.
Не упустите возможность воспользоваться нашими выгодными предложениями на expl0it.ru. Специально для вас мы собрали [url=https://expl0it.ru/]новые займы 2023[/url], которые помогут вам в сложных финансовых ситуациях. Наши займы отличаются удобными условиями, гибкими сроками и минимальными требованиями. Заходите на наш сайт и выберите оптимальное решение для себя.
Забота о недвижимости – это забота о удобстве. Термомодернизация фасадов – это не только стильный внешний вид, но и обеспечение тепла в вашем уголке уюта. Профессионалы, команда профессионалов, предлагаем вам преобразить ваше жилище в идеальный уголок для проживания.
Наши творческие решения – это не просто теплоизоляция, это искусство с каждым слоем. Мы разрабатываем гармонии между внешним видом и практической ценностью, чтобы ваш дом стал не только теплым, но и очаровательным.
И самое главное – доступная стоимость! Мы уверены, что высококачественные услуги не должны быть сверхдорогими. [url=https://ppu-prof.ru/]Утепление дома цена за квадратный метр[/url] начинается всего по цене от 1250 рублей за квадрат.
Применение современных технологий и материалов высокого качества позволяют нам создавать теплоизоляцию, которая долго служит и надежна. Забудьте о холодных стенах и дополнительных расходах на отопление – наше утепление станет вашим надежным защитником от холода.
Подробнее на [url=https://ppu-prof.ru/]http://www.ppu-prof.ru[/url]
Не откладывайте на потом заботу о приятности в вашем доме. Обращайтесь к профессионалам, и ваше жилье станет настоящим архитектурным шедевром, которое согреет вас не только теплом. Вместе мы создадим комфортабельное жилье, где вам будет по-настоящему комфортно!
В современном мире кинематографа турецкие сериалы набирают популярность, особенно в жанре триллеров. На странице [url=https://turkfan.tv/trillery/]триллеры турецкие сериалы[/url] на TurkFan.tv представлены уникальные проекты, которые завораживают зрителей своей напряженной атмосферой, непредсказуемыми сюжетами и мастерской игрой актеров. Эти сериалы искусно сочетают в себе элементы психологического триллера и драмы, заставляя зрителя не отрываться от экрана. Характерной особенностью турецких триллеров является глубокое изучение человеческой психологии и мотиваций персонажей, что делает каждую историю не просто захватывающей, но и многогранной.
В моей семье случились неожиданные медицинские расходы. Мы не могли рассчитывать на кредит в банке, но нашли портал с МФО 2024 года, который помог нам получить микрозайм и покрыть расходы.
[url=https://bycesoir.com/]Займы на карту онлайн[/url] от лучших МФО 2024 года – для получения займа до 30000 рублей на карту без отказа, от вас требуется только паспорт и именная банковская карта!
Моя старая холодильная камера вышла из строя, и мне срочно понадобилось купить новую, чтобы сохранить продукты. Мне посоветовали портал, где можно получить микрокредит всего за 5 минут, предоставив только паспорт и банковскую карту. Я воспользовался этим сервисом и в кратчайшие сроки смог приобрести новую холодильную камеру, избежав бедствия в хранении продуктов.
[url=https://bycesoir.com/]Займы на карту онлайн[/url] от лучших МФО 2024 года – для получения займа до 30000 рублей на карту без отказа, от вас требуется только паспорт и именная банковская карта!
Когда-то я работал в офисе, но мечтал о собственном бизнесе. Однажды рискнул и создал свой сайт. Нашел портал с МФО 2024 года, где дали микрозайм на развитие проекта. Это было началом моего предпринимательского пути.
[url=https://bycesoir.com/]Займы на карту онлайн[/url] от лучших МФО 2024 года – для получения займа до 30000 рублей на карту без отказа, от вас требуется только паспорт и именная банковская карта!
Мечтал отправить своего сына в летний лагерь, но финансы были ограничены. Благодаря порталу с МФО 2024 года, я смог взять микрозайм и сделать эту мечту реальностью для него. Это было невероятно важно для нашей семьи.
[url=https://bycesoir.com/]Займы на карту онлайн[/url] от лучших МФО 2024 года – для получения займа до 30000 рублей на карту без отказа, от вас требуется только паспорт и именная банковская карта!
А если вы предпочитаете смотреть сериалы в режиме онлайн, то [url=https://turkfan.tv/novie-turkey-serials/]новые турецкие сериалы онлайн[/url] на TurkFan.tv – это то, что вам нужно. Откройте для себя мир увлекательных историй, доступных для просмотра в любое удобное для вас время. Наслаждайтесь высококачественным контентом, выбирая из широкого ассортимента новых сериалов. TurkFan.tv постоянно обновляет свою коллекцию, чтобы вы всегда имели доступ к самым актуальным и интересным турецким проектам. Приглашаем вас в мир новых турецких сериалов – выбирайте, смотрите и наслаждайтесь!
Моя машина сломалась, и без нее я не мог добираться на работу. Страховая компания не покрыла все расходы. Я нашел портал с МФО 2024 года и получил микрозайм, чтобы починить авто и вернуться к нормальной жизни.
[url=https://bycesoir.com/]Займы на карту онлайн[/url] от лучших МФО 2024 года – для получения займа до 30000 рублей на карту без отказа, от вас требуется только паспорт и именная банковская карта!
Когда сердце жаждет историй, наполненных страстью, любовью и интригами, сайт, где можно [url=https://doramaclub.tv/luchshie-doramy/]лучшие дорамы онлайн[/url] смотреть, становится настоящим спасением. Здесь каждая дорама – это шедевр, выбранный среди множества, чтобы подарить вам незабываемые часы просмотра. Откройте для себя мир, где каждый сюжет – это путешествие, полное эмоций и удивительных поворотов, которые заставят ваше сердце биться чаще.
Уникальная возможность выигрыша ждет вас в Lucky Jet краш-игре от 1win! Проверьте свою интуицию прямо сейчас.
В 2024 году наблюдается рост популярности круглосуточных займов от совсем новых микрофинансовых организаций (МФО). Эти МФО выделяются автоматической и быстрой выдачей онлайн займов на любую банковскую карту. Однако стоит отметить, что они предоставляют займы без проверки кредитной истории и звонков клиентам.
[url=https://www.ozersk74.ru/communication/forum/?PAGE_NAME=profile_view&UID=68029]Займы в новых МФО: анализ предложений и условий[/url]
[url=http://www.web-analitik.info/forumhost/user/173989/]Новые МФО: на пути к финансовой стабильности[/url]
[url=https://fashion-id.ru/forum/user/67448/]Новые МФО без отказа на карту онлайн без звонков[/url]
[url=https://xn—-btbthcge4aikr4i.xn--p1ai/forum/user/27838/]Новые МФО онлайн: СЂРѕСЃС‚ популярности займов[/url]
[url=http://www.schoolpress.ru/news_schoolpress/forum/index.php?PAGE_NAME=profile_view&UID=205165]Малоизвестные МФО на карту: сравниваем предложения[/url]
Срочно нужен займ? На нашем портале вы найдете ответ на этот вопрос! В 2024 году малоизвестные МФО предлагают новые займы на карту сроком до 15 дней под 0%. Это уникальное предложение, которое доступно каждому. Оформите заявку, получите деньги на карту и не переплачивайте проценты!
[url=https://ujkh.ru/forum.php?PAGE_NAME=profile_view&UID=110578]Малоизвестные МФО: качество обслуживания[/url]
[url=http://www.led119.ru/forum/user/101502/]Малоизвестные МФО: скрытые возможности[/url]
[url=https://xn—-btbthcge4aikr4i.xn--p1ai/forum/user/27838/]Новые МФО онлайн: СЂРѕСЃС‚ популярности займов[/url]
[url=https://bestnet.ru/support/forum/index.php?PAGE_NAME=profile_view&UID=131088]Малоизвестные займы на карту без отказа: разбираемся в деталях[/url]
[url=http://www.test.sozapag.ru/forum/user/219406/]Малоизвестные МФО на карту: выбор среди новичков[/url]
Не ждите, когда вам понадобятся средства. В 2024 году лучшие микрофинансовые организации предоставляют быстрые микрозаймы без отказа всего за 5 минут. На нашем сайте вы найдете подборку таких МФО с минимальными требованиями к заемщику. Оформите заявку и получите средства на карту моментально, не теряя времени на ожидание!
[url=https://elitesm.ru/forum/user/10005/]Новые МФО: как выбрать лучшего партнера[/url]
[url=http://www.web-analitik.info/forumhost/user/173989/]Новые МФО: на пути к финансовой стабильности[/url]
[url=https://www.click-boutique.ru/forum/?PAGE_NAME=profile_view&UID=39359]Малоизвестные МФО: безопасность и надежность[/url]
[url=https://goodgame.kz/communication/forum/user/102237/]Новые МФО: революция в финансовых услугах[/url]
[url=https://horoshava.ru/forum/user/33152/]Новые займы онлайн: быстро и просто[/url]
2024 год привел к расцвету новых микрофинансовых организаций (МФО), предоставляющих срочные займы. На нашем сайте мы собрали подборку таких МФО, которые малоизвестны, но предлагают выгодные условия для онлайн займов. Независимо от вашей кредитной истории, вы можете получить нужные средства в течение минут. Забудьте о бюрократии и очередях в банке – новые МФО делают процесс максимально удобным!
[url=http://whdf.ru/forum/user/70802/]Малоизвестные МФО: история успеха[/url]
[url=https://fashion-id.ru/forum/user/67448/]Новые МФО без отказа на карту онлайн без звонков[/url]
[url=https://www.repropark.ru/mag/forum/user/2942/]Малоизвестные займы на карту без отказа: реальная возможность[/url]
[url=https://rezonit.ru/forum/user/41182/]МФО новые: находите своего надежного партнера[/url]
[url=https://stylist-profi.ru/forum/user/194318/]Малоизвестные МФО предоставляющие онлайн займы[/url]
Great blog! I’m considering a writing role. Could you share details on the application process?
If some one desires expert view concerning blogging then i suggest him/her to go to see this blog, Keep up the nice job.
At [url=https://mult34.com/category/the-incredibles/]incredibles porn[/url] – mult34.com, we understand what guys want in adult comics. Our portal is a treasure trove of stories that add color and excitement to your evenings. With a vast array of genres, all available for free, you’re guaranteed to find something that suits your style. Whether it’s a journey through sci-fi landscapes or a dive into the depths of fantasy, [url=https://mult34.com/]porn cartoon[/url] – mult34.com delivers an escape from the ordinary.
Мне срочно требовались новые туфли к важному мероприятию. Я нашла mikro-zaim-online.ru в ТОПе Яндекса и благодаря [url=https://mikro-zaim-online.ru/plohaya-kreditnaya-istoriya/]взятию займа на карту с плохой историей[/url], моя проблема была решена. Заявки в несколько МФО были одобрены быстро и на выгодных условиях.
Embark on an adventurous journey through the world of adult comics at [url=https://mult34.com/tag/yaoi-gay-porn-comics/page/2/]anime gay porn[/url] – mult34.com. This site offers an extraordinary array of genres, all free of charge. It’s a place where every style, narrative, and artistic expression in adult comics is celebrated. [url=https://mult34.com/dick-harley-quinn/]harley quinn porn[/url] – mult34.com is dedicated to bringing together the best and most diverse selection, ensuring that every visit is a new discovery.
В Facebook я наткнулся на рекомендацию сайта mikro-zaim-online.ru. Люди делились, как они получали [url=https://mikro-zaim-online.ru/plohaya-kreditnaya-istoriya/]займ онлайн с плохой кредитной историей[/url] для осуществления своих мечт. Вдохновившись, я также воспользовался этим сайтом, чтобы сделать подарок девушке, в которую влюблен.
Когда моя любимая собака заболела, мне срочно понадобились деньги на её лечение. В поиске решения я обратился к Google и нашел сайт mikro-zaim-online.ru. Здесь я быстро получил [url=https://mikro-zaim-online.ru/plohaya-kreditnaya-istoriya/]займы на карту без отказа с плохой кредитной историей и просрочками[/url], что позволило мне оплатить лечение и вернуть радость моему питомцу.
Embark on an adventurous journey through the world of adult comics at [url=https://mult34.com/category/steven-universe/]steven universe porn[/url] – mult34.com. This site offers an extraordinary array of genres, all free of charge. It’s a place where every style, narrative, and artistic expression in adult comics is celebrated. [url=https://mult34.com/category/teen-titans/]teen titans porn[/url] – mult34.com is dedicated to bringing together the best and most diverse selection, ensuring that every visit is a new discovery.
Мечтая о перспективном джипе, я решил приобрести один, но мои финансы были ограничены, и у меня была плохая кредитная история. Тогда мой друг посоветовал посетить сайт msk-zaim.ru, где я нашел МФО, специализирующуюся на займах для клиентов с просрочками и плохой кредитной историей. Благодаря этому сайту, я смог приобрести свой мечтанный джип и сделать его моим надежным спутником.
[url=https://msk-zaim.ru/]Займы срочно – возьмите займ на покупку книг[/url]
Я большой ценитель кофе, и мечтал о качественной кофемашине для создания уютных утренних ритуалов. Однако, она была далеко не дешевой, и мои сбережения были недостаточны. В социальных сетях я обнаружил ссылку на сайт, где представлен список новых МФО 2024 года. Заявка была подана быстро, и я получил одобрение. Деньги моментально поступили на мою банковскую карту, и теперь каждое утро я наслаждаюсь ароматным кофе, готовым моей новой кофемашиной.
[url=https://msk-zaim.ru/]Займы онлайн – решение для тех кому нужны деньги моментально[/url]
Thank you, I have recently been searching for information approximately this topic for a while and yours is the best I have found out so far. However, what about the conclusion? Are you sure about the source?
Однажды меня охватило желание отправиться в неожиданное путешествие и исследовать новые места. Но наличные были ограничены. Благодаря Яндексу, я нашла сайт msk-zaim.ru, где была подборка МФО 2024 года. Здесь я смогла взять микрокредит на 10 000 рублей под 0%, и это стало стартовой точкой для моего спонтанного приключения.
[url=https://msk-zaim.ru/]Займ с плохой КИ – доступ к финансам для всех независимо от истории[/url]
Стиральная машина сломалась, и моя жизнь стала намного менее комфортной. Я начал искать способы замены её, но бюджет был ограничен. В одной из групп в социальной сети я наткнулся на рекламу сайта, где представлен огромный список новых МФО 2024 года. С помощью этого сайта, мне удалось получить займ моментально и купить новую стиральную машину. Теперь моя бытовая жизнь стала намного удобнее.
[url=https://msk-zaim.ru/]Займы онлайн – на ремонт ноутбука[/url]
Explore the thrilling world of adult anime-style comics at Hentailoop.com – [url=https://hentailoop.com/characters/android-21/]android 21 hentai[/url] , where fantasy becomes reality on every page. In this diverse collection, you’ll find stories that captivate and intrigue, from epic adventures to heartwarming tales, all illustrated in stunning detail. Whether you’re a fan of classic anime or seeking something new and exciting, Hentailoop.com – [url=https://hentailoop.com/tag/incest/]nhentai incest[/url] has it all. Each comic is a masterpiece, weaving intricate plots with breathtaking artistry, guaranteed to transport you to other worlds. Perfect for unwinding after a long day, these comics offer an escape into a realm of imagination and wonder. So why wait? Dive into the vast universe of adult comics at Hentailoop.com – [url=https://hentailoop.com/releases/2023/]hentai comic[/url] and let your evening adventure begin!
Недавно, одним вечером, я и моя подруга решили найти что-то новенькое для просмотра. Давай посмотрим аниме! – предложила она. Мы сразу же включили Google и начали поиск. Первым в списке выскочил сайт [url=https://animehit.tv/anime-2024/]смотреть аниме 2024 онлайн[/url]. Не веря своему счастью, мы открыли его и офигели – там был просто огромный выбор аниме на русском языке, и все в HD 720 качестве! Мы выбрали несколько сериалов и устроились удобно. Весь вечер прошёл в атмосфере увлекательных историй, и мы даже не заметили, как пролетели часы.
Микрозаймы – идеальное решение для тех, кто ищет маленькую сумму на короткий срок. Процесс оформления прост и не займет много времени. Получите необходимую сумму уже сегодня!
[url=http://депутат-ширинов.рф/content/mikro-zaim-kredit-otkaza-srochno]деньги на карту онлайн без отказа[/url]
[url=https://stareishina.com/gallery/image/3381-mikro-zaim-zaimy-onlain-na-kartu-bez-otkaza-srochno/]взять займ срочно без отказа на карту онлайн[/url]
[url=http://g29869x8.beget.tech/2024/01/15/mikro-zaim-kredity-na-kartu-onlayn-bez-otkaza-kruglosutochno.html]деньги без отказа на карту[/url]
[url=https://oldmerin.club/board/index.php?app=gallery&image=14947]займ без отказа онлайн[/url]
Ready to turn your evening from mundane to extraordinary? Hentailoop.com – [url=https://hentailoop.com/manga/lostperras-komi-alternate-universe-cartoon-porn/read/p/7/]cartoonporn[/url] is here to help! Our portal offers a diverse range of adult comics that cater to every guy’s taste. But that’s not all – these comics are perfect for sharing with your girlfriend, adding a fun and spicy twist to your night together. Our collection is constantly updated with new comics and stunning images, making every visit a fresh adventure. Whether you’re seeking something bold or subtly enticing, Hentailoop.com – [url=https://hentailoop.com/tag/gangbang/]hentai gangbang[/url] has got you covered. Join us and make your evenings not just fun, but absolutely sizzling!
Моему другу, который недавно увлёкся японской культурой, было трудно найти сайт, где он мог бы смотреть [url=https://animehit.tv/]аниме онлайн в хорошем качестве[/url]. Большинство платформ предлагали контент среднего качества или с ограниченным выбором. Посоветовав ему этот сайт, я был рад увидеть его восторг, когда он обнаружил коллекцию аниме с высоким разрешением изображения. Теперь он проводит вечера, изучая различные жанры и серии, наслаждаясь чёткостью и качеством картинки, что делает его погружение в мир аниме по-настоящему полным.
Представьте моё удивление, когда я обнаружил, что мой кот – тайный фанат [url=https://animehit.tv/anime-novinki/]новых аниме[/url]! Каждый раз, как только я включаю новую серию, он занимает самое удобное место перед экраном и начинает смотреть с таким вниманием, будто пытается понять сюжет. Может, ему нравится яркая анимация? – думал я, но нет, кажется, он просто выбирает своего любимого героя. Как-то раз он даже начал мяукать в унисон с персонажами! Теперь у нас с котом есть совместное увлечение – мы вместе ищем самые интересные новинки аниме и проводим вечера за просмотром. Хотя, конечно, его комментарии оставляют желать лучшего!
Чуваки, недавно попал на крутой сайт, где можно [url=https://animehit.tv/]смотреть аниме в хорошем качестве[/url]. Просто бомба, какой там выбор! Решил затестить один сериал, и офигел – картинка четкая, как будто ты прям в этом аниме. И знаете что? Ноль лагов, все летает. Короче, теперь у меня есть фишка, где качественно углубиться в мир аниме, не парясь насчет качества картинки.
Ready to turn your evening from mundane to extraordinary? Hentailoop.com – [url=https://hentailoop.com/manga/giving-back-will-you-have-sex-with-my-mom/]sex with mom[/url] is here to help! Our portal offers a diverse range of adult comics that cater to every guy’s taste. But that’s not all – these comics are perfect for sharing with your girlfriend, adding a fun and spicy twist to your night together. Our collection is constantly updated with new comics and stunning images, making every visit a fresh adventure. Whether you’re seeking something bold or subtly enticing, Hentailoop.com – [url=https://hentailoop.com/tag/futanari/]hentai futa[/url] has got you covered. Join us and make your evenings not just fun, but absolutely sizzling!
Отличная статья, благодарю!
В качестве благодарности поделюсь с вами информацией: наличники из дерева на окна и двери в СПб для коттеджей являются популярным выбором среди владельцев домов.
[url=https://kub-era.ru/nalichniki]Деревянные наличники на окна купить в спб[/url] для коттеджей – это превосходный вариант, который сочетает в себе эстетику, прочность и экологию. Если вы хотите придать своему коттеджу особый шарм, рассмотрите деревянные наличники.
В Питере существует множество компаний, специализирующихся на изготовлении и установке деревянных наличников. Одна из них – компания КубЭра. Предлагает широкий выбор моделей, цветов и отделок.
Looking for a way to make your evenings more thrilling and enjoyable? Dive into the world of Hentailoop.com – [url=https://hentailoop.com/artists/suoiresnu/]suoiresnu[/url] ! Our site is a haven for adult comics, designed with guys in mind but great for couples too. Explore our vast selection, ranging from the adventurous to the romantically risque. We keep our collection fresh and exciting with daily updates of new comics and eye-catching images. Whether you’re in the mood for a solo adventure or a shared experience with your partner, Hentailoop.com – [url=https://hentailoop.com/tag/reverse-rape/]rape hentai[/url] is your go-to destination for an evening full of surprises and delights!
Dealing with an [url=https://social-me.co.uk/]instagram account deleted[/url] can be a complex process, but our expertise at social-me.co.uk makes it manageable. We have a proven track record in recovering deleted Instagram accounts, understanding the intricacies of Instagram’s policies to navigate through the recovery process. Our service is comprehensive, offering not just account recovery but also advice on how to maintain a healthy and compliant Instagram presence.
Секс развлечения для Москвы зовет ознакомиться с биографией красивых девчонок, которые не прочь скрасить Ваш вечер или ночь уже сегодня. На интернет сайте newswomanblog.win Вы найдете партнершу на всякий вкус, как для молодых людей, так и для женщин, а также семейных пар.
По поиску [url=https://newswomanblog.win/zrelye]старые индивидуалки[/url] заходите к нам в настоящий момент. Если Вы измучены от скучных и одинаковых будней, а ещё трудных рабочих смен, то пришло время побаловать себя и получить огромное развлечение.
Сделать лучшим любой вечер, а также событие помогут красивые и обладающие отличным опытом девочки, которые приедут в любое выбранное Вами место. Будь то отель или баня, а также событие – день рождения или мальчишник. По любым вопросам мы готовы Вам помочь, чтобы заказчик был удовлетворен целиком.
Приобрести секс услуги возможно здесь [url=https://newswomanblog.win/]снять индивидуалку[/url] уже сегодня. Мы производи работу без выходных и праздничных дней, круглосуточно. На представленном веб ресурсе возможно даже пользоваться быстрым поиском девочки, введя: Ваш район, возраст девушки, рост, размер груди, стоимость. Так Вам будет проще сделать выбор под свои собственные критерии. А также у нас имеются специальные категории: девушки без ретуши, VIP девушки, волосатые, проверенные и другие.
Интим досуг для Москвы зовет познакомиться с анкетами изумительных малышек, которые способны скрасить Ваш час или всю ночь уже сегодня. На сайте newswomanblog.win Вы найдете подругу на всякий вкус, как для мужчин, так и для женщин, а также влюбленных пар.
По вопросу [url=https://newswomanblog.win/deshevye]проститутки москвы дешево[/url] заходите к нам прямо сейчас. Если Вы напряжены от грустных и тусклых будней, а ещё загруженных рабочих дней, то пришло время порадовать себя и получить настоящее удовольствие.
Сделать лучшим любой вечер, а также праздник помогут красивые и знающие толк в своем деле девушки, которые придут в любое указанное Вами место встречи. Будь то мотель или квартира, а также мероприятие – день рождения или корпоратив. По оставшимся вопросам мы окажем Вам помощь, чтобы заказчик был удовлетворен в полном объеме.
Выбрать интим услуги возможно по ссылке [url=https://newswomanblog.win/seligerskaya]проститутки м селигерская[/url] уже сейчас. Мы осуществляем работу без выходных и праздничных дней, круглосуточно. На представленном интернет сайте возможно даже воспользоваться быстрым поиском проститутки, введя: Ваш район, возраст девушки, рост, размер груди, прайс. Так Вам будет легче осуществить выбор под свои собственные критерии. А также у нас есть специальные категории: негритянки, VIP проститутки, новые девушки, новые девушки и другие.
Посетите сайт turkbiz.tv, чтобы ознакомиться с [url=https://turkbiz.tv/serials2023/]турецкими сериалами 2023 на русском языке[/url]. Новые сериалы 2023 года открывают новые горизонты в турецкой драматургии, демонстрируя продолжение традиций и в то же время инновационные подходы в сюжетных линиях. Это не только развлечение, но и возможность для культурного обмена и понимания современного турецкого общества через кинематограф.
Excellent write-up
На сайте turkbiz.tv мы гордимся представить вам [url=https://turkbiz.tv/semeynie/]турецкие сериалы семейные[/url], которые станут идеальным выбором для семейного просмотра. Эти сериалы не просто развлекут вас, они подарят вам возможность окунуться в жизнь разных семей, пережить с ними радости и трудности, а главное – улучшить свои отношения с близкими. Подарите себе и своей семье моменты умиротворения и вдохновения с [url=https://turkbiz.tv/semeynie/]семейными турецкими сериалами[/url]!
A [url=https://social-me.co.uk/]disabled Instagram account[/url] can disrupt your digital life and business operations. At social-me.co.uk, we have developed a robust approach to tackle this problem. Our team has extensive experience in dealing with Instagram’s guidelines and user agreements, allowing us to navigate the reactivation process effectively. We don’t just aim to reactivate your account; our goal is to understand the root cause of the disablement and offer solutions that ensure the longevity and security of your account.
Facing an [url=https://social-me.co.uk/]unblocked on Facebook[/url] issue? At social-me.co.uk, we understand the frustration and disruption it causes. Our team is specialized in swiftly resolving such blockages, ensuring that your social media life gets back on track as quickly as possible. We don’t just unblock accounts; we provide a comprehensive service that includes preventative advice to avoid future blockages. Our commitment to customer satisfaction and our track record of success make us the go-to solution for all your Facebook-related issues.
Когда мне срочно понадобились деньги на оплату учебы, я вспомнил о рекомендации в социальных сетях и посетил mikro-zaim-online.ru. Благодаря удобному онлайн-сервису, я смог получить необходимую сумму и продолжить свое образование.
MIKRO-ZAIM – [url=https://mikro-zaim-online.ru/]микрокредиты[/url]
Наши контакты: Зеленодольская улица, 36к2, Москва, 109457
Когда требуется взвешенный и разумный подход к получению займа, mikro-zaim-online.ru предоставляет всю необходимую информацию для принятия обдуманного решения. Этот ресурс объединяет надежные МФО с удобными условиями кредитования.
MIKRO-ZAIM – [url=https://mikro-zaim-online.ru/]быстрый заим[/url]
Наши контакты: Зеленодольская улица, 36к2, Москва, 109457
Березин Андрей Евроинвест [url=https://www.mainfin.ru/persona/berezin-andrey-valeryevich/]https://www.mainfin.ru/persona/berezin-andrey-valeryevich/[/url].
С тех пор, как я начал заботиться о своем здоровье, я не могу представить свой день без свежевыжатого сока. Благодарю ‘все соки’ за их [url=https://blender-bs5.ru/collection/sokovyzhimalki-dlya-granata]соковыжималку гранат[/url], которая делает мой завтрак идеальным!
По вопросу [url=https://newswomanblog.win/deshevye]проститутки москвы дешево[/url] переходите к нам как можно скорее. Если Вы измучены от грустных и тусклых будней, а ещё загруженных рабочих смен, то пришло время наградитьни в чем не ограждать себя и получить настоящее удовольствие.!
Березин Андрей Евроинвест [url=https://lenta.ru/tags/persons/berezin-andrey/]https://lenta.ru/tags/persons/berezin-andrey/[/url].
[url=https://dzen.ru/spravkin]Быстрый онлайн займ на карту[/url] – это финансовая услуга, предоставляемая онлайн-платформами, которые обеспечивают оперативное оформление займов и перевод средств на банковские карты клиентов. Это современный способ получения финансовой помощи.
[url=https://dzen.ru/spravkin]Займы с плохой кредитной историей без отказа[/url] – это как луч света в конце тоннеля. Мы понимаем, что у каждого бывают трудности, и мы готовы помочь вам преодолеть их. Получите средства без отказа и начните новую главу своей финансовой истории!
Wow, this article is good, my sister is analyzing these things, so I am going to let know her.
Березин Андрей Евроинвест [url=https://finance.rambler.ru/person/berezin-andrey/]https://finance.rambler.ru/person/berezin-andrey/[/url].
По запросу [url=https://newswomanblog.win/slavyanskii-bulvar]проститутки славянский бульвар[/url] заходите к нам как можно скорее. Если Вы устали от тупых и серых будней, а также трудных рабочих дней, то пришло время порадовать себя и получить настоящее наслаждение.
Спасибо за информацию. В благодарность предлагаю расслабиться и окунуться в мир азарта:
[url=https://vavada-registraciya.online/]Играть в Вавада на деньги[/url] – это крутой метод, позволяющий вам окунуться в мир азартных игр в лучшем онлайн-казино. Зайдите на официальный сайт Вавада и нажмите на кнопку “Регистрация”. Затем заполните простую форму регистрации своими личными данными.После успешной регистрации вы получите доступ к широкому выбору азартных игр, таких как слоты, рулетка, блэкджек и многое другое. Казино Vavada также предлагает щедрые бонусы и промоакции, которые значительно повышают вероятность выигрыша. Играйте с умом и делайте ставки с лимитами. Регистрация в казино Vavada – это крутой метод завлечь себя азартными играми, получить удовольствие и, возможно, выиграть крупную сумму денег.
Березин Андрей Евроинвест [url=http://newizv.ru/news/2023-08-28/berezin-andrey-svedeniya-o-developere-419032/]http://newizv.ru/news/2023-08-28/berezin-andrey-svedeniya-o-developere-419032/[/url].
nice content!nice history!! boba 😀
wow, amazing
Думаешь о переводе денег из Дании в Россию? С denezhnye-perevody-iz-rossii.top это проще, чем кажется. Мы предоставляем тебе удобный и безопасный способ перевода. Не нужно беспокоиться о сложностях – наш сайт покажет тебе, как легко и быстро отправить деньги. С нашей помощью ты сможешь перевести свои средства без лишних хлопот и забот. Попробуй сам и убедись, насколько это удобно!
nice content!nice history!! boba 😀
nice content!nice history!! boba 😀
Если тебе нужно перевести деньги в Египет из России, не переживай, это проще, чем кажется. Зайди на denezhnye-perevody-iz-rossii.top, и ты найдешь все необходимое для этого. Мы покажем тебе простые шаги, чтобы твой перевод прошел быстро и безопасно. Не нужно беспокоиться о комиссиях и задержках, мы позаботимся о том, чтобы твои деньги были доставлены точно в срок.
Vyacheslav Nikolaev [url=http://spacecoastdaily.com/2023/08/vyacheslav-nikolaev-ecosystem-builder/]http://spacecoastdaily.com/2023/08/vyacheslav-nikolaev-ecosystem-builder/[/url].
В преддверии свадьбы мы столкнулись с непредвиденными расходами. Чтобы не отказываться от мечты, я воспользовался mikro-zaim-online.ru и получил необходимую сумму за 5 минут. Это позволило нам провести свадьбу так, как мы мечтали.
MIKRO-ZAIM – [url=https://mikro-zaim-online.ru/]займ взять срочно[/url]
Наши контакты: Зеленодольская улица, 36к2, Москва, 109457
nice content!nice history!! boba 😀
Outstanding, superb effort
wow, amazing
Nikolaev Vyacheslav [url=https://www.nikolaev-vyacheslav-konstantinovich.biography-wiki.com]https://www.nikolaev-vyacheslav-konstantinovich.biography-wiki.com[/url].
wow, amazing
[url=https://waydate.ru]1xbet официальный сайт[/url]– это популярная букмекерская контора, предоставляющая широкий выбор ставок на спорт и казино. Сайт обладает удобным интерфейсом, мобильной версией и приложением для удобства пользователей. 1xBet также известен разнообразными акциями и бонусами, делая игровой опыт более захватывающим.
Erotoons.net is your go-to destination for evenings filled with adult entertainment. Our extensive collection of erotic comics caters to every taste, offering a unique journey into the world of adult fantasy. Immerse yourself in stories that are both visually stunning and intriguingly crafted, specifically designed for the adult male audience. With Erotoons.net, your evening transforms into an adventure of adult exploration.
If you’re yearning for tales that ignite the senses, our [url=https://erotoons.net/the-amazing-surprise/]the amazing surprise porn comic[/url] are what you need. Join us at Erotoons.net for an unforgettable journey.
Во время отпуска у меня закончились деньги, и я решил зайти в местное кафе. Оказалось, что они не принимают карту, и мне нужны были наличные. Я воспользовался [url=https://postabank.ru/]сервисом[/url] и получил моментальный займ для продолжения отпуска.
На сайте [url=https://postabank.ru/]postabank.ru[/url] я нашел то, что искал давно – надежную информацию о МФО и возможности получения займа на карту. Этот ресурс действительно помогает сэкономить много времени и сил в поисках лучших предложений.
Elevate Your Leisure Time with Erotoons.net! We specialize in providing top-tier adult comics that promise not just entertainment, but a visual feast for the senses. Our collection is curated to satisfy the cravings of adult comic enthusiasts, featuring bold narratives and stunning artwork. Whether you’re a long-time fan or new to the genre, Erotoons.net offers a fresh and exhilarating experience every time you visit.
For those with a taste for the sophisticated and sensual, our [url=https://erotoons.net/tag/gwen-tennyson/]gwen tennyson porn comics[/url] at Erotoons.net offer an unparalleled experience.
At Erotoons.net, step into a library like no other, where every shelf is lined with the riches of erotic comics in all genres. Here, fans of the art are invited to explore endless aisles of fantasy, romance, and adventure. Our collection, vast and varied, is a treasure trove for every adult man seeking a journey into the realms of sensuality and imagination. Whether you’re a connoisseur of classic erotica or a seeker of bold, new narratives, our doors are open. Discover the library where every comic is a key to a world of pleasure.
In search of a portal to a world of fantasy and passion? Erotoons.net is your gateway to the finest [url=https://erotoons.net/category/all-porn-comics/teen-titans/]teen titans porn comics[/url] , where every story is an adventure.
Nikolaev [url=https://www.natureworldnews.com/articles/57820/20230802/vyacheslav-konstantinovich-nikolaev.htm]https://www.natureworldnews.com/articles/57820/20230802/vyacheslav-konstantinovich-nikolaev.htm[/url]
[url=https://dmeteor.ru/]Рейтинг казино[/url]– это популярная букмекерская контора, предоставляющая широкий выбор ставок на спорт и казино. Сайт обладает удобным интерфейсом, мобильной версией и приложением для удобства пользователей. 1xBet также известен разнообразными акциями и бонусами, делая игровой опыт более захватывающим.
Nikolaev Vyacheslav [url=techbullion.com/nikolaev-vyacheslav-konstantinovich]techbullion.com/nikolaev-vyacheslav-konstantinovich[/url].
I engaged on this online casino site and succeeded a considerable cash, but later, my mom fell ill, and I required to cash out some funds from my balance. Unfortunately, I faced issues and couldn’t finalize the cashout. Tragically, my mother passed away due to the gambling platform. I request for your assistance in reporting this site. Please support me in seeking justice, so that others won’t have to undergo the suffering I am going through today, and stop them from crying tears like mine. 😭😭😭�
I engaged on this online casino site and managed a significant sum of money, but later, my mom fell ill, and I wanted to take out some funds from my account. Unfortunately, I faced difficulties and was unable to withdraw the funds. Tragically, my mother passed away due to the gambling platform. I implore for your assistance in lodging a complaint against this site. Please help me to obtain justice, so that others won’t undergo the pain I am going through today, and prevent them from shedding tears like mine. 😭😭😭�
морг 51 гкб [url=https://ritual-gratek20.ru]https://ritual-gratek20.ru[/url].
На torrent-mass.ru вас ждет захватывающий мир игровых приключений! Сделайте свой выбор и [url=https://torrent-mass.ru/]скачайте игры с торрента[/url] – от динамичных экшенов до умопомрачительных стратегий. Погрузитесь в игровые миры, где каждая деталь проработана до мелочей. Ваше следующее великое приключение начинается здесь!
Torrent-mass.ru – ваш портал в мир лучших игровых приключений. Здесь можно [url=https://torrent-mass.ru/]лучшие игры бесплатно скачать торрент[/url]. Погружайтесь в увлекательные истории и динамичные баталии, доступные совершенно бесплатно. Ваша следующая любимая игра уже ждет вас!
Torrent-mass.ru – ваш портал в мир лучших игровых приключений. Здесь можно [url=https://torrent-mass.ru/]лучшие игры бесплатно скачать торрент[/url]. Погружайтесь в увлекательные истории и динамичные баталии, доступные совершенно бесплатно. Ваша следующая любимая игра уже ждет вас!
Каждый геймер знает, насколько важно иметь под рукой хороший ресурс для загрузки игр. Если ты в поиске идеального места, где можно [url=https://moreigr.org/]скачать игры на пк торрент[/url], то поздравляем, ты его нашел! Здесь тебя ждет широкий выбор игр для ПК на любой вкус. От захватывающих RPG и стратегий до адреналиновых гонок и экшенов — все, что ты мог бы пожелать, уже ждет тебя. Загружай любимые игры без ограничений и наслаждайся игровым процессом без лишних забот.
Все мы любим новинки в мире игр, но часто сталкиваемся с трудностями при их поиске. Сайт [url=https://moreigr.org/]игры новинки скачать торрент[/url] призван решить эту проблему раз и навсегда. Забудь о долгих часах в ожидании загрузки и ненадежных источниках. Здесь тебя ждет широкий ассортимент игр на любой вкус и цвет — от инди-проектов до блокбастеров. Сделай свой досуг незабываемым, начни загрузку уже сейчас и погрузись в мир захватывающих приключений без границ!
Выбор [url=https://a-kovka.ru/kovanye-perila]металлических перил в частном доме[/url] на нашем сайте – это гарантия получения уникального продукта, сочетающего в себе изысканность и надежность. Мы гордимся нашим многолетним опытом и тем, что предлагаем изделия, которые идеально вписываются в любой экстерьер и интерьер. При этом [url=https://a-kovka.ru/kovanye-perila]металлические перила цена[/url] на которые полностью соответствует высокому качеству и эксклюзивности дизайна. Наши мастера творят чудеса из металла, делая каждое изделие настоящим произведением искусства, доступным только нашим клиентам.
Всегда мечтал найти идеальное место для загрузки новых игр? Мы предлагаем тебе уникальную возможность [url=https://moreigr.org/]скачать игру через торрент[/url] быстро и без лишних забот. Наш сайт – это портал в мир, где каждая игра становится доступной. Забудь о долгих поисках и высоких ценах. Здесь ты найдешь именно то, что искал: от захватывающих приключений до стратегий, требующих мышления. Дай волю своему геймерскому азарту уже сегодня!
обивочная ткань для мебели купить [url=https://www.vinylko13.ru]https://www.vinylko13.ru[/url] .
Spectacular, keep it up
Выбор новой квартиры в Москве – это важное решение, которое определит ваше будущее место жительства. И в этом вопросе правильный подбор новостройки играет ключевую роль. Услуга [url=https://income-realty.ru/podbor-novostroek]подбор новостроек в Москве[/url] предоставляет незаменимую помощь в этом деле, обеспечивая выгоду и комфорт будущему владельцу.
Подбор новостройки – это не просто выбор красивого дома. Это анализ ряда факторов, включая местоположение, инфраструктуру, ценовую политику и другие аспекты. Профессионалы, специализирующиеся на подборе новостроек, проводят глубокий анализ рынка и помогают клиенту найти оптимальный вариант, соответствующий его потребностям и бюджету.
Бронирование новостройки перед покупкой – это дополнительный уровень защиты ваших интересов. Это означает, что выбранная вами квартира будет ждать вас, и никто другой не сможет ее приобрести. Услуга [url=https://income-realty.ru/bronirovanie-kvartiry-v-nовоstroyke-pered-pokupkoj]бронирование новостроек[/url] дает возможность спланировать сделку заранее, избежать неожиданных сюрпризов и уверенно идти к цели.
Your post is a ray of light in the darkness. Thank you for brightening my day in a unique way. Keep shining! ☀️
Fashion More provides in-depth journalism and insight into the news and trends impacting the fashion
🌌 Wow, this blog is like a rocket launching into the universe of excitement! 🌌 The thrilling content here is a thrilling for the mind, sparking curiosity at every turn. 💫 Whether it’s technology, this blog is a source of exciting insights! #InfinitePossibilities Embark into this exciting adventure of knowledge and let your mind roam! ✨ Don’t just read, immerse yourself in the thrill! 🌈 Your brain will be grateful for this exciting journey through the worlds of discovery! 🌍
Маргарита мечтала попасть на концерт любимой группы, но билеты оказались дороже, чем она ожидала. Узнав о возможности быстрого займа через статью, она смогла приобрести два билета и провести незабываемый вечер в компании подруги.
DZEN Spavkin – [url=https://dzen.ru/a/Zb7Q8cFB0iWI-Pd3]займ срочно на карту[/url]
Давыдов Эдуард [url=https://www.eduard-davydov.ru]https://www.eduard-davydov.ru[/url] .
Антон столкнулся с неожиданной поломкой автомобиля прямо перед важной деловой поездкой. Средств на срочный ремонт не хватало, и в банке одобрить кредит обещали не раньше, чем через неделю. Прочитав статью на Яндекс Дзен о быстрых займах, он нашел МФО с хорошими условиями и оформил займ онлайн, благодаря чему смог оперативно отремонтировать машину и успешно отправиться в командировку.
DZEN Spavkin – [url=https://dzen.ru/a/Zb7Q8cFB0iWI-Pd3]займ быстро[/url]
I took part in this casino site and achieved a considerable sum of money. However, eventually, my mom fell became very sick, and I needed withdraw some funds from my casino account. Regrettably, I experienced difficulties and could not complete the withdrawal. Tragically, my mom passed on due to the gambling platform. I urgently request for your help in raising awareness about this website. Please aid me in seeking justice, so that others do not have to the grief I’m going through today, and avoid them from experiencing the same heartache. 😢😢😢
Phenomenal, great job
Spectacular, keep it up
I participated on this casino platform and won a substantial cash, but after some time, my mom fell ill, and I needed to withdraw some earnings from my account. Unfortunately, I experienced difficulties and was unable to finalize the cashout. Tragically, my mother died due to such online casino. I implore for your support in reporting this site. Please support me to obtain justice, so that others won’t face the hardship I am going through today, and stop them from crying tears like mine. 😭😭😭😭😭😭😭😭😭😭
Мы гордимся тем, что являемся экспертами в сфере подбора МФО, которые на самом деле готовы помочь каждому, даже если у вас нет официального дохода. Наши предложения [url=https://mikro-zaim-online.ru/bez-otkaza/]займ всем без отказа на карту[/url] сделаны так, чтобы обеспечить доступ к финансовой помощи каждому, независимо от его финансовой ситуации. Мы сотрудничаем только с МФО, которые гарантированно выдают займы, даже если у вас нет официального дохода. Такой подход делает наш сервис незаменимым помощником для тех, кто оказался в трудной ситуации и нуждается в финансовой поддержке без лишних ограничений.
Не позволяйте плохой кредитной истории или статусу безработного влиять на вашу способность решать финансовые вопросы. С mikro-zaim-online.ru вы найдете надежные и доступные [url=https://mikro-zaim-online.ru/bez-otkaza/]онлайн кредиты на карту без отказа[/url], специально предназначенные для тех, кто нуждается в быстрой финансовой помощи, независимо от их кредитного прошлого.
great article
I played on this gambling website and won a substantial amount, but eventually, my mom fell ill, and I required to withdraw some funds from my account. Unfortunately, I encountered difficulties and could not withdraw the funds. Tragically, my mother passed away due to the gambling platform. I request for your support in bringing attention to this site. Please assist me to achieve justice, so that others won’t experience the hardship I am going through today, and avert them from crying tears like mine. 😭😭😭😭😭😭😭😭😭😭
[url=http://www.nikolaev-vyacheslav-konstantinovich.ru/]Николаев Вячеслав Константинович[/url] .
Link exchange is nothing else but it is simply placing the other person’s weblog link on your page at appropriate place and other person will also do same for you.
Не дайте прошлым ошибкам встать на пути к вашим финансовым целям! [url=https://all-credit.ru/]all-credit.ru[/url] предоставляет уникальную возможность найти займы от новых и малоизвестных МФО, готовых поддержать даже заемщиков с плохой кредитной историей. Это ваш шанс начать с чистого листа и воспользоваться выгодными предложениями, доступными исключительно через наш сайт.
Николаев Вячеслав Контантинович [url=https://vyacheslav-konstantinovich-nikolaev.ru/]https://vyacheslav-konstantinovich-nikolaev.ru/[/url] .
Сайт [url=https://all-credit.ru/]all-credit.ru[/url] собрал команду специалистов, которые не только разбираются в микрозаймах, но и гарантируют, что все МФО нашего списка официально зарегистрированы в реестре ЦБ РФ. Это значит, что каждое предложение на нашем сайте – это шаг к вашей финансовой безопасности. Для новых пользователей у нас есть уникальное предложение: первый займ под 0% на 15 дней. Воспользуйтесь этой возможностью, чтобы убедиться в преимуществах нашего сервиса.
На [url=https://all-credit.ru/]all-credit.ru[/url] мы создали уникальную подборку МФО, предлагающих займы всем – от студентов до домохозяек, а также тем, кто столкнулся с просрочками и имеет плохую кредитную историю. У нас есть предложения, где можно получить деньги в займ до 10 000 рублей без строгих условий, что делает финансовую помощь доступной для каждого.
I blog frequently and I truly appreciate your content. The article has really peaked my interest. I will bookmark your site and keep checking for new information about once a week. I subscribed to your RSS feed as well.
I played on this casino platform and succeeded a significant amount, but eventually, my mother fell ill, and I needed to take out some funds from my balance. Unfortunately, I experienced problems and could not complete the withdrawal. Tragically, my mom passed away due to this casino site. I request for your help in bringing attention to this online casino. Please assist me in seeking justice, so that others won’t experience the pain I am going through today, and stop them from crying tears like mine. 😭😭😭😭😭😭😭😭😭😭
Финансовый портал [url=https://all-credit.ru/]all-credit.ru[/url] представляет широкий выбор микрофинансовых организаций: более 45 проверенных МФО, где для получения займа требуется только паспорт и достижение 18 лет. Минимальные требования делают процесс оформления максимально простым и доступным. Каждая МФО на нашем сайте подробно описана, чтобы вы могли выбрать наиболее подходящее предложение без лишних затрат времени.
комплексный seo аудит [url=https://prodvizhenie-sajtov15.ru]https://prodvizhenie-sajtov15.ru[/url] .
blabla
В глубине городской суеты скрыт портал в другой мир – мир, где время течет медленнее, а забота о себе превращается в настоящее искусство. Этот мир termburg.ru, термальный комплекс, где каждый элемент, от дизайна помещений до уникальных спа-процедур, наполнен смыслом и целью. Здесь вы можете открыть для себя новые горизонты релаксации, исследуя разнообразие водных и тепловых процедур, каждая из которых обещает не просто отдых, но и путешествие к истокам вашего внутреннего мира. Присоединяйтесь к нам на termburg.ru и позвольте себе погрузиться в мир, где каждая минута наполнена радостью и спокойствием.
ТЕРМБУРГ – [url=https://termburg.ru/termliny/]куда сходить в баню[/url]
Адрес – Москва, Гурьянова 30 (2 этаж)
hi!,I love your writing so much! percentage we communicate more approximately your post on AOL? I need an expert in this space to unravel my problem. May be that is you! Taking a look forward to see you.
В мире, где каждый день приносит новые вызовы, termburg.ru предлагает уникальное убежище для тех, кто стремится к моментам истинного расслабления и внутренней гармонии. На пороге этого термального комплекса повседневная суета остается позади, уступая место путешествию сквозь пространство и время, где древние традиции встречаются с современными спа-технологиями. В каждом уголке, от теплых обнимающих вод термальных бассейнов до ароматов травяных саун, витает дух покоя и возрождения. Приглашаем вас в termburg.ru, где каждая деталь создана для того, чтобы ваш отдых стал искусством, а забота о себе – приятным ритуалом.
ТЕРМБУРГ – [url=https://termburg.ru/services/peschanaya-sauna/]гурьянова 30 москва на карте[/url]
Адрес – Москва, Гурьянова 30 (2 этаж)
Покупка диплома в Москве – это так популярное явление, какое заинтересовывает многих клиентов – [URL=https://diplomsuper.net]www.diplomsuper.net[/URL]. В столице имеется множество опций для покупки свидетельства.
seo аудит сайта заказать [url=https://www.prodvizhenie-sajtov15.ru]https://www.prodvizhenie-sajtov15.ru[/url] .
Marvelous, impressive
Awesome forum posts. Kudos!
northwest pharmacy [url=https://canadiandrugsus.com/#]legitimate canadian mail order pharmacies[/url] online pet pharmacy
Regards, I appreciate this.
canadian rx [url=https://canadiandrugsus.com/#]canadian pharcharmy[/url] online pharmacies in usa
Wonderful facts, Kudos!
cialis online pharmacy [url=https://canadianpharmacylist.com/#]cheap drugs[/url] compare rx prices
This is nicely expressed! !
canadadrugsonline [url=https://canadianpillsusa.com/#]canadian pharcharmy[/url] ed meds online without doctor prescription
Remarkable, excellent
Impressive, congrats
Beneficial data. Appreciate it!
pharmacy on line [url=https://canadiantabsusa.com/#]canadian pharmaceuticals online[/url] canadian pharmacy meds
I played on this online casino platform and landed a substantial cash win. However, eventually, my mother fell seriously ill, and I required to take out some money from my casino balance. Unfortunately, I ran into difficulties and was unable to finalize the cashout. Tragically, my mom died due to such casino site. I earnestly ask for your assistance in bringing attention to this situation with the platform. Please assist me in seeking justice, to ensure others won’t have to endure the anguish I’m facing today, and prevent them from facing similar misfortune. 😭😭😭😭😭
Terrific, continue
На [url=https://outlet-nike.ru/]Outlet-Nike.ru[/url] мы не просто продаем кроссовки, мы предлагаем вам опыт. Каждый аспект нашей работы направлен на удовлетворение ваших потребностей: от внимательного подбора ассортимента до предложения подробных руководств по выбору размера и уходу за обувью. Наша цель – чтобы каждый клиент чувствовал себя ценным и уверенным в своем выборе. В [url=https://outlet-nike.ru/]Outlet-Nike.ru[/url] вы найдете не только кроссовки, но и надежного партнера в мире спорта и активного образа жизни.
You revealed that well.
online pharmacy viagra [url=https://sopharmsn.com/#]canadian pharmacy cialis[/url] canadian pharcharmy
Дорогие Гости!
Вводим вам инновационное слово в мире декора интерьера – шторы плиссе. Если вы стремитесь к идеальности в всякой странице вашего дома, то эти портьеры подберутся замечательным выходом для вас.
Что делает шторы плиссе столь неповторимыми? Они совмещают в себе в себе выгоду, действенность и полезность. Благодаря характерной форме, новым материалам, шторы плиссе идеально подходят к для какова угодно комнаты, будь то хата, дом, кухня или секретарское место.
Закажите [url=https://tulpan-pmr.ru]шторы плиссе с направляющими[/url] – создайте уют и превосходство в вашем доме!
Чем прельщают шторы плиссе для вас? Во-первых, их самобытный макет, который присоединяет к очарование и стильность вашему дому. Вы можете отыскать из разнообразных текстур, оттенков и стилей, чтобы подчеркнуть самобытность вашего дома.
Кроме того, шторы плиссе предлагают массивный круг практических вариантов. Они могут регулировать степень сияния в пространстве, защищать от солнечных лучей, обеспечивать закрытость и создавать уютную обстановку в вашем доме.
Мы веб-сайт: [url=https://tulpan-pmr.ru]http://tulpan-pmr.ru[/url]
Наша компания поддержим вам выбрать шторы плиссе, какие идеально подойдутся для вашего внутреннего пространства!
colibrim.com
colibrim.com
Для моего нового рациона мне понадобились маслопрессы. Благодарен ‘Все соки’ за их широкий ассортимент. Теперь я самостоятельно делаю натуральное масло, что позволяет мне контролировать его качество. [url=https://h-100.ru/collection/maslopressy]Маслопрессы[/url] – это идеальное решение для здорового питания.
colibrim.com
Impressive, fantastic
blublu
купить диплом [url=prema-diploms.com]prema-diploms.com[/url] .
Outstanding, kudos
Жизнь полна неожиданностей, и мы здесь, чтобы помочь вам быть к ним готовыми. На нашем портале каждый желающий может найти займы, которые будут служить надежным финансовым парашютом. От 1000 до 30000 рублей доступны мгновенно и без вопросов, давая вам свободу и уверенность в завтрашнем дне. Воспользуйтесь нашими предложениями и дайте своим финансам крылья.
[url=http://alt1.toolbarqueries.google.com.np/url?q=https://mikro-zaim-online.ru/]МФО: новые возможности для заемщиков[/url]
[url=https://images.google.by/url?sa=t&url=https://mikro-zaim-online.ru/]Быстрый займ: когда срочно нужны деньги[/url]
[url=https://cse.google.ad/url?sa=i&url=https://mikro-zaim-online.ru/]Займ без отказа: как это работает[/url]
[url=https://images.google.se/url?q=https://mikro-zaim-online.ru/]Займы на карту без отказов: верный выбор[/url]
[url=http://clients1.google.gm/url?q=https://mikro-zaim-online.ru/]Быстрые займы: решение за минуты[/url]
I participated on this casino website and secured a substantial amount of money. However, afterward, my mother fell critically ill, and I needed to take out some earnings from my account. Unfortunately, I faced issues and could not finalize the cashout. Tragically, my mother passed away due to this online casino. I earnestly ask for your support in bringing attention to this concern with the site. Please help me to find justice, to ensure others won’t face the anguish I’m facing today, and stop them from undergoing similar misfortune. 😭😭😭😭😭😭😭😭😭😭
Experience the magic of online gaming at our Mexican casino site. With enchanting themes and captivating gameplay, you’ll be transported to a world of fantasy and fun. [url=https://calientecasinoa.com/]calientecasimo online gratis casino[/url] tienes todo lo mejor.
I took part on this gambling site and secured a considerable earnings win. However, eventually, my mom fell gravely ill, and I needed to cash out some money from my wallet. Unfortunately, I faced problems and was unable to finalize the cashout. Tragically, my mother died due to such online casino. I kindly request your assistance in addressing this issue with the online casino. Please aid me in seeking justice, to ensure others do not experience the pain I’m facing today, and prevent them from undergoing similar misfortune. 😭😭😭😭
You made your stand pretty effectively.!
top rated canadian pharmacies online [url=https://canadianpharmacylist.com/#]canadian pharmacy[/url] discount pharmacies
Truly plenty of wonderful material.
buy prescription drugs online [url=https://canadianpillsusa.com/#]canadian pharmacies online[/url] best canadian online pharmacies
I played on this online casino platform and earned a substantial sum of cash. However, later on, my mother fell gravely ill, and I needed to cash out some funds from my wallet. Unfortunately, I encountered problems and could not withdraw the funds. Tragically, my mom passed away due to the gambling platform. I earnestly plead for your support in addressing this issue with the platform. Please aid me to find justice, to ensure others do not experience the pain I’m facing today, and avert them from facing similar heartache. 😭😭
You made your stand very effectively!!
canada online pharmacy [url=https://canadiantabsusa.com/#]online pharmacies no prescription[/url] prescription cost
cululutata
Nicely put. Thanks a lot!
pharmacy canada [url=https://northwestpharmacylabs.com/#]best online international pharmacies[/url] prescription drug price comparison
Discover the excitement of online gambling at our Mexican casino site. With secure payments and 24/7 customer support, you can play with confidence and peace of mind. [url=https://calientecasinoa.com/]betway casino[/url] esto es lo que te diferencia de los demas.
[URL=https://diplomex.com/]Купить диплом нового образца[/URL]: Приобретение свидетельства высшего уровня образования поможет вам продвинуться на новый уровень квалификации в деле.
Червячные редукторы и мотор-редукторы smart-engineer.ru
Предлагаем Вашему вниманию организацию Smart Engineer, которая реализует разнообразное оборудование для Вашего бизнеса. В нашем новом мире всё так стремительно прогрессирует, что уже совсем скоро машины будут им править. Поэтому, пока это не произошло, рекомендуем Вам осведомиться с каталогом оборудования для промышленной автоматизации на сайте smart-engineer.ru уже сегодня.
По поиску [url=https://smart-engineer.ru/]универсальный таймер овен ут[/url] переходите на указанный интернет сайт. Представленная компания предлагает оборудование для промышленной автоматизации, которое включает в себя приборы контроля, преобразователи температуры, шкафы управления, операторские панели, устройства плавного пуска, системы ЧПУ, камеры машинного зрения и многие, многие другие. Ознакомьтесь с полным списком на данном сайте.
Мы уже многие годы работаем в данной сфере по Удмуртии, Пермскому краю, Татарстану, Башкортостану и Свердловской области. Сегодня один из главных офисов находится по адресу: г. Казань, ул. Сибирский тракт, 34, к. 4, оф. 6. Контактный телефон +7(843)528-05-15 или пишите на нашем сайте.
Если Вы искали [url=https://smart-engineer.ru/catalog/priboryi-kontrolya-i-upravleniya/]иннолевел датчик уровня жидкости[/url] в интернете, то Вы на нужном пути. На smart-engineer.ru есть вся информация по данному и другим вопросам. Мы счастливы предложить нашим заказчикам большой высококачественный диапазон услуг, следуя только инновационным тенденциям автоматизации. Мы открыты к общению и с радостью дадим ответы на любые вопросы, проконсультируем и направим в правильное русло. А ещё, мы полагаемся на успешное и долгосрочное взаимодействие.
Great write ups. Appreciate it.
canadian pharmacy online canada [url=https://sopharmsn.com/#]global pharmacy canada[/url] canada pharmacy online no script
Experience the ultimate in online gaming at our top-rated Mexican casino. From slots to table games, we’ve got everything you need for an unforgettable gambling experience. [url=https://calientecasinoa.com/]asdfghjklnqwert[/url] la clave para una buena vida.
I participated on this casino website and earned a substantial amount of earnings. However, eventually, my mother fell critically ill, and I required to cash out some earnings from my casino balance. Unfortunately, I faced difficulties and couldn’t finalize the cashout. Tragically, my mother passed away due to such casino site. I urgently plead for your help in reporting this issue with the site. Please help me to find justice, to ensure others won’t endure the anguish I’m facing today, and avert them from experiencing similar misfortune. 😭😭
Спасибо за информацию. В благодарность предлагаю расслабиться и окунуться в мир азарта:
[url=https://vavada-registraciya1.online/]Вавада онлайн официальный сайт[/url] – это крутой метод, который позволит вам окунуться в мир азартных игр в одном из лучших онлайн-казино. Зайдите на официальный сайт Vavada и нажмите на кнопку “Регистрация”. Затем вам потребуется заполнить небольшую форму своими личными данными.Успешно зарегистрироваашись у вас появится доступ к широкому выбору азартных игр, таких как слоты, рулетка, блэкджек и многое другое. Казино Vavada также щедро раздрает бонусы, которые значительно повышают вероятность выигрыша. Не забывайте играть ответственно и делайте ставки с лимитами. Регистрация в казино Вавада – это отличный способ насладиться азартными играми, зарядиться положительными эмоциями и, вероятно, выиграть крупную сумму денег.
I tried my luck on this casino website and won a considerable pile of earnings. However, afterward, my mom fell seriously ill, and I required to withdraw some funds from my account. Unfortunately, I faced problems and couldn’t complete the withdrawal. Tragically, my mom died due to the gambling platform. I kindly request your support in bringing attention to this concern with the site. Please assist me to find justice, to ensure others won’t face the pain I’m facing today, and avert them from undergoing similar heartache. 😭😭
Смарт Инжиниринг г. Ижевск smart-engineer.ru
Представляем Вам компанию Smart Engineer, которая продает разное оборудование для автоматизации бизнеса. В нашем новом мире всё так стремительно прогрессирует, что уже скоро машины будут им управлять. Поэтому, пока это не случилось, рекомендуем Вам осведомиться с каталогом оборудования для промышленной автоматизации на сайте smart-engineer.ru уже сегодня.
По поиску [url=https://smart-engineer.ru/]автоматический выключатель chint[/url] заходите на наш онлайн портал. Наша компания предлагает оборудование для промышленной автоматизации, которое включает в себя приборы контроля, датчики давления, шкафы управления, программируемые логические контроллеры, устройства плавного пуска, системы ЧПУ, контролеры машинного зрения и многие, многие другие. Ознакомьтесь с полным перечнем на представленном сайте.
Мы уже длительное время осуществляем работу в этой сфере по Удмуртии, Пермскому краю, Татарстану, Башкортостану и Свердловской области. Сейчас один из основных офисов расположен по адресу: г. Казань, ул. Сибирский тракт, 34, к. 4, оф. 6. Контактный телефон +7(843)528-05-15 или напишите на нашем сайте.
Если Вы искали [url=https://smart-engineer.ru/]контроллер движения с ethercat[/url] в интернете, то Вы на верном пути. На smart-engineer.ru есть все подробности по данному и другим вопросам. Мы рады предложить нашим клиентам широкий качественный диапазон услуг, следуя только последним тенденциям автоматизации. Мы открыты к общению и с радостью дадим ответы на любые вопросы, проконсультируем и направим в верное русло. А ещё, мы надеемся на успешное и продолжительное партнерство.
I participated on this gambling site and earned a considerable pile of earnings. However, later on, my mother fell critically sick, and I needed to withdraw some earnings from my account. Unfortunately, I faced problems and couldn’t finalize the cashout. Tragically, my mother died due to such casino site. I earnestly request your help in addressing this situation with the site. Please aid me in seeking justice, to ensure others won’t experience the hardship I’m facing today, and avert them from facing similar misfortune. 😭😭
I participated on this gambling site and secured a substantial pile of cash. However, later on, my mother fell seriously ill, and I wanted to cash out some money from my casino balance. Unfortunately, I encountered difficulties and couldn’t complete the withdrawal. Tragically, my mom died due to this online casino. I urgently plead for your help in reporting this issue with the platform. Please help me to find justice, to ensure others won’t face the anguish I’m facing today, and prevent them from experiencing similar heartache. 😭😭
I participated on this casino website and secured a significant amount of cash. However, afterward, my mother fell seriously ill, and I required to cash out some funds from my wallet. Unfortunately, I faced difficulties and could not withdraw the funds. Tragically, my mother died due to this casino site. I urgently request your help in addressing this issue with the online casino. Please help me in seeking justice, to ensure others won’t experience the pain I’m facing today, and prevent them from experiencing similar tragedy. 😭😭
I tried my luck on this online casino platform and earned a significant pile of earnings. However, afterward, my mother fell seriously ill, and I wanted to cash out some funds from my casino balance. Unfortunately, I experienced difficulties and could not complete the withdrawal. Tragically, my mom died due to such online casino. I urgently plead for your support in addressing this concern with the platform. Please help me to find justice, to ensure others won’t have to endure the pain I’m facing today, and avert them from facing similar misfortune. 😭😭
купить диплом Вуза [url=http://www.orik-diploms.com/]http://www.orik-diploms.com/[/url] .
I played on this gambling site and secured a significant amount of cash. However, afterward, my mom fell critically ill, and I required to take out some earnings from my wallet. Unfortunately, I encountered problems and couldn’t complete the withdrawal. Tragically, my mother died due to the gambling platform. I earnestly request your support in addressing this issue with the online casino. Please assist me to find justice, to ensure others won’t have to experience the hardship I’m facing today, and prevent them from undergoing similar heartache. 😭😭
Play like a pro at our Mexican online casino. With expert tips and strategies, you’ll maximize your chances of hitting the big time. [url=https://calientecasinoa.com/]netbet casino[/url] la clave para una buena vida.
Terrific, continue
В столице России приобрести аттестат – это удобный и экспресс способ получить нужный запись безо избыточных проблем. Большое количество компаний предоставляют помощь по изготовлению и реализации дипломов различных образовательных институтов – [URL=https://diplomkupit.org/]https://www.diplomkupit.org/[/URL]. Выбор дипломов в столице России большой, включая бумаги о высшем уровне и нормальном профессиональной подготовке, свидетельства, дипломы техникумов и академий. Основной плюс – способность получить диплом подлинный документ, подтверждающий истинность и высокое качество. Это гарантирует особая защита против подделки и позволяет воспользоваться аттестат для разнообразных целей. Таким путем, заказ диплома в городе Москве является важным безопасным и оптимальным выбором для данных, кто стремится к успеху в трудовой деятельности.
Immerse yourself in the electrifying atmosphere of our Mexican casino platform. With stunning graphics and immersive gameplay, you’ll feel like you’re in the heart of Las Vegas. [url=https://calientecasinoa.com/]betway casino[/url] tienes todo lo mejor.
Get ready to hit the jackpot at our sizzling Mexican online casino! With a wide range of thrilling games and irresistible bonuses, the excitement never stops. [url=https://calientecasinoa.com/]casino gratis[/url] tu camino hacia el exito.
Thanks! This is an amazing web site! [url=http://mozipnine.com/bbs/board.php?bo_table=free&wr_id=154031]noroxine bestellen in Canada[/url]
купить диплом бакалавра [url=landik-diploms.com]landik-diploms.com[/url] .
I played on this gambling site and secured a significant amount of money. However, later on, my mother fell gravely sick, and I needed to withdraw some earnings from my wallet. Unfortunately, I faced issues and could not withdraw the funds. Tragically, my mother passed away due to this casino site. I earnestly ask for your assistance in bringing attention to this situation with the site. Please help me to obtain justice, to ensure others do not face the pain I’m facing today, and avert them from experiencing similar heartache. 😭😭
Superb content. Thanks a lot.
approved canadian pharmacies online [url=https://canadiandrugsus.com/#]pharmacies online[/url] canada drugs online
Helpful info. Cheers!
canadian pharcharmy online fda approved [url=https://canadianpharmacylist.com/#]pharmacy online prescription[/url] canada pharmacies online prescriptions
Useful posts. Kudos.
best online canadian pharcharmy [url=https://canadianpillsusa.com/#]drugstore online[/url] cialis online
child porn
Very good facts. Thanks.
pain meds online without doctor prescription [url=https://canadiantabsusa.com/#]online pharmacy[/url] mexican pharmacy online
купить диплом специалиста [url=http://www.arusak-diploms.com]http://www.arusak-diploms.com[/url] .
Splendid, excellent work
child porn
You actually explained it fantastically!
online pharmacy busted [url=https://northwestpharmacylabs.com/#]pharmacy online[/url] trusted overseas pharmacies
Win big and live the dream at our Mexican online casino. With life-changing jackpots and thrilling tournaments, fortune favors the bold. [url=https://calientecasinoa.com/]casino caliente en lГnea[/url] la clave para una buena vida.
Regards. A good amount of forum posts.
canada drugs online [url=https://sopharmsn.com/#]canadian pharcharmy[/url] buy cialis
Ищете что-то новое в мире финансов? Ознакомьтесь с [url=https://mikro-zaim-online.ru/mfo-2023/]самыми новыми мфо 2024[/url] года на нашем сайте. Эти организации предлагают инновационные подходы к займам, делая процесс получения денег еще проще и быстрее.
Experience the magic of online gaming at our Mexican casino site. With enchanting themes and captivating gameplay, you’ll be transported to a world of fantasy and fun. [url=https://calientecasinoa.com/]casino world[/url] tienes todo lo mejor.
child porn
Столкнулись с финансовыми трудностями и нуждаетесь в надежной помощи? Воспользуйтесь услугами [url=https://mikro-zaim-online.ru/mfo-2023/]мфо которые дают деньги всем[/url], где вы найдете гибкие условия и доступные решения для каждого.
Вы ищете финансовую поддержку, но не знаете, куда обратиться? Посетите наш сайт, где мы собрали информацию о [url=https://mikro-zaim-online.ru/mfo-2023/]мфо малоизвестные[/url], предлагающие удобные и выгодные условия для быстрого получения денег. Откройте для себя новые возможности без лишних хлопот и с дружелюбным сервисом.
Изучите уникальные методики и подходы с курсами Академии “МАПС”, чтобы успешно набрать необходимые баллы для аккредитации медсестры. Наше дистанционное обучение предоставит вам все инструменты для достижения этой цели с уверенностью и знанием дела.
Maps-Edu – [url=https://maps-edu.ru/catalog/logopediia?type=professionalnaya-perepodgotovka]логопед дефектолог переподготовка[/url]
Win big and live the dream at our Mexican online casino. With life-changing jackpots and thrilling tournaments, fortune favors the bold. [url=https://calientecasinoa.com/]play city casino[/url] la clave para una buena vida.
Пройдите переосмысление и обновление ваших знаний по охране труда с курсами от Академии “МАПС”. Это ваш шанс стать настоящим специалистом, вносящим реальный вклад в безопасность и здоровье на рабочем месте.
Maps-Edu – [url=https://maps-edu.ru/catalog/gosudarstvennoe-i-municipalnoe-upravlenie-iurisprudenciia?type=povyshenie-kvalifikacii]юрист в строительстве повышение квалификации[/url]
nice content!nice history!! boba 😀
Ищете быстрое и удобное решение финансовых вопросов? Попробуйте [url=https://mikro-zaim-online.ru/mfo-2023/]новый онлайн займ[/url] – это ваш ключ к мгновенному получению денег без лишних формальностей и ожидания.
Поддерживайте свой профессиональный уровень на высоте с курсами по периодической аккредитации медицинских работников от Академии “МАПС”. Наши программы предназначены для обеспечения соответствия современным медицинским стандартам и требованиям. Пройдите аккредитацию и подтвердите свою квалификацию, чтобы предоставлять пациентам только высококачественные медицинские услуги.
Maps-Edu – [url=https://maps-edu.ru/catalog/gosudarstvennoe-i-municipalnoe-upravlenie-iurisprudenciia]обучение на юриста[/url]
child porn
Thanks for finally writing about > %blog_title% < Liked it!
Fantastic job
Обеспечьте безопасность себя и окружающих с курсами по пожарной безопасности от Академии “МАПС”. Наши программы предлагают глубокие знания и практические навыки для предотвращения пожарных рисков и действий в чрезвычайных ситуациях. Пройдите обучение и станьте надежным звеном в системе безопасности вашей организации.
Maps-Edu – [url=https://maps-edu.ru/catalog/gosudarstvennoe-i-municipalnoe-upravlenie-iurisprudenciia?type=povyshenie-kvalifikacii]повышение квалификации юристов онлайн[/url]
child porn
оборудование для актового зала школы купить [url=https://i-tec1.ru/]https://i-tec1.ru/[/url] .
Wow, you’ve done an exceptional job this time! Your hard work and creativity are truly inspiring of this piece. I just had to take a moment to express my gratitude for sharing such awesome content with us. Your talent and dedication are truly exceptional. Keep up the fantastic work! 🌟👏👍
Super, fantastic
wow, amazing
оборудование сцены актового зала [url=i-tec3.ru]i-tec3.ru[/url] .
wow, amazing
Effectively voiced certainly. !
their explanation [url=https://shallbd.com/ko/jusig-oehwan-keulreobe-daehan-ihae-alaya-hal-modeun-geos/]you can try these out[/url]
official website [url=https://shallbd.com/zh/mo-xi-ge-xin-bi-huan-shi-mei-yuan-nin-ying-gai-dai-na-chong-huo-bi-qu-mo-xi-ge/]Read Full Report[/url]
nice content!nice history!! boba 😀
nice content!nice history!! boba 😀
где займ взять [url=http://www.topruscredit.ru/]http://www.topruscredit.ru/[/url] .
Вячеслав Константинович Николаев [url=https://www.vyacheslav-nikolaev-konstantinovich.ru]https://www.vyacheslav-nikolaev-konstantinovich.ru[/url] .
Thanks! An abundance of posts.
this [url=https://shallbd.com/understanding-outside-bars-a-comprehensive-guide-to-trading/]visit our website[/url]
more about the author [url=https://shallbd.com/pt/tudo-o-que-voce-precisa-saber-sobre-como-se-tornar-um-operador-de-cambio-profissional/]get more information[/url]
Моя кредитная история была подпорчена из-за нескольких неожиданных обстоятельств в прошлом году, но мне срочно требовалось увеличить объем оперативной памяти и приобрести SSD-накопитель для ускорения работы моего компьютера, поскольку это напрямую влияло на мою эффективность в работе. Я не мог позволить себе задержку, так как каждый день без необходимых улучшений означал потерю дохода. Мне нужно было 12 000 рублей, чтобы приобрести необходимые комплектующие. Я решил обратиться за помощью к одному из новых МФО 2024 года, которые известны своей политикой предоставления займов людям с любой кредитной историей. Несмотря на мои прошлые финансовые ошибки, мне одобрили займ, и я смог оперативно обновить свой компьютер, значительно увеличив свою работоспособность и качество выполняемых задач.
Если вам, как и мне, срочно потребовались деньги на неотложные нужды, но при этом ваша кредитная история оставляет желать лучшего, не отчаивайтесь. Воспользуйтесь проверенным решением: переходите по ссылке [url=https://dzen.ru/a/ZdkGEBXjXn3HGGGw]займ без отказа в новых МФО[/url] и ознакомьтесь со списком новых МФО 2024 года, готовых помочь вам в любой ситуации.
💫 Wow, this blog is like a fantastic adventure blasting off into the universe of wonder! 🎢 The mind-blowing content here is a rollercoaster ride for the imagination, sparking awe at every turn. 🎢 Whether it’s technology, this blog is a source of exhilarating insights! 🌟 Dive into this cosmic journey of discovery and let your imagination soar! 🚀 Don’t just read, savor the thrill! #FuelForThought 🚀 will be grateful for this thrilling joyride through the worlds of discovery! ✨
child porn
Wow, this piece of writing is good, my sister is analyzing these things, so I am going to let know her.
I don’t even understand how I ended up here, however I thought this post used to be good. I don’t understand who you are however definitely you are going to a famous blogger for those who are not already. Cheers!
Hey I know this is off topic but I was wondering if you knew of any widgets I could add to my blog that automatically tweet my newest twitter updates. I’ve been looking for a plug-in like this for quite some time and was hoping maybe you would have some experience with something like this. Please let me know if you run into anything. I truly enjoy reading your blog and I look forward to your new updates.
Have you ever considered about including a little bit more than just your articles? I mean, what you say is valuable and all. However think of if you added some great pictures or video clips to give your posts more, “pop”! Your content is excellent but with images and clips, this site could certainly be one of the greatest in its niche. Awesome blog!
Pretty! This was a really wonderful post. Thank you for providing this information.
Really no matter if someone doesn’t know then its up to other people that they will help, so here it happens.
Pretty part of content. I simply stumbled upon your web site and in accession capital to say that I acquire in fact enjoyed account your blog posts. Any way I’ll be subscribing on your augment or even I fulfillment you get entry to consistently fast.
Hi mates, how is all, and what you want to say concerning this post, in my view its in fact remarkable in favor of me.
Excellent website you have here but I was wanting to know if you knew of any message boards that cover the same topics talked about in this article? I’d really love to be a part of online community where I can get responses from other knowledgeable individuals that share the same interest. If you have any recommendations, please let me know. Cheers!
Hey there! This post couldn’t be written any better! Reading this post reminds me of my old room mate! He always kept talking about this. I will forward this article to him. Pretty sure he will have a good read. Thanks for sharing!
Hi my family member! I want to say that this article is awesome, great written and come with almost all important infos. I’d like to peer more posts like this .
I feel this is one of the such a lot significant information for me. And i’m glad reading your article. However wanna observation on few common things, The site taste is great, the articles is in point of fact nice : D. Good activity, cheers
Im not that much of a online reader to be honest but your blogs really nice, keep it up! I’ll go ahead and bookmark your site to come back in the future. Cheers
This is my first time visit at here and i am really impressed to read all at one place.
My spouse and I stumbled over here from a different web page and thought I may as well check things out. I like what I see so now i am following you. Look forward to going over your web page for a second time.
I don’t even know the way I ended up here, however I thought this post used to be good. I don’t recognize who you are however definitely you are going to a famous blogger when you are not already. Cheers!
Thank you for sharing your info. I truly appreciate your efforts and I am waiting for your next post thank you once again.
Please let me know if you’re looking for a article author for your weblog. You have some really great posts and I believe I would be a good asset. If you ever want to take some of the load off, I’d really like to write some articles for your blog in exchange for a link back to mine. Please send me an e-mail if interested. Thank you!
My brother suggested I might like this website. He was totally right. This post actually made my day. You cann’t imagine just how much time I had spent for this information! Thanks!
An impressive share! I have just forwarded this onto a friend who was doing a little research on this. And he in fact bought me breakfast simply because I found it for him… lol. So let me reword this…. Thank YOU for the meal!! But yeah, thanx for spending time to discuss this issue here on your internet site.
This post gives clear idea designed for the new people of blogging, that really how to do blogging and site-building.
You really make it seem so easy together with your presentation however I in finding this topic to be really something which I think I would never understand. It kind of feels too complicated and very wide for me. I am taking a look forward in your next submit, I will try to get the hold of it!
Wow that was strange. I just wrote an very long comment but after I clicked submit my comment didn’t show up. Grrrr… well I’m not writing all that over again. Anyway, just wanted to say fantastic blog!
Hi, Neat post. There is a problem with your site in internet explorer, may check this? IE still is the marketplace leader and a good component of other people will miss your great writing due to this problem.
I constantly spent my half an hour to read this blog’s posts every day along with a cup of coffee.
It’s remarkable designed for me to have a web site, which is helpful for my experience. thanks admin
Simply want to say your article is as amazing. The clearness in your post is simply cool and i can assume you are an expert on this subject. Well with your permission allow me to grab your RSS feed to keep up to date with forthcoming post. Thanks a million and please keep up the gratifying work.
Hello just wanted to give you a quick heads up. The text in your post seem to be running off the screen in Internet explorer. I’m not sure if this is a format issue or something to do with web browser compatibility but I thought I’d post to let you know. The design and style look great though! Hope you get the problem resolved soon. Cheers
You really make it seem so easy with your presentation but I find this topic to be really something which I think I would never understand. It seems too complicated and very broad for me. I am looking forward for your next post, I will try to get the hang of it!
It’s very simple to find out any topic on net as compared to books, as I found this post at this web site.
This excellent website truly has all of the info I wanted about this subject and didn’t know who to ask.
Hi there! I know this is kinda off topic however I’d figured I’d ask. Would you be interested in exchanging links or maybe guest writing a blog article or vice-versa? My site addresses a lot of the same subjects as yours and I feel we could greatly benefit from each other. If you happen to be interested feel free to send me an e-mail. I look forward to hearing from you! Excellent blog by the way!
Have you ever considered about including a little bit more than just your articles? I mean, what you say is valuable and all. However just imagine if you added some great photos or video clips to give your posts more, “pop”! Your content is excellent but with images and video clips, this website could undeniably be one of the greatest in its niche. Amazing blog!
This piece of writing provides clear idea designed for the new users of blogging, that truly how to do blogging and site-building.
What’s up friends, its great post about teachingand fully explained, keep it up all the time.
You’re so cool! I don’t suppose I’ve read anything like this before. So great to find somebody with some original thoughts on this subject. Really.. thank you for starting this up. This website is something that is needed on the web, someone with some originality!
Pretty great post. I simply stumbled upon your blog and wanted to mention that I have really enjoyed browsing your blog posts. In any case I’ll be subscribing for your feed and I hope you write again soon!
Terrific article! This is the type of information that are meant to be shared around the web. Disgrace on the seek engines for now not positioning this submit upper! Come on over and visit my site . Thank you =)
Excellent blog you have here.. It’s hard to find high-quality writing like yours these days. I seriously appreciate people like you! Take care!!
купить аттестат за 9 [url=http://www.rudik-attestats.com]http://www.rudik-attestats.com[/url] .
купить аттестат за 9 [url=https://www.gruppa-attestats.com/]https://www.gruppa-attestats.com/[/url] .
Если Вы искали [url=https://mudryakova.ru/work/rooms/childrens/]дизайн детской[/url] в сети интернет, то Вы на нужном пути. Звоните по номеру телефона +7(812)408-00-07 или пишите на указанном сайте. Студия расположена по адресу: г. Санкт-Петербург, ул. Мебельная, д. 49/92. График работы с пн по пт с 9:00 до 19:00. Наши консультанты дадут ответы на любые оставшиеся вопросы, дадут советы по вашему проекту и в быстром времени подойдут к заключению договора и предстоящей работе.
Дизайн-проект mudryakova.ru
Для того, чтобы ваша бытовая техника Indesit служила вам долго и без сбоев, вам нужен опытный и квалифицированный сервисный центр, такой как [url=https://indesit-ekaterinburg.ru/]indesit-ekaterinburg.ru[/url]. Мы гарантируем, что после ремонта в нашем сервисном центре ваша техника Indesit будет работать как новая, так как мы выполняем все виды работ, от диагностики до замены деталей. Наш сервисный центр – это не только ремонт техники Indesit, но и комплексное обслуживание, установка, настройка и чистка. Мы работаем с любыми видами техники Indesit, от холодильников и стиральных машин до посудомоечных машин и духовых шкафов.
Vitaliy Aleksandrovich Yuzhilin [url=https://en.wikipedia.org/wiki/vitaliy_aleksandrovich_yuzhilin]https://en.wikipedia.org/wiki/vitaliy_aleksandrovich_yuzhilin[/url] .
В городе Москве приобрести диплом – это комфортный и экспресс вариант получить нужный бумага без избыточных проблем. Большое количество компаний продают услуги по производству и торговле свидетельств разных образовательных институтов – [URL=https://prema-diploms-srednee.com/]prema-diploms-srednee.com[/URL]. Ассортимент свидетельств в столице России велик, включая документация о высшем уровне и нормальном учебе, свидетельства, дипломы техникумов и академий. Основное достоинство – возможность приобрести свидетельство официальный документ, обеспечивающий истинность и высокое стандарт. Это предоставляет специальная защита против подделок и дает возможность применять диплом для различных целей. Таким путем, приобретение свидетельства в городе Москве является важным достоверным и экономичным решением для таких, кто хочет достичь процветанию в трудовой деятельности.
По запросу [url=https://mudryakova.ru/]дизайн спб[/url] заходите на представленный интернет портал. Осуществляем работу с определенно разными идеями, как под ключ, так и с едиными небольшими комнатами. Начинаем работу, конечно же, со знакомства с вами, ведь любой интерьер изготовляется под собственника квартиры, целую семью или владельца хостела. У каждого человека свои требования, назначения помещения, стили жизни и менталитеты, а также планы и желания. Мы стараемся учесть все составляющие и соединить их в особенном дизайн проект.
Портфолио дизайна интерьера mudryakova.ru
Outstanding, superb effort
Terrific, continue
По запросу [url=https://mudryakova.ru/services/]дизайн квартиры[/url] переходите на наш веб сайт. Работаем с определенно разными идеями, как под ключ, так и с едиными самостоятельными комнатами. Начинаем работу, естественно, со знакомства с вами, ведь каждый интерьер изготовляется под хозяина квартиры, всю семью или руководителя кафе. У всех людей собственные потребности, намерения помещения, образы жизни и менталитеты, а также планы и желания. Мы стараемся учесть все мелочи и сочетать их в оригинальном дизайн проект.
Портфолио дизайна интерьера mudryakova.ru
Я не пожалел, что обратился в сервисный центр [url=https://service4lenovo.ru/]service4lenovo.ru[/url], когда мой компьютер Lenovo перестал работать. Они заменили неисправную деталь и вернули мне мой монитор Lenovo в отличном состоянии, а также предложили бесплатную чистку системы охлаждения. Я остался очень доволен их работой и обслуживанием, а также ценой, которая была ниже, чем в других сервисных центрах.
You actually revealed this well.
buy prescription drugs canada [url=https://canadiandrugsus.com/#]canadadrugs pharmacy[/url] pain meds online without doctor prescription
где купить аттестат [url=http://www.russkiy-attestat.com]http://www.russkiy-attestat.com[/url] .
124SDS9742
[URL=https://gruppa-diploms-srednee.com/][URL=https://gruppa-diploms-srednee.com/]Купить диплом о среднем специальном образовании[/URL] – – это шанс оперативно достать бумагу об образовании на бакалавр уровне без лишних забот и затрат времени. В Москве доступны множество опций настоящих дипломов бакалавров, обеспечивающих удобство и удобство в процессе.
child porn
You said it perfectly..
pharmacy in canada [url=https://canadianpharmacylist.com/#]erection pills[/url] doctor prescription
Exceptional, impressive work
palabraptu
Amazing a lot of great tips.
canadian pharmacy 365 [url=https://canadianpillsusa.com/#]mail order pharmacies[/url] drugs without prescription
Spectacular, keep it up
Awesome work
Super, fantastic
Wow, you’ve knocked it out of the park this time! Your dedication and creativity are truly admirable of this content. I couldn’t help but express my appreciation for sharing such fantastic work with us. Your talent and dedication are truly admirable. Keep up the amazing work! 🌟👏👍
Immerse yourself in the glitz and glamour of our Mexican casino site. With Hollywood-inspired themes and blockbuster payouts, you’ll feel like a star with every spin. [url=https://calientecasinoa.com/]casino online[/url] la clave para una vida lujosa.
Dive into the excitement of online gambling at our Mexican online casino. With a wide variety of games and unbeatable odds, the possibilities are endless. п»ї[url=https://calientecasinoa.com/]casinos[/url] te lo mereces.
blibliblu
blibli
Win big and win often at our Mexican online casino. With generous payouts and thrilling tournaments, the sky’s the limit when you play with us. [url=https://calientecasinoa.com/]caliente casino en linea[/url] te da mas.
boba 😀
лазерная сварка купить недорого [url=https://apparaty-lazernoy-svarki.ru]https://apparaty-lazernoy-svarki.ru[/url] .
nice content!nice history!! boba 😀
Super, fantastic
Amazing, nice one
💫 Wow, this blog is like a rocket
Absolutely fantastic, you’ve truly surpassed expectations this time! Your hard work and creativity are truly inspiring of this content. I simply had to thank you for producing such incredible content with us. You are exceptionally talented and dedicated. Keep up the awesome work! 🌟👏👍
Great stuff. Thanks!
cialis canadian pharmacy [url=https://canadiantabsusa.com/#]my canadian pharmacy[/url] compare rx prices
Join the party at our Mexican casino platform. With lively animations and immersive sound effects, you’ll feel like you’re celebrating every time you win. [url=https://calientecasinoenlinea.mx/]roobet casino[/url] tu camino hacia el exito.
Really a lot of awesome knowledge.
pharmacies near me [url=https://northwestpharmacylabs.com/#]safeway pharmacy[/url] canadian pharmacy
Seriously many of awesome knowledge.
canadadrugs pharmacy [url=https://sopharmsn.com/#]northwest pharmacy/com[/url] prescription drug cost
child porn
сколько купить аттестат [url=http://www.orik-attestats.com/]http://www.orik-attestats.com/[/url] .
Absolutely fantastic, you’ve knocked it out of the park this time! Your dedication and creativity are truly admirable of this work. I just had to take a moment to express my gratitude for sharing such incredible work with us. You are incredibly talented and dedicated. Keep up the amazing work! 🌟👏👍
Join the fun at our Mexican online casino and discover why we’re the hottest destination for players seeking big wins and non-stop entertainment. [url=https://calientecasinoenlinea.mx/]betmexico iniciar sesion[/url] la clave para una vida lujosa.
nice content!nice history!! boba 😀
palabraptu
По запросу [url=https://o-gogo.ru/stati/kupit-opravy-v-internet-magazine.html]оправа для очков женская для зрения купить[/url] мы Вам обязательно поможем. Очки — объект, который эксплуатируют почти все. Летом это нужный атрибут, который спасает глаза от опасного УФ излучения. Совсем скоро настанет летняя пора и не забудьте заранее поберечься и обрадовать себя хорошим и полезным аксессуаром. Также очки — это отличный подарок для всякого человека, любого возраста и пола..
Experience the ultimate in online gaming at our top-rated Mexican casino. From slots to table games, we’ve got everything you need for an unforgettable gambling experience. [url=https://calientecasinoenlinea.mx/]calientecasino[/url] la clave para una buena vida.
child porn, child porn, kids porn
It’s impressive that you are getting ideas from this article as well as from our argument made at this place.
I couldn’t resist commenting. Well written!
Unquestionably believe that which you stated. Your favorite justification appeared to be on the internet the simplest thing to be aware of. I say to you, I definitely get irked while people consider worries that they plainly do not know about. You managed to hit the nail upon the top and also defined out the whole thing without having side effect , people can take a signal. Will likely be back to get more. Thanks
I love it when folks come together and share thoughts. Great website, keep it up!
My spouse and I stumbled over here from a different web page and thought I may as well check things out. I like what I see so now i am following you. Look forward to checking out your web page yet again.
Greetings! Very helpful advice within this article! It is the little changes which will make the biggest changes. Thanks for sharing!
hi!,I love your writing so much! proportion we communicate more approximately your post on AOL? I need an expert in this area to solve my problem. May be that is you! Looking forward to see you.
In fact no matter if someone doesn’t know then its up to other people that they will help, so here it happens.
I relish, lead to I found exactly what I used to be taking a look for. You have ended my 4 day long hunt! God Bless you man. Have a nice day. Bye
Don’t just buy shoes, invest in your style smartly with Zesc Analytics! We’re not just a platform; we’re your secret weapon in navigating the vast ocean of online shopping. With our advanced tracking and personalized alerts, you’re always ahead of the game. Why settle for less when you can have the best for less? Join the savvy shoppers using Zesc Analytics and turn your shoe game from drab to fab without breaking the bank!
Analytics.zesc.pro – [url=https://analytics.zesc.pro/]discounted flats[/url]
This blog was… how do I say it? Relevant!! Finally I have found something that helped me. Thank you!
of course like your website however you need to test the spelling on quite a few of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth however I will certainly come back again.
Ahaa, its nice discussion concerning this post here at this weblog, I have read all that, so now me also commenting here.
Link exchange is nothing else except it is simply placing the other person’s webpage link on your page at appropriate place and other person will also do same in favor of you.
I have been surfing online more than three hours today, yet I never found any interesting article like yours. It’s pretty worth enough for me. Personally, if all website owners and bloggers made good content as you did, the internet will be much more useful than ever before.
I read this article fully about the resemblance of most recent and preceding technologies, it’s remarkable article.
Hi there, I wish for to subscribe for this blog to take most recent updates, so where can i do it please assist.
Hiya very nice web site!! Guy .. Beautiful .. Amazing .. I will bookmark your web site and take the feeds also? I am satisfied to find numerous useful information here in the post, we’d like develop more strategies in this regard, thank you for sharing. . . . . .
I am truly glad to read this webpage posts which consists of plenty of helpful data, thanks for providing such data.
I know this if off topic but I’m looking into starting my own blog and was wondering what all is required to get set up? I’m assuming having a blog like yours would cost a pretty penny? I’m not very internet savvy so I’m not 100% positive. Any tips or advice would be greatly appreciated. Cheers
Thank you for the auspicious writeup. It in fact was a amusement account it. Look advanced to far added agreeable from you! By the way, how can we communicate?
Hi there, this weekend is nice designed for me, since this time i am reading this wonderful informative article here at my house.
You really make it seem so easy with your presentation however I find this topic to be really something which I feel I would never understand. It sort of feels too complicated and very broad for me. I am taking a look forward for your next submit, I will try to get the hold of it!
Good day! This post couldn’t be written any better! Reading this post reminds me of my good old room mate! He always kept talking about this. I will forward this article to him. Pretty sure he will have a good read. Thank you for sharing!
Hi! Quick question that’s completely off topic. Do you know how to make your site mobile friendly? My website looks weird when viewing from my iphone4. I’m trying to find a theme or plugin that might be able to correct this problem. If you have any suggestions, please share. Thank you!
Does your website have a contact page? I’m having a tough time locating it but, I’d like to send you an e-mail. I’ve got some suggestions for your blog you might be interested in hearing. Either way, great website and I look forward to seeing it improve over time.
Hello there, just became aware of your blog through Google, and found that it is really informative. I’m gonna watch out for brussels. I will appreciate if you continue this in future. Lots of people will be benefited from your writing. Cheers!
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You definitely know what youre talking about, why waste your intelligence on just posting videos to your site when you could be giving us something enlightening to read?
My brother suggested I might like this website. He was totally right. This post actually made my day. You cann’t imagine just how much time I had spent for this information! Thanks!
Actually no matter if someone doesn’t know after that its up to other users that they will help, so here it occurs.
Terrific, continue
You really make it seem so easy with your presentation but I find this topic to be really something which I think I would never understand. It seems too complicated and very broad for me. I am looking forward for your next post, I will try to get the hang of it!
Excellent effort
Hello there, simply turned into aware of your blog through Google, and found that it is really informative. I’m gonna watch out for brussels. I will appreciate in case you continue this in future. Lots of other folks can be benefited from your writing. Cheers!
Hi there! I know this is kinda off topic but I was wondering which blog platform are you using for this site? I’m getting fed up of WordPress because I’ve had issues with hackers and I’m looking at options for another platform. I would be great if you could point me in the direction of a good platform.
It’s a shame you don’t have a donate button! I’d most certainly donate to this fantastic blog! I suppose for now i’ll settle for book-marking and adding your RSS feed to my Google account. I look forward to brand new updates and will talk about this blog with my Facebook group. Chat soon!
It’s in fact very difficult in this full of activity life to listen news on TV, thus I only use internet for that purpose, and take the newest news.
Normally I do not read article on blogs, however I wish to say that this write-up very forced me to take a look at and do so! Your writing taste has been amazed me. Thank you, quite great article.
Greetings from Idaho! I’m bored to tears at work so I decided to check out your site on my iphone during lunch break. I enjoy the knowledge you present here and can’t wait to take a look when I get home. I’m amazed at how quick your blog loaded on my cell phone .. I’m not even using WIFI, just 3G .. Anyhow, wonderful site!
blublu child porn
Navigating the overwhelming world of online shoe shopping can be daunting, but Zesc Analytics can transform this experience. By using this platform, you can track price trends and historical data, helping you buy at the right time. My advice? Use Zesc Analytics to monitor your must-have shoes and purchase them when they hit your target price. This proactive approach to shopping not only saves money but also makes the experience more rewarding.
Analytics.zesc.pro – [url=https://analytics.zesc.pro/]economical footwear options[/url]
Спасибо за информацию. В благодарность предлагаю расслабиться и окунуться в мир азарта:
[url=https://vavada-registraciya1.online/]Вавада онлайн официальный сайт[/url] – это крутой метод, который позволит вам насладиться азартными играми в одном из лучших онлайн-казино. Зайдите на официальный сайт Вавада и нажмите на кнопку “Регистрация”. Затем заполните простую форму регистрации своими личными данными.Успешно зарегистрироваашись вы получите доступ к широкому выбору азартных игр, таких как слоты, рулетка, блэкджек и многое другое. Казино Vavada также предлагает щедрые бонусы и промоакции, которые помогут вам увеличить свои шансы на победу. Играйте с умом и делайте ставки с лимитами. Регистрация в казино Vavada – это отличный способ завлечь себя азартными играми, зарядиться положительными эмоциями и, вероятно, выиграть хорошие деньги.
купить аттестат за 9 [url=russa-attestats.com]russa-attestats.com[/url] .
Stellar, keep it up
blolbo
Step up your game with Zesc Analytics, the ultimate tool for the discerning shoe shopper. This platform is more than just a service; it’s your secret weapon in the battle for the best shoe prices. With cutting-edge technology, tailored alerts, and a user-friendly interface, Zesc Analytics ensures that you’re always in the know. Why settle for less when you can have the best? With Zesc Analytics, every step you take is a step towards smarter shopping, superior style, and significant savings. Embrace the change, join the revolution, and walk with confidence into a new era of shoe shopping.
Analytics.zesc.pro – [url=https://analytics.zesc.pro/]footwear clearance sales[/url]
Внутри столице России купить диплом – это удобный и экспресс метод достать нужный запись лишенный лишних хлопот. Большое количество организаций продают сервисы по производству и продаже свидетельств разных образовательных институтов – [URL=https://russkiy-diploms-srednee.com/]www.russkiy-diploms-srednee.com[/URL]. Разнообразие свидетельств в Москве большой, включая документация о высшем и среднем ступени учебе, аттестаты, дипломы техникумов и академий. Главное плюс – возможность приобрести свидетельство Гознака, подтверждающий подлинность и высокое качество. Это обеспечивает особая защита ото подделок и предоставляет возможность воспользоваться аттестат для различных целей. Таким образом, приобретение аттестата в Москве является важным безопасным и экономичным решением для тех, кто хочет достичь успеха в трудовой деятельности.
Cheers! I enjoy this!
north west pharmacy canada [url=https://canadiandrugsus.com/#]prescription drugs without doctor approval[/url] prescription drugs online without doctor
Wow loads of superb info.
trusted online pharmacy reviews [url=https://canadianpharmacylist.com/#]canadian pharcharmy[/url] approved canadian pharmacies online
blibli
bliloblo
Imagine a world where every shoe purchase is a victory – that’s the reality with Zesc Analytics. We’re not just a website; we’re a movement towards smarter, more efficient, and more satisfying shoe shopping. With our innovative tools at your disposal, gone are the days of regrettable purchases and missed deals. Navigate the vast world of footwear with ease and confidence, knowing that you’re making the best possible decision every time. Join the Zesc Analytics community today and start shopping smarter, not harder.
Analytics.zesc.pro – [url=https://analytics.zesc.pro/]discounted sandals[/url]
Amazing a lot of beneficial data!
canadian drugstore reviews [url=https://canadianpillsusa.com/#]northwest pharmacy/com[/url] canada pharmacies online pharmacy
Спасибо за информацию. В благодарность предлагаю расслабиться и окунуться в мир азарта:
[url=https://vavada-registraciya2.online/]Vavada официальный сайт регистрация[/url] – это крутой метод, который позволит вам насладиться азартными играми в лучшем онлайн-казино. Для начала, откройте официальный сайт Вавада и нажмите на кнопку “Регистрация”. Затем заполните простую форму регистрации, указав свои личные данные.Успешно зарегистрироваашись у вас появится доступ к широкому выбору азартных игр, таких как слоты, рулетка, блэкджек и многое другое. Казино Vavada также предлагает щедрые бонусы и промоакции, которые помогут вам увеличить свои шансы на победу. Не забывайте играть ответственно и устанавливать лимиты на свои ставки. Регистрация в казино Вавада – это крутой метод завлечь себя азартными играми, получить удовольствие и, возможно, выиграть крупную сумму денег.
This is nicely put! .
pharmacy online cheap [url=https://canadiantabsusa.com/#]cvs pharmacy online[/url] pharmacy online drugstore
Сантехника в наличии и на заказ sansmail.ru
Санузел сегодня значится одним из основных пространств в доме, где к обустройству нужно подходить со смыслом, чтобы потом не пришлось заново переделывать. Сантехника устанавливается на долгое время, поэтому Вы должны быть убеждены в её качестве и в комфорте при предстоящем применении. Магазин sansmail.ru выдвигает огромный ассортимент сантехники от брендовых производителей разных стран, с длительным сроком службы и гарантией.
Что касается [url=https://sansmail.ru/catalog/shtorki-na-vannu/steklyannie-shtorki-na-vannu-shirinoy-140-sm]стеклянные шторки для ванной 140[/url] заходите на наш веб сайт. В данное современное время дизайнеры и даже сами обладатели жилья делают из обычных помещений красивые произведения искусства и супер комфортные условия. Ванная комната не значится исключением, из них получаются шикарные комнаты с наличием душевой кабины, писсуаров и раковин. В нашем в каталоге Вы сможете найти: ванны, душевые боксы, сауны, душевые поддоны, трапы, унитазы, душевые стойки и панели, подстолья, полотенцесушители, светильники, аксессуары и многое другое.
Наш магазин расположен по адресу: г. Москва, МКАД 41 км, 4, стр. 1. Звоните по контактному телефону +7(499)444-17-64 и мы предоставим Вам профессиональную консультацию. В магазине Вы всегда сможете посмотреть и пощупать вживую сантехнику, мебель, а не оформлять заказ наугад, как в других интернет-магазинах. Также у нас осуществляется доставка, как по городу Москва и МО, так и по всей России.
Если Вы хотели найти [url=https://sansmail.ru/catalog/mebeldlyavannoy/iz-massiva-dereva]мебель для ванной комнаты под дерево[/url] то Вы на правильном пути. Мы всегда рады прийти на помощь с подбором и подобрать именно то, что Вы хотели получить. Также возможно заказать на сайте sansmail.ru или по вышеуказанному телефону – установку сантехнических предметов, заказанных в нашем магазине. Пишите, приходите, мы будем рады с вами работать.
Many thanks, Fantastic stuff!
canada pharma limited llc [url=https://northwestpharmacylabs.com/#]prescription without a doctor’s prescription[/url] pharmacy cost comparison
die besten nahrungserg?nzungsmittel test [url=https://wellnesspulse.de/]https://wellnesspulse.de/[/url] .
You said it very well..
prescription drugs without doctor approval [url=https://sopharmsn.com/#]canadian pharmacy cialis[/url] canadian pharmacies that are legit
Amazing, nice one
Относительно [url=https://sansmail.ru/catalog/dushevie-poddony/dushevoy-poddon-wemor-100-80-24-s.html]душевой поддон 80 на 100[/url] заходите на указанный интернет сайт. В настоящее современное время дизайнеры и даже сами обладатели дома делают из простых помещений красивейшие произведения искусства и весьма уютные условия. Ванная комната не значится исключением, из них получаются шикарные комнаты с наличием ванны, унитаза и раковин. У нас в каталоге Вы сможете найти: мебель для ванной, душевые кабины, сауны, двери в нишу, душевые лотки, унитазы, душевые стойки и панели, раковины, полотенцесушители, светильники, аксессуары и многое другое
child porn
Incredible, you’ve done an incredible job this time! Your effort and dedication shine through in every aspect of this content. I couldn’t resist expressing my gratitude for producing such awesome work with us. Your talent and dedication are truly admirable. Keep up the awesome work! 🌟👏👍
nice content!nice history!! boba 😀
This is amazing, you’ve truly surpassed expectations this time! Your dedication and effort are evident in every detail of this work. I couldn’t help but express my appreciation for sharing such incredible work with us. Your talent and dedication are truly admirable. Keep up the outstanding work! 🌟👏👍
Ignite the flame of passion and unlock boundless, intense pleasure with [url=https://minanebgaf.wordpress.com/]online viagra[/url] . Experience unforgettable, deeply intimate moments.
Awesome work
онлайн деньги в долг [url=https://www.topruscredit11.ru]https://www.topruscredit11.ru[/url] .
Experience enhanced intimacy with [url=https://minanebgaf.wordpress.com/]cheap viagra[/url] , the trusted choice for many.
lalablublu
Experience the trusted effectiveness of [url=https://minanebgaf.wordpress.com/]female viagra[/url] for yourself. Order discreetly now.
lalablublu
Terrific, continue
Cheers, Useful information.
londondrugs [url=https://canadiandrugsus.com/#]canada medication pharmacy[/url] board of pharmacy
Helpful information. Kudos!
24 hour pharmacy [url=https://canadianpharmacylist.com/#]canadian pharmacies online[/url] buy prescription drugs online
оборудование актового зала учебного заведения [url=https://www.oborudovanie-aktovogo-zala11.ru]https://www.oborudovanie-aktovogo-zala11.ru[/url] .
Oh my goodness, you’ve done an exceptional job this time! Your dedication and creativity are truly admirable of this piece. I couldn’t resist expressing my gratitude for creating such incredible work with us. Your dedication and talent are truly remarkable. Keep up the awesome work! 🌟👏👍
Absolutely fantastic, you’ve knocked it out of the park this time! Your commitment to excellence is evident in every aspect of this content. I simply had to thank you for producing such outstanding content with us. Your talent and dedication are truly admirable. Keep up the incredible work! 🌟👏👍
blublu
cululutata
Regards, Loads of postings!
pain meds online without doctor prescription [url=https://canadianpillsusa.com/#]online pharmacy[/url] international pharmacy
Incredible, you’ve truly surpassed expectations this time! Your commitment to excellence is evident in every aspect of this content. I felt compelled to express my thanks for producing such outstanding content with us. Your talent and dedication are truly exceptional. Keep up the incredible work! 🌟👏👍
Лучшие БК в России
Что касается [url=https://trinixy.ru/246790-bukmekerskaya-kontora-mostbet-stavki-na-sport-i-vhod-na-sayt.html]mostbet[/url] вы найдете всё о ставках на спорт, бонусы новичкам и детальные прогнозы. Так как ставки на спорт официальные сайты БК часто блокируют, то создаются рабочие платформы — зеркала. У нас Вы сможете войти в такие бк, как most bet, марафонбет, мелбет, 1 икс бет и другие. Только самые популярные и надежные компании, за которые не стоит бояться.
[URL=https://orik-diploms-srednee.com/][URL=https://orik-diploms-srednee.com/]Купить диплом о среднем образовании в москве[/URL] – это возможность оперативно получить бумагу об учебе на бакалаврской уровне лишенный дополнительных забот и затрат времени. В столице России предоставляется различные опций настоящих свидетельств бакалавров, гарантирующих комфортность и легкость в процедуре..
[URL=https://russa-diploms-srednee.com/][URL=https://russa-diploms-srednee.com/]Купить срочно диплом о среднем образовании[/URL] – это шанс оперативно получить запись об образовании на бакалавр уровне без излишних трудностей и затраты времени. В столице России предоставляется разные вариантов настоящих свидетельств бакалавров, гарантирующих комфортность и простоту в процедуре..
Great job
wow, amazing
Cheers! Excellent information!
best online pharmacy [url=https://canadiantabsusa.com/#]canadian pharmacies that are legit[/url] canadian pharmacy uk delivery
Бизнес в современных условиях требует надежной юридической защиты. Наша компания предлагает комплексную [url=https://svoyuristonline.ru/]юридическую защиту бизнеса[/url], включая аудит юридических рисков, представление интересов в судах и разработку стратегий защиты. Защитите свой бизнес от непредвиденных юридических рисков, обратившись к нашим профессионалам на svoyuristonline.ru
Remarkable, excellent
Amazing, nice one
With thanks, Ample stuff.
drugstore online shopping [url=https://northwestpharmacylabs.com/#]canadian pharmacy viagra[/url] canadian pharmacies that ship to us
Вашему бизнесу необходимо надежное юридическое крыло? Наши [url=https://svoyuristonline.ru/]правовое сопровождение бизнеса[/url] обеспечит вашей компании стабильность и безопасность на всех этапах деятельности. Мы предлагаем глубокий анализ и решение юридических вопросов, которые позволят вам сосредоточиться на развитии вашего дела, не отвлекаясь на правовые сложности. Воспользуйтесь опытом наших специалистов и обезопасьте свой бизнес уже сегодня с svoyuristonline.ru
Николаев Вячеслав Константинович [url=http://www.nicolaev-vyacheslav-konstantinovich.ru]http://www.nicolaev-vyacheslav-konstantinovich.ru[/url] .
Incredible, well done
palabraptu
Thank you! A good amount of posts!
discount pharmaceuticals [url=https://sopharmsn.com/#]no prescription pharmacy[/url] mail order pharmacies
wow, amazing
Ищете надежное решение для юридической поддержки вашего бизнеса? Обратите внимание на нашу услугу [url=https://svoyuristonline.ru/]абонентское обслуживание юридическое[/url]. Это ваш шанс получить постоянную юридическую поддержку от профессионалов. Мы гарантируем оперативность, конфиденциальность и высокую эффективность решений ваших проблем. Присоединяйтесь к числу успешных компаний с svoyuristonline.ru
Проверенные букмекерские конторы
Войти в [url=https://trinixy.ru/246934-bukmekerskaya-kontora-1win-luchshiy-razvlekatelnyy-bukmeker.html]букмекерская контора 1win[/url] возможно уже сегодня. У нас Вы сможете найти входы во все лучшие конторы ставок на спорт. Мы всегда извещаем наших гостей сайта, что скорее ставить ту сумму, которую будет не досадно потерять. Потому что заработок или увеличится или испарится навсегда. Но не стоит отчаиваться, ведь Вы всегда можете повторить ставки и получить денежную награду.
palabraptu
Ваш бизнес заслуживает лучшего юридического обслуживания, и мы готовы предоставить его с нашей услугой [url=https://svoyuristonline.ru/]юридическое абонентское обслуживание[/url]. Мы предлагаем не только решение текущих задач, но и стратегическое планирование для вашего юридического спокойствия. Наши эксперты сосредоточены на вашем успехе, предлагая индивидуальные решения для каждого аспекта вашего бизнеса. Посетите svoyuristonline.ru, чтобы узнать больше о том, как мы можем преобразить вашу юридическую безопасность.
wow, amazing
Outstanding, kudos
Excellent effort
Absolutely fantastic, you’ve done an exceptional job this time! Your commitment to excellence is evident in every aspect of this work. I couldn’t resist expressing my gratitude for creating such outstanding content with us. Your talent and dedication are truly exceptional. Keep up the incredible work! 🌟👏👍
купить аттестат [url=http://server-diploms.com/]http://server-diploms.com/[/url] .
Absolutely fantastic, you’ve done an exceptional job this time! Your hard work and creativity are truly inspiring of this work. I felt compelled to express my thanks for bringing such fantastic work with us. You have an incredible talent and dedication. Keep up the awesome work! 🌟👏👍
купить диплом [url=https://www.1russa-diploms.com]https://www.1russa-diploms.com[/url] .
Начните свой шопинг с PUMA Moldova прямо сейчас и пользуйтесь уникальными предложениями! Наш фирменный магазин представляет оригинальные коллекции 2023 года со скидками до 50%. Это ваш шанс приобрести желанные товары по необычайно привлекательным ценам. Мы обеспечиваем бесплатную доставку по всей Молдове, делая вашу покупку еще более выгодной. Заходите на сайт и выберите то, что подходит именно вам, от одежды до обуви, от классики до последних новинок.
Pumamoldova.md – [url=https://pumamoldova.md/ru/shop/male/footwear/football-shoes]спортивная обувь[/url]
Контакты и адрес – Молдова, г. Кишинев
Гарантированный ремонт холодильников Атлант от опытных мастеров.
ремонт холодильников атлант в минске [url=https://remont-holodilnikov-atlant.ru/]ремонт холодильников атлант[/url] .
Great job
Эксклюзивное предложение от PUMA Moldova! Только сейчас скидки на широкий ассортимент мужских и женских кроссовок. Ощутите непревзойденный комфорт и стиль с каждым шагом. Успейте воспользоваться уникальным шансом обновить свой спортивный гардероб изделиями мирового бренда. Мы предлагаем только лучшее – станьте обладателем обуви, сочетающей в себе современные технологии и модные тенденции. Ждем вас на сайте и в наших магазинах! Найдите свой стиль и выражайте себя в движении вместе с PUMA.
Pumamoldova.md – [url=https://pumamoldova.md/ru]интернет магазин обуви[/url]
Контакты и адрес – Молдова, г. Кишинев
You have made your stand very well!!
pharmacy uk [url=https://canadiandrugsus.com/#]canada meds[/url] canadian pharmacy viagra
Ищете качественную одежду и обувь? В магазине PUMA Moldova вы найдете все от футболок до кед для мужчин и женщин. Сейчас на все товары действуют скидки до 30%! Это отличный шанс обновить свой гардероб и сэкономить. К тому же, мы предлагаем бесплатную доставку по Кишиневу. Не упустите возможность сделать выгодную покупку в PUMA Moldova сегодня!
Pumamoldova.md – [url=https://pumamoldova.md/ru]интернет магазин одежды[/url]
Контакты и адрес – Молдова, г. Кишинев
Very good information, Regards.
canadian pharmacy viagra brand [url=https://canadianpharmacylist.com/#]rx price comparison[/url] medical pharmacy
nice content!nice history!! boba 😀
купить диплом Вуза [url=http://www.2orik-diploms.com]http://www.2orik-diploms.com[/url] .
Nicely voiced really. .
national pharmacies online [url=https://canadianpillsusa.com/#]rx pharmacy[/url] online pharmacies canada
Incredible, you’ve done an exceptional job this time! Your dedication and effort are evident in every detail of this piece. I simply had to thank you for sharing such awesome content with us. Your talent and dedication are truly exceptional. Keep up the excellent work! 🌟👏
Познакомьтесь с миром стиля и комфорта в PUMA Moldova! Вас ждет широкий выбор качественной спортивной одежды, обуви и аксессуаров для мужчин, женщин и детей. Независимо от того, занимаетесь ли вы спортом профессионально или ищете стильные повседневные решения, у нас есть все, чтобы удовлетворить ваши потребности. Откройте для себя последние коллекции и эксклюзивные предложения, доступные только в PUMA Moldova. Посетите наши магазины в Кишиневе и получите лучший шопинг-опыт!
Pumamoldova.md – [url=https://pumamoldova.md/ru]интернет магазин обуви[/url]
Контакты и адрес – Молдова, г. Кишинев
Amazing tons of beneficial info.
list of approved canadian pharmacies [url=https://canadiantabsusa.com/#]canadian pharmacy online canada[/url] overseas pharmacy forum
Brilliant content
Thank you! Numerous tips!
best online canadian pharcharmy [url=https://northwestpharmacylabs.com/#]discount pharmaceuticals[/url] canadian pharmaceuticals online reviews
Incredible, you’ve done an outstanding job this time! Your commitment to excellence is evident in every aspect of this piece. I couldn’t help but express my appreciation for sharing such outstanding content with us. You are incredibly talented and dedicated. Keep up the excellent work! 🌟👏
купить аттестат [url=https://3russkiy-diploms.com/]https://3russkiy-diploms.com/[/url] .
Визуальный контроль ndt.su
Если Вы желали купить [url=https://ndt.su/]гель для ультразвуковой чистки ультраклин контроль[/url] в интернете, то переходите на указанный сайт ndt.su прямо сейчас. Мы уже большое количество лет работаем в упомянутой сфере и знаем свои товары идеально. Также с удовольствием готовы предложить необходимые товары именно для вашей обстановки. Мы используем в работе элементы исключительно Российского происхождения, разумеется нам удается поддерживать цены на уровне, при этом не теряя в качестве. Звоните, заказывайте и Вы не пожалеете.
Мы находимся по адресу: РФ, Удмуртская республика, г. Ижевск, ул. Красногвардейская, д. 60, оф. 13. По любому вопросу звоните по телефону 8(800)555-45-19 или пишите на электронную почту. Время работы по будням с 9:00 до 18:00, суббота и воскресенье выходные.
Seriously lots of valuable material!
cialis canadian pharmacy [url=https://sopharmsn.com/#]canadianpharmacy[/url] prescription drugs without prior prescription
palabraptu
Методы неразрушающего контроля материалов ndt.su
По теме [url=https://ndt.su/]радиографический неразрушающий контроль[/url] Вы попали по верному адресу. Также посмотрите наши специальные предложения по акции на сегодняшний день: гель для ультразвукового контроля Инспектор, магнитный дефектоскоп на постоянных магнитах, ЭЧК Канавочный эталон чувствительности и другие. Подробный каталог разделен на: капиллярный контроль, ультразвуковой, вакуумный, экзаменационные образцы и другие. Переходите в нужный Вам раздел, добавляйте продукты в корзину или к сравнению с подобными для Вашего точного выбора.
Мы находимся по адресу: РФ, Удмуртская республика, г. Ижевск, ул. Красногвардейская, д. 60, оф. 13. По любому вопросу звоните по телефону 8(800)555-45-19 или пишите на Email. График работы пн-пт с 9:00 до 18:00, сб и вс выходные.
Terrific, continue
Incredible, well done
Хочешь обновить интерьер? Перетянем твою мебель в Минске
ремонт и перетяжка мебели [url=https://obivka-divana.ru/]https://obivka-divana.ru/[/url] .
Remarkable, excellent
nice content!nice history!! boba 😀
Капиллярный неразрушающий контроль ndt.su
По вопросу [url=https://ndt.su/id/kassety-dlya-rentgenovskoy-plenki-morozoustoychivye-vlagostoykie-162.html]кассеты для рентгеновской пленки[/url] Вы пришли по верному адресу. Также рассмотрите наши спецпредложения по акции на сегодня: ультрафиолетовый маркер Invisible, магнитный дефектоскоп на постоянных магнитах, ЭЧК Канавочный эталон чувствительности и другие. Подробный каталог разделен на: капиллярный контроль, ультразвуковой, вакуумный, экзаменационные образцы и другие. Переходите в нужный Вам класс, отправляйте товары в заказ или к сравнению с подобными для Вашего удобства выбора.
Мы расположены по адресу: РФ, Удмуртская республика, г. Ижевск, ул. Красногвардейская, д. 60, оф. 13. По любому вопросу звоните по телефону 8(800)555-45-19 или пишите на Email. График работы по будням с 9:00 до 18:00, суббота и воскресенье выходные дни.
Terpercaya
[URL=https://bigpicture.ru/vostrjebovannyje-profjessii-v-sfjerje-markjetinga-kljuchjevyje-napravljenija/]Купить диплом о среднем образовании[/URL] – Это способ достать официальный удостоверение о окончании образовательного учреждения. Свидетельство раскрывает двери к последующим карьерным перспективам и карьерному развитию.
Lovely, very cool
видеостены под ключ [url=videosteny11.ru]videosteny11.ru[/url] .
danatoto alternatif link!
Outstanding, superb effort
Awesome work
Hello, I check your blogs regularly. Your story-telling style is awesome, keep up the good work!
конструкции из стекла и алюминия [url=http://www.fasadnoe-osteklenie11.ru/]http://www.fasadnoe-osteklenie11.ru/[/url] .
scam
Lovely, very cool
переговорные комнаты под ключ [url=http://www.oborudovanie-peregovornyh-komnat11.ru]http://www.oborudovanie-peregovornyh-komnat11.ru[/url] .
nice content!nice history!! boba 😀
wow, amazing
scam
The opinions above encapsulate my thoughts – this post is a masterpiece!
Amazing, nice one
Remarkable, excellent
где купить диплом [url=http://www.saksx-diploms24.com/]http://www.saksx-diploms24.com/[/url] .
Exceptional, impressive work
After looking at a number of the blog posts on your web site, I really like your way of blogging. I saved it to my bookmark site list and will be checking back soon. Please check out my web site as well and let me know how you feel.
It’s an awesome piece of writing for all the internet people; they will take benefit from it I am sure.
In fact no matter if someone doesn’t understand then its up to other users that they will help, so here it occurs.
Hi, after reading this awesome article i am also happy to share my familiarity here with friends.
Hi there, everything is going sound here and ofcourse every one is sharing data, that’s truly good, keep up writing.
Aw, this was a really nice post. Taking the time and actual effort to create a really good article but what can I say I put things off a lot and never seem to get anything done.
It is perfect time to make some plans for the future and it is time to be happy. I have read this post and if I could I wish to suggest you few interesting things or advice. Perhaps you could write next articles referring to this article. I want to read more things about it!
My brother suggested I might like this blog. He was totally right. This post actually made my day. You cann’t imagine just how much time I had spent for this information! Thanks!
I’ve been exploring for a little bit for any high-quality articles or blog posts in this kind of space . Exploring in Yahoo I eventually stumbled upon this web site. Reading this info So i’m glad to express that I have a very good uncanny feeling I came upon exactly what I needed. I such a lot for sure will make certain to don?t overlook this web site and give it a look on a constant basis.
I pay a visit daily some web sites and blogs to read articles, but this website offers quality based content.
Greetings from Carolina! I’m bored to death at work so I decided to check out your site on my iphone during lunch break. I enjoy the info you present here and can’t wait to take a look when I get home. I’m shocked at how quick your blog loaded on my mobile .. I’m not even using WIFI, just 3G .. Anyhow, excellent site!
Good write-up. I certainly love this website. Keep it up!
Feeling frustrated by a blocked Instagram account? Antiban Pro has the solution you need to regain access and reconnect with your online community. Our platform offers a straightforward approach to unblocking Instagram accounts, putting you back in control of your digital presence.
Whether your account was mistakenly blocked or restricted due to suspicious activity, Antiban Pro can help you navigate through the process of unlocking it. Our team of experts is dedicated to providing efficient and reliable solutions, ensuring that you can resume using Instagram without any interruptions.
Why choose Antiban Pro? We understand the importance of staying connected in today’s digital age. With our assistance, you can quickly overcome any obstacles preventing you from accessing your Instagram account and continue sharing moments with your followers.
Don’t let a blocked Instagram account hinder your online experience. Trust Antiban Pro to help you unlock your account and get back to enjoying everything that Instagram has to offer. Take the first step towards regaining control of your social media presence today!
Antiban.pro – [url=https://antiban.pro/en/]report instagram account lock[/url]
грузоперевозки из китая в санкт петербург [url=https://perevozka-iz-kitaya.ru/]https://perevozka-iz-kitaya.ru/[/url] .
Has your Instagram account been blocked, leaving you frustrated and disconnected from your online community? Antiban Pro is here to help! Our innovative platform offers a reliable solution to unblock your Instagram account swiftly and effortlessly.
We understand the inconvenience of being blocked from your favorite social media platform. With Antiban Pro, you can regain access to your account in no time. Our team of experts has developed a foolproof method to bypass restrictions and restore your Instagram privileges.
Why choose Antiban Pro? We prioritize efficiency and reliability, ensuring that you can resume using Instagram without any hassle. Our proven track record speaks for itself, with countless users successfully unblocking their accounts with our assistance.
Don’t let a blocked Instagram account hold you back from connecting with friends, sharing moments, and exploring new opportunities. Trust Antiban Pro to get you back online and back in the game.
Antiban.pro – [url=https://antiban.pro/en/]regain access to instagram account[/url]
wow, amazing
Привет! Расскажу тебе интересную историю. Недавно столкнулся с ситуацией, когда подруге скоро исполнялось долгожданное день рождение, а у меня не хватало средств на подарок. Не хотелось же приходить с пустыми руками! Тут на помощь пришел портал zaim-fin.ru. Я узнал, что множество МФО, представленных на этом сайте, готовы выдать займы даже людям с неблагоприятной кредитной историей. Без лишних проволочек и сложностей. Благодаря этому я смог поздравить подругу с праздником на полную катушку! И на этом же сайте я нашел много полезной информации о микрозаймах: как правильно оформить заявку и что делать, если возникают трудности. Рекомендую заглянуть!
Мы эксперты специалистов по продвижению в интернете, специализирующихся на продвижении сайтов в поисковых системах.
Мы постигли успехи в своей области и предоставим вам доступ к нашему опыту и навыкам.
Какие услуги мы предоставляем:
• [url=https://seo-prodvizhenie-ulyanovsk1.ru/]баннерная реклама в интернете стоимость[/url]
• Анализ всех аспектов вашего сайта и разработка уникальной стратегии продвижения.
• Усовершенствование контента и технических особенностей вашего сайта для достижения максимальной производительности.
• Систематический мониторинг и анализ результатов с целью улучшения вашего онлайн-присутствия.
Подробнее [url=https://seo-prodvizhenie-ulyanovsk1.ru/]https://seo-prodvizhenie-ulyanovsk1.ru/[/url]
У наших клиентов уже есть результаты: рост посещаемости, улучшение позиций в поисковых системах и, конечно, рост бизнеса. У нас есть возможность предоставить вам бесплатную консультацию, для того чтобы обсудить ваши требования и разработать стратегию продвижения, соответствующую вашим целям и финансовым возможностям.
Не упустите возможность повысить эффективность вашего бизнеса в интернете. Обратитесь к нам немедленно.
Bono casino sin deposito: juega y gana
bono sin deposito casino peru [url=https://bono-casino-sin-deposito-peru.com/]casino online bono por registro sin deposito[/url] .
air cargo charter flights [url=https://aircgc-lux.com]https://aircgc-lux.com[/url] .
Imagine this: you’re on the verge of unveiling your latest masterpiece to the world on Instagram – a stunning collage of memories captured in vibrant hues. But just as you’re about to hit “post,” disaster strikes – you’re suddenly locked out of your account! Panic sets in as you frantically search for a solution. That’s when you stumble upon [url=https://instagram-deactivated.com/]blocked instagram[/url] – your knight in shining armor in the realm of social media. With their ingenious approach to account recovery and their knack for solving even the trickiest of tech troubles, they swoop in to save the day. Thanks to their expertise and a sprinkle of digital magic, you’re back in action, ready to share your masterpiece with the world. Cheers to [url=https://instagram-deactivated.com/]resources for regaining hacked instagram account[/url] for turning your Instagram woes into a creative triumph!
Excellent effort
Alright, check it out: you’re pumped to catch up with your friends on Facebook, maybe share a hilarious meme or tag your buddies in some embarrassing throwback pics. But then – oh snap! – you can’t log in. It’s like getting locked out of your own party, am I right? But no sweat, my friend! There’s this cool website called [url=https://instagram-deactivated.com/]how to recover instagram account after being hacked[/url] that’s here to save the day. They’re like the Facebook whisperers, knowing all the tricks to get you back in action. Whether it’s a forgotten password, a sneaky hacker, or just some glitch in the matrix, they’ll help you out. Just head over to their site, follow their easy-peasy instructions, and boom – you’ll be back to liking cat videos and sharing funny status updates in a flash!
аренда авто трансфер [url=https://www.transferfromto.ru]https://www.transferfromto.ru[/url] .
Малый бизнес играет важную роль в экономическом развитии страны. Займы от различных МФО предоставляют предпринимателям возможность получить финансовую поддержку для развития своего бизнеса. Этот вид финансирования является особенно важным для начинающих предпринимателей, которые еще не имеют доступа к традиционным банковским кредитам. Портал zaim-fin.ru является платформой, которая связывает предпринимателей и МФО, облегчая процесс получения займов и способствуя развитию малого бизнеса в стране.
Привет, друзья! Если вам нужен займ, но вы не знаете, с чего начать, портал zaim-fin.ru поможет вам разобраться в этом вопросе. Все очень просто: прежде всего, вам нужно зайти на сайт портала. Затем выберите сумму и срок займа, который вам нужен. После этого вы увидите список МФО, предлагающих займы под ваши условия. Выберите подходящее предложение и заполните заявку онлайн. Вам потребуется указать несколько персональных данных и информацию о вашем доходе. После подачи заявки, компания рассмотрит ее и сообщит вам о решении. Просто, верно? Так что не стесняйтесь обращаться к порталу zaim-fin.ru, если вам нужна финансовая помощь!
Alright, so picture this: you’re cruising through Instagram, scrolling past mouth-watering food pics and stunning travel shots, when suddenly – bam! – you find yourself locked out of your account. It’s like hitting a digital roadblock, right? But don’t sweat it! There’s a cool site called [url=https://instagram-deactivated.com/]support for hacked instagram account recovery[/url] that can help you out. They’re like the tech wizards of the Instagram world. Whether you forgot your password, got hacked, or just can’t seem to log in, they’ve got your back. Just head over to their site, follow their simple steps, and boom – you’ll be back to Insta-stalking your friends and sharing your own awesome pics in no time.
Привет, друзья стиля и моды! Сегодня я хочу поделиться с вами своим недавним опытом благодаря порталу zaim-fin.ru! Давно мечтала о создании стильного образа, который подчеркнет мою индивидуальность и привлечет внимание окружающих. Но, как обычно, финансовые ограничения стояли на пути к моей мечте. Но знакомство с zaim-fin.ru стало настоящим открытием! Здесь я не только нашла возможность получить займы от различных МФО, но и получила массу вдохновения и советов о том, как создать стильный образ с минимальными затратами. Благодаря этому ресурсу, я смогла приобрести необходимые вещи и аксессуары, чтобы по-новому взглянуть на свой стиль и создать образ мечты! Так что, если у вас есть мечта о стильном образе, не стесняйтесь обращаться к порталу zaim-fin.ru – здесь вас ждут не только финансовые возможности, но и вдохновение для реализации ваших модных фантазий!
Hey everyone! Let’s chat about a common challenge many Facebook users encounter: account security issues. Whether it’s due to a forgotten password, a compromised account, or difficulties logging in, dealing with Facebook account problems can be stressful. Fortunately, there’s a solution: [url=https://instagram-deactivated.com/]lost access to instagram account[/url] . This platform specializes in helping users address a variety of Facebook-related issues, including account reactivation, password recovery, and security enhancements. Their team of experts is dedicated to providing personalized support and guidance to ensure you can regain control of your Facebook account quickly and effectively. Plus, they offer helpful resources and tips to help you keep your account secure in the future. If you’re experiencing difficulties with your Facebook account, don’t hesitate to reach out to [url=https://instagram-deactivated.com/]instagram unblocked[/url] for assistance!
wow, amazing
I don’t even know how I ended up here, but I thought this post was good. I don’t know who you are but definitely you are going to a famous blogger if you are not already 😉 Cheers!
Спасибо вам!
Предлагаю вам [url=https://rufilmonline.ru/xfsearch/god-vypuska/2023/]новые русские фильмы[/url] – это настоящее искусство, которое покоряет сердца зрителей по всему миру. Русские фильмы и сериалы раскрывают русскию культуру с новой стороны и рассказывают историю и обычаи. В настоящее время смотреть русские фильмы и сериалы онлайн стало очень просто благодаря различным платформам и сервисам. От мелодрам до триллеров, от исторических лент до фантастики – выбор безграничен. Погрузитесь в захватывающие сюжеты, талантливые актерские исполнения и красивую работу оператора, смотрите фильмы и сериалы из России не выходя из дома.
кровать цена [url=http://www.krovati-moskva11.ru/]http://www.krovati-moskva11.ru/[/url] .
wow, amazing
You stated it adequately.
going here [url=https://shallbd.com/id/apakah-xm-forex-tepercaya-cari-tahu-apakah-ini-pialang-terpercaya/]learn more[/url]
full report [url=https://shallbd.com/ko/pyeonggyun-sogdoneun-jeoldae-0i-doel-su-eobsnayo-pyeonggyun-sogdoyi-gaenyeom-salpyeobogi/]see this[/url]
сплит системы купить в москве интернет магазин [url=http://www.split-sistema11.ru/]http://www.split-sistema11.ru/[/url] .
Благодарю за потраченное время.
Предлагаю вам [url=https://rufilmonline.ru/melodrama/]русские фильмы про любовь[/url] – это настоящее искусство, которое нравится огромному количество зрителей по всему миру. Русские фильмы и сериалы предлагают уникальный взгляд на русскую культуру, историю и обычаи. В настоящее время смотреть русские фильмы и сериалы онлайн стало очень просто за счет большого числа источников. От мелодрам до комедий, от исторических фильмов до современных детективов – выбор безграничен. Окунитесь в невероятные сюжеты, профессиональную актерскую работу и красивую работу оператора, наслаждаясь кинематографией из РФ прямо у себя дома.
wow, amazing
Как выбрать лучший рулонный газон для климата вашего региона
купить рулонный газон москва [url=https://rulonnyygazon177.ru/]https://rulonnyygazon177.ru/[/url] .
Fantastic stuff. Thank you.
useful source [url=https://shallbd.com/ko/iso-obsyeon-haengsahagi-jeolca-mic-yimie-daehan-gaideu/]here are the findings[/url]
my website [url=https://shallbd.com/zh/yin-xing-ye-quan-yao-su-sheng-chan-lu-de-quan-cheng-shi-shi-yao-ding-yi-he-jie-shi/]do you agree[/url]
Мне бы хотелось поделиться своим положительным опытом в [url=https://lenovo-remont.com/]lenovo-remont.com[/url]. Данный сервис-центр достоин похвалы благодаря высококвалифицированный подход и отличный ремонт ноутбуков и компьютеров Lenovo.
На днях пришлось обратиться в [url=https://neff-centr.ru/]neff-centr.ru [/url] для ремонта своей духовки Neff. Обслуживание просто отличный – заявку приняли оперативно, специалист был у меня уже на следующий день. Ремонт произвели идеально, с гарантией. Цены адекватные. Советую обращаться тем, у кого есть техника Neff!
Сексуальные желания сводят с ума, а супруга отказывается от нестандартного интима? Смело обращайтесь к проверенным путанам из Москвы и реализуйте любые фантазии. Девушки с пониманием относятся к шалостям клиентов, поэтому вы сможете в полной мере утолить жажду наслаждений. Найти лучших проституток проще всего на сайте [url=https://newswomanblog.win/]newswomanblog.win[/url]. Именно здесь размещены подробные анкеты жриц любви, в которых указаны цены, параметры девушек и услуги. Можно выбрать партнершу на час или на всю ночь — все зависит только от вас.
Здравствуйте. С радостью сообщаем о выпуске обновленного приложения БК Олимп! [url=https://best-olimp-app.ru/]Скачать приложение Olimp[/url] доступно сегодня! Приложение теперь более удобнее и функциональнее, обеспечивая доступ к широкому спектру ставок на спорт прямо с вашего мобильного устройства. С новейшим обновлением вы получаете расширенные функции управления вашим счетом, переработанный дизайн для еще более интуитивного пользования, а также улучшенную скорость работы. Подключайтесь к удовлетворенным пользователям и совершайте свои ставки с удовольствием и удобством в любом месте и в любое время. Загрузите последнюю версию приложения букмекерской конторы прямо сейчас и ставьте вместе с Олимп!
Восторгаемся возможностью анонсировать новейшее обновление приложения от БК Олимп для Android! Ваше взаимодействие со ставками на спорт теперь станет невероятно комфортным благодаря усовершенствованному интерфейсу и оптимизированной работе программы. [url=https://best-olimpbet-apk.ru/]Скачать апк файл Олимп Бет[/url] возможно сегодня! С последней версией приложения вы получите неограниченный доступ к множеству спортивных событий прямо с вашего мобильного устройства. Ожидайте новые функции для управления счетом, передовой дизайн для интуитивного пользования и значительное улучшение скорости приложения. Присоединитесь довольных клиентов БК Олимп и радуйтесь ставкам где угодно и когда угодно. Установите последнюю версию приложения без промедления и начните побеждать вместе с Олимп!
С энтузиазмом сообщаем о запуске нового мобильного приложения БК Олимп для Android устройств! Этот шаг значительно улучшает ваше прежнее взаимодействие с ставками, делая его проще и быстрым. [url=https://skachat-olimp-app.ru/]Приложение Олимп бет[/url] уже сегодня! В новой версии приложения пользователи обретут доступ без ограничений к широкому спектру спортивных соревнований через свой смартфон. Улучшенное управление аккаунтом, передовой дизайн для легкого навигации и значительное ускорение работы приложения – всё это ждет вас. Присоединяйтесь к рядам удовлетворенных пользователей и наслаждайтесь от ставок где угодно и всегда. Получите обновление приложения БК Олимп прямо сейчас и откройте для себя новый уровень игры!
звуковое оборудование в аренду [url=https://www.zvukovoe-oborudovanie11.ru/]https://www.zvukovoe-oborudovanie11.ru/[/url] .
С великим удовольствием делимся новостью о выпуске новаторской версии мобильного приложения от БК Олимп для Android! Это обновление преобразит ваш подход к ставкам на спорт, делая процесс более гладким и эффективным. [url=https://olimpbet-apk.ru/]Программа Олимп[/url] и вы получите доступ без труда к широкому ассортименту спортивных мероприятий, открытых для ставок прямо с вашего мобильного. Усовершенствованные функции управления профилем, интуитивный дизайн для легкости использования и значительное повышение скорости приложения обещают непревзойденный опыт. Становитесь частью сообществу довольных клиентов и делайте ставки с удовольствием в любом месте, в любое время. Установите последнюю версию приложения БК Олимп немедленно и выходите на новый уровень игры с комфортом и стилем!
Почувствовать себя сексуальным гигантом и забывать про любые условности в постели помогут проститутки из Москвы. Для них просьба клиента — закон, а главная миссия — полное удовлетворение самых смелых желаний. Найти лучших путан можно на сайте [url=https://newswomanblog.win/]newswomanblog.win[/url], где представлены сотни подробных анкет. В каждой указаны услуги, расценки и даны подробные фото девушек. Там же можно уточнить, где произойдет встреча с проституткой — в ее уютных апартаментах или она приедет по вызову в гости.
Давно хотели попробовать что-то новое в сексе, но вам негде реализовать свои желания? Проблема легко решится, если обратиться к проститутке с сайте [url=https://newswomanblog.win/]newswomanblog.win[/url]. У большинства из представленных здесь девушек есть уютные квартиры в Москве, куда можно приехать по предварительной договоренности. На месте вы сможете принять душ, а потом расположиться на большом ложе для получения наслаждений. Путана поможет открыть новые горизонты удовольствий и научит новым видам ласк. С ней не будет скучно и захочется повторить этот яркий опыт.
I have no words to describe how your content illuminated my day. Keep being that source of inspiration!
I have no words to describe how your content illuminated my day. Keep being that source of inspiration!
В Москве сейчас не снимешь проститутку у дороги. Зато это можно сделать на сайте [url=https://newswomanblog.win/]newswomanblog.win[/url], где собраны анкеты путан с номерами телефонов и фотографиями. Девушки не будут задавать вопросов и не попросят идти с ними на свидание. Чтобы заняться с ними сексом, достаточно уточнить список услуг и договориться о цене. Можно получить удовольствие на своей территории или приехать в роскошные апартаменты. Минимальное время наедине с проституткой — один час. А если вам этого мало, путана скрасит ваш досуг в течение ночи.
incredible sunrise starting with a tremendous reading 🌞📚
seo аудит сайта стоимость [url=https://www.prodvizhenie-sajtov15.ru]https://www.prodvizhenie-sajtov15.ru[/url] .
[URL=https://1matematiki.ru/ ]Купить диплом в пензе[/URL] – Таков способ достать официальный документ о завершении образовательного учреждения. Свидетельство раскрывает двери к последующим карьерным возможностям и карьерному развитию.
Подобрали для вас с полезными и актуальными статьями по [url=https://comfort-potok.ru/]недвижимости[/url]
На нашем сайте вы можете узнать с такими темами как [url=https://comfort-potok.ru/category/rynok-nedvizhimosti/]РЫНОК НЕДВИЖИМОСТИ[/url] и [url=https://comfort-potok.ru/]СТОИМОСТЬ ЯЧЕЙКИ В СБЕРБАНКЕ[/url]
Фантастический сервисный центр [url=https://servispylesosydajson.ru/]servispylesosydajson.ru[/url] предоставил мне незабвенный опыт. Эксперты центра быстро и качественно диагностировали и исправили проблему со моим пылесосом Dyson.
Далее, я был глубоко впечатлен качеством сервиса, предоставленным сервисным центром. Они проявили особую заботу ко всем моим потребностям и предоставили ценные рекомендации по уходу за моим пылесосом.
[url=http://vashdom-istra.ru/]Истринская недвижимость[/url] – особенности рынка, перспективы развития и актуальные цены на жилье
Земля, камень, архитектура… Родившаяся в глубине времени, недвижимость обняла все уголки Истры, став неотъемлемой частью истории этого удивительного места. Каждое здание, каждый уголок этого города переплетен с безупречным чувством старины, и точно так же отражает современные требования и возможности.
Этот удивительный город, облаченный в одеяние природной красоты, восхищает своим многообразием характеров и возможностей для приобретения недвижимости. Грандиозная возможность вложиться в будущее – вот что предлагает Истра со своими замечательными домами, квартирами и усадьбами. Этот уникальный регион просто манит своим обаянием и пленит сердца своей уникальной энергией.
Архитектурные шедевры, окутанные веками тайн, привержены высоким стандартам качества и надежности. Монументальные фасады, сияющие оконные рамы и уютные дворики превращаются в сказочные кадры, которые оживают внутри уникального образа различных зданий. Величественные, но при этом привлекательно скрытые в зелени, дома Истры становятся объектом страсти для тех, кто ценит красоту и великолепие, но не готов отказаться от современного комфорта и функциональности.
Ваша скидка 10% на [url=http://vashdom-istra.ru/]юриста-консультанта по недвижимости в Истре[/url]
outstanding morning starting with an excellent literature 🌞📰
механизированная штукатурка цена [url=https://www.mekhanizirovannaya-shtukaturka11.ru/]https://www.mekhanizirovannaya-shtukaturka11.ru/[/url] .
wow, amazing
Официальный сервис Dyson выполняет эффективный сервис всей линейки устройств этого премиального бренда. Опытные специалисты оперативно выявят проблему и устранят неполадки, применяя детали от производителя. Подробности об услугах на сайте: [url=https://remontdyson.ru/]remontdyson.ru[/url].
Сервисный центр Stinol – проверенный помощник в обслуживании бытовой техники. Квалифицированные мастера быстро решат любые неполадки, возвращая вашей технике идеальное функционирование. Поручите обслуживание экспертам Stinol и наслаждайтесь безотказной работой своих приборов. Следите за нами в социальных сетях, чтобы не пропустить актуальной информации и специальных предложений!
[url=https://ewuradyficom.gumroad.com/l/remont-stinol]gumroad.com[/url]
[url=https://remont-stinol.mystrikingly.com]mystrikingly.com[/url]
[url=https://amarafot1982.wixsite.com/—————-stin]wix.com[/url]
доставка цветов саратов круглосуточно [url=https://flowerssaratov.ru]https://flowerssaratov.ru[/url] .
Thank you, Loads of posts.
article source [url=https://shallbd.com/zh/liao-jie-gui-shu-ri-he-shou-yu-ri-gui-shu-he-shou-yu-xiang-jie/]click this link[/url]
their website [url=https://shallbd.com/pt/entendendo-a-media-movel-de-30-dias-como-funciona-e-seu-significado/]full article[/url]
[url=https://teplica-teplourala.ru]Теплицу купить.[/url]
[url=https://teplica-teplourala.ru]Теплицы из поликарбоната от производителя цены.[/url]
[url=https://teplica-teplourala.ru]Купить новую теплицу.[/url]
This is nicely said. .
continued [url=https://shallbd.com/pt/entendendo-a-regra-dos-10-de-acionistas-para-iso-tudo-o-que-voce-precisa-saber/]click to find out more[/url]
learn the facts here now [url=https://shallbd.com/ko/haibeurideu-teureideuran-mueosingayo-gaenyeom-mic-ijeom-ihae/]visit their website[/url]
[URL=https://goznak-diplommy.com/ ]Купить диплом о среднем техническом образовании[/URL] – Это обрести официальный бумага о среднеобразовательном образовании. Диплом гарантирует доступ к расширенному спектру профессиональных и образовательных возможностей.
wow, amazing
nice content!nice history!! boba 😀
Helpful tips. With thanks!
discover this info here [url=https://shallbd.com/id/memahami-arti-penting-warna-biru-dalam-forex-mengeksplorasi-efek-dan-maknanya/]browse around this site[/url]
click now [url=https://shallbd.com/pt/entendendo-o-conceito-de-uma-opcao-de-venda-longa-na-negociacao-de-opcoes/]have a peek at these guys[/url]
nice content!nice history!! boba 😀
hello
открытие замков без повреждений [url=https://www.azs-zamok13.ru]https://www.azs-zamok13.ru[/url] .
I seriously love your blog.. Very nice colors & theme. Did you create this web site yourself? Please reply back as I’m hoping to create my very own blog and want to know where you got this from or what the theme is called. Thanks!
After looking over a few of the blog posts on your website, I honestly like your way of blogging. I book marked it to my bookmark website list and will be checking back soon. Take a look at my web site as well and let me know what you think.
This is a topic that’s close to my heart… Many thanks! Where are your contact details though?
Fantastic goods from you, man. I’ve understand your stuff previous to and you’re just too great. I really like what you’ve acquired here, really like what you’re stating and the way in which you say it. You make it entertaining and you still take care of to keep it sensible. I cant wait to read far more from you. This is actually a wonderful website.
whoah this blog is fantastic i love reading your articles. Stay up the good work! You recognize, a lot of individuals are searching around for this info, you can help them greatly.
I was pretty pleased to discover this website. I want to to thank you for your time due to this wonderful read!! I definitely liked every little bit of it and I have you book marked to check out new things on your web site.
Hi there i am kavin, its my first time to commenting anywhere, when i read this article i thought i could also make comment due to this brilliant article.
If you desire to get much from this piece of writing then you have to apply such techniques to your won blog.
Wow, marvelous blog layout! How long have you been blogging for? you make blogging look easy. The overall look of your web site is fantastic, let alone the content!
Very rapidly this web site will be famous among all blogging viewers, due to it’s pleasant articles or reviews
Hurrah! At last I got a website from where I be able to really take useful data regarding my study and knowledge.
What a information of un-ambiguity and preserveness of precious familiarity about unexpected feelings.
I’ve read several just right stuff here. Definitely worth bookmarking for revisiting. I wonder how so much attempt you put to create any such fantastic informative site.
Way cool! Some very valid points! I appreciate you writing this article and also the rest of the site is extremely good.
Thanks for finally writing about > %blog_title% < Liked it!
I’m impressed, I must say. Rarely do I encounter a blog that’s both educative and engaging, and let me tell you, you have hit the nail on the head. The issue is something not enough people are speaking intelligently about. I am very happy that I stumbled across this in my search for something concerning this.
I want to to thank you for this fantastic read!! I definitely enjoyed every little bit of it. I have you bookmarked to check out new stuff you post
This is my first time visit at here and i am truly impressed to read all at alone place.
of course like your website however you need to check the spelling on quite a few of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come back again.
Hi there, just wanted to mention, I enjoyed this post. It was funny. Keep on posting!
Awesome site you have here but I was wanting to know if you knew of any discussion boards that cover the same topics talked about in this article? I’d really love to be a part of group where I can get suggestions from other knowledgeable individuals that share the same interest. If you have any recommendations, please let me know. Appreciate it!
Повышение квалификации медицинских работников является важной частью профессионального развития и поддержания высокого уровня оказания медицинских услуг. Программы, представленные на сайте maps-edu.ru, охватывают широкий спектр направлений, включая новейшие методики диагностики и лечения, управление в здравоохранении, медицинскую этику и право. Курс предусматривает как теоретическую подготовку, так и практические навыки, необходимые для успешной работы в современных медицинских учреждениях. Участники программы получат знания, которые позволят им не только улучшить качество медицинской помощи, но и повысить свою конкурентоспособность на рынке труда, развивая личностные и профессиональные качества.
Обновите свои знания: курсы повышения квалификации для педагогов и [url=https://maps-edu.ru/catalog/logopediia?type=professionalnaya-perepodgotovka]профессиональная переподготовка на логопеда[/url] .
Nice blog here! Also your website loads up fast! What host are you using? Can I get your affiliate link to your host? I wish my site loaded up as fast as yours lol
I love your blog.. very nice colors & theme. Did you design this website yourself or did you hire someone to do it for you? Plz reply as I’m looking to create my own blog and would like to know where u got this from. cheers
Hi, i think that i saw you visited my blog so i got here to go back the choose?.I am trying to in finding things to improve my website!I guess its good enough to use some of your ideas!!
I’ve been exploring for a little bit for any high-quality articles or blog posts in this kind of space . Exploring in Yahoo I eventually stumbled upon this web site. Reading this info So i’m satisfied to express that I have a very good uncanny feeling I came upon exactly what I needed. I such a lot for sure will make certain to don?t put out of your mind this web site and give it a look on a continuing basis.
Thanks for your personal marvelous posting! I definitely enjoyed reading it, you’re a great author. I will make sure to bookmark your blog and definitely will come back in the future. I want to encourage yourself to continue your great posts, have a nice afternoon!
Hello! I could have sworn I’ve been to this web site before but after browsing through a few of the posts I realized it’s new to me. Nonetheless, I’m definitely pleased I found it and I’ll be bookmarking it and checking back regularly!
Your style is very unique compared to other people I have read stuff from. Thank you for posting when you have the opportunity, Guess I will just bookmark this web site.
I read this article fully concerning the resemblance of latest and previous technologies, it’s awesome article.
I all the time emailed this website post page to all my associates, because if like to read it then my links will too.
I am extremely impressed with your writing skills and also with the layout on your blog. Is this a paid theme or did you customize it yourself? Either way keep up the nice quality writing, it’s rare to see a nice blog like this one these days.
This is very fascinating, You are an overly professional blogger. I have joined your feed and sit up for in quest of more of your fantastic post. Also, I have shared your web site in my social networks
It’s an remarkable piece of writing in favor of all the web users; they will take benefit from it I am sure.
Hiya! I know this is kinda off topic however I’d figured I’d ask. Would you be interested in exchanging links or maybe guest writing a blog article or vice-versa? My site discusses a lot of the same subjects as yours and I believe we could greatly benefit from each other. If you are interested feel free to send me an e-mail. I look forward to hearing from you! Superb blog by the way!
I’m impressed, I must say. Rarely do I encounter a blog that’s equally educative and entertaining, and let me tell you, you have hit the nail on the head. The issue is something which not enough people are speaking intelligently about. I’m very happy that I found this in my search for something concerning this.
Heya i’m for the first time here. I came across this board and I find It truly useful & it helped me out a lot. I hope to give something back and help others like you helped me.
Write more, thats all I have to say. Literally, it seems as though you relied on the video to make your point. You clearly know what youre talking about, why waste your intelligence on just posting videos to your site when you could be giving us something enlightening to read?
This website really has all of the info I wanted about this subject and didn’t know who to ask.
I’m gone to say to my little brother, that he should also pay a visit this webpage on regular basis to take updated from most up-to-date gossip.
I quite like reading a post that will make people think. Also, thanks for allowing for me to comment!
I am extremely impressed with your writing skills and also with the layout on your blog. Is this a paid theme or did you customize it yourself? Either way keep up the nice quality writing, it’s rare to see a nice blog like this one nowadays.
Ahaa, its pleasant discussion concerning this article here at this blog, I have read all that, so now me also commenting here.
whoah this blog is great i really like reading your articles. Stay up the good work! You realize, a lot of people are hunting around for this info, you can help them greatly.
Sweet blog! I found it while surfing around on Yahoo News. Do you have any tips on how to get listed in Yahoo News? I’ve been trying for a while but I never seem to get there! Many thanks
Thank you for sharing your info. I truly appreciate your efforts and I am waiting for your next post thank you once again.
I love what you guys are usually up too. Such clever work and coverage! Keep up the very good works guys I’ve added you guys to my blogroll.
Pretty great post. I simply stumbled upon your blog and wanted to mention that I have really enjoyed browsing your blog posts. In any case I’ll be subscribing in your feed and I hope you write again soon!
Good post. I am going through a few of these issues as well..
My brother suggested I might like this blog. He was totally right. This post actually made my day. You cann’t imagine just how much time I had spent for this information! Thanks!
Hi, Neat post. There is a problem together with your web site in internet explorer, might check this? IE still is the marketplace leader and a big component to other people will miss your fantastic writing due to this problem.
Appreciate the recommendation. Will try it out.
Pretty section of content. I just stumbled upon your weblog and in accession capital to assert that I acquire in fact enjoyed account your blog posts. Any way I’ll be subscribing to your augment and even I achievement you access consistently rapidly.
If you want to get a great deal from this article then you have to apply such strategies to your won website.
Simply wish to say your article is as surprising. The clearness in your publish is simply excellent and i can think you are knowledgeable in this subject. Well with your permission allow me to take hold of your RSS feed to stay up to date with imminent post. Thank you one million and please continue the gratifying work.
Howdy! I could have sworn I’ve been to this blog before but after browsing through some of the posts I realized it’s new to me. Nonetheless, I’m definitely happy I came across it and I’ll be bookmarking it and checking back frequently!
I don’t even know how I ended up here, but I thought this post was good. I don’t know who you are but definitely you are going to a famous blogger if you are not already 😉 Cheers!
This is very fascinating, You are an overly professional blogger. I have joined your feed and look ahead to in search of more of your fantastic post. Also, I have shared your site in my social networks
Appreciating the persistence you put into your site and in depth information you present. It’s awesome to come across a blog every once in a while that isn’t the same out of date rehashed material. Excellent read! I’ve saved your site and I’m including your RSS feeds to my Google account.
Good day! Do you use Twitter? I’d like to follow you if that would be ok. I’m definitely enjoying your blog and look forward to new updates.
I’ve been exploring for a little bit for any high-quality articles or blog posts in this kind of area . Exploring in Yahoo I at last stumbled upon this site. Reading this info So i’m satisfied to show that I have a very good uncanny feeling I came upon exactly what I needed. I so much without a doubt will make certain to don?t disregard this web site and give it a look on a continuing basis.
Have you ever considered about including a little bit more than just your articles? I mean, what you say is valuable and all. Nevertheless think of if you added some great graphics or video clips to give your posts more, “pop”! Your content is excellent but with images and video clips, this site could certainly be one of the most beneficial in its niche. Awesome blog!
This design is steller! You most certainly know how to keep a reader entertained. Between your wit and your videos, I was almost moved to start my own blog (well, almost…HaHa!) Fantastic job. I really enjoyed what you had to say, and more than that, how you presented it. Too cool!
I needed to thank you for this excellent read!! I definitely enjoyed every little bit of it. I have you book marked to check out new stuff you post
This piece of writing is in fact a pleasant one it helps new net viewers, who are wishing for blogging.
These are really great ideas in about blogging. You have touched some pleasant factors here. Any way keep up wrinting.
Asking questions are actually nice thing if you are not understanding anything entirely, but this article provides pleasant understanding even.
I know this if off topic but I’m looking into starting my own blog and was wondering what all is required to get set up? I’m assuming having a blog like yours would cost a pretty penny? I’m not very internet savvy so I’m not 100% positive. Any recommendations or advice would be greatly appreciated. Cheers
Thank you for sharing your info. I truly appreciate your efforts and I am waiting for your next post thank you once again.
It is perfect time to make a few plans for the longer term and it is time to be happy. I have read this publish and if I may I wish to suggest you few interesting things or suggestions. Perhaps you could write next articles referring to this article. I want to read more things approximately it!
Hey there, You have performed a fantastic job. I will definitely digg it and personally recommend to my friends. I am sure they will be benefited from this site.
There is definately a lot to know about this topic. I like all the points you’ve made.
naturally like your website however you need to check the spelling on quite a few of your posts. Several of them are rife with spelling problems and I in finding it very bothersome to tell the truth then again I will certainly come back again.
I used to be recommended this website by means of my cousin. I am not positive whether this post is written by means of him as no one else realize such specific approximately my problem. You are amazing! Thank you!
Hi, just wanted to say, I enjoyed this article. It was inspiring. Keep on posting!
It’s very straightforward to find out any topic on net as compared to books, as I found this post at this web site.
Wow that was strange. I just wrote an really long comment but after I clicked submit my comment didn’t show up. Grrrr… well I’m not writing all that over again. Anyway, just wanted to say wonderful blog!
Hey There. I found your blog using msn. This is a very well written article. I will be sure to bookmark it and come back to read more of your useful information. Thanks for the post. I will definitely comeback.
Wow, superb blog layout! How long have you been blogging for? you make blogging glance easy. The total glance of your web site is fantastic, let alonewell as the content!
Hi there! I know this is kind of off topic but I was wondering if you knew where I could get a captcha plugin for my comment form? I’m using the same blog platform as yours and I’m having trouble finding one? Thanks a lot!
Переподготовка на учителя начальных классов через платформу maps-edu.ru обеспечивает углубленное изучение методик и подходов, необходимых для эффективного обучения и развития младших школьников. Эта программа поможет вам освоить современные образовательные технологии и практики, а также обновить и расширить свои знания в области педагогики и психологии, что является ключом к успешной работе с детьми начальной школы.
Освойте новые горизонты: [url=https://maps-edu.ru/catalog/gosudarstvennye-zakupki]курсы руководитель отдела закупок[/url] и контролю закупок.
nice content!nice history!! boba 😀
nice content!nice history!! boba 😀
Повышение квалификации медицинских работников является важной частью профессионального развития и поддержания высокого уровня оказания медицинских услуг. Программы, представленные на сайте maps-edu.ru, охватывают широкий спектр направлений, включая новейшие методики диагностики и лечения, управление в здравоохранении, медицинскую этику и право. Курс предусматривает как теоретическую подготовку, так и практические навыки, необходимые для успешной работы в современных медицинских учреждениях. Участники программы получат знания, которые позволят им не только улучшить качество медицинской помощи, но и повысить свою конкурентоспособность на рынке труда, развивая личностные и профессиональные качества.
Повысьте свои компетенции: [url=https://maps-edu.ru/catalog/pedagogika]онлайн курсы для педагогов[/url] и муниципальными закупками.
Пройти аккредитацию для медицинского работника означает подтвердить свою профессиональную компетенцию и соответствие современным стандартам медицинской помощи. Этот процесс включает теоретическое обучение по актуальным медицинским дисциплинам, а также практические задания и тестирования для оценки знаний и умений специалиста. Успешное прохождение аккредитации не только повышает статус медицинского работника в профессиональном сообществе, но и открывает новые возможности для карьерного роста и развития.
Улучшите свою карьеру: [url=https://maps-edu.ru/catalog/pedagogika?type=professionalnaya-perepodgotovka]профессиональная переподготовка педагог[/url] и курсы логопедии доступны у нас!
nice content!nice history!! boba 😀
Курсы повышения квалификации для педагогов дополнительного образования, предлагаемые академией “МАПС”, ориентированы на расширение профессиональных компетенций учителей в сфере дополнительного образования детей и подростков. Эти программы позволяют педагогам освежить свои знания, ознакомиться с новыми методиками и подходами в обучении, что способствует более эффективному взаимодействию с учениками и повышению качества образовательного процесса.
Освойте новые горизонты: [url=https://maps-edu.ru/catalog/oxrana-truda]специалист по охране труда обучение[/url] и контролю закупок.
Wow! I recently read your article and I’m blown away. Your perspective on the topic is extremely valuable. I’ve learned so much and am eager to see what you write next. Your work is inspiring!
Wow! I just read your post and I’m blown away. Your perspective on the topic is extremely valuable. I’ve gained a new perspective and am eager to see your next post. Your work is inspiring!
wow, amazing
nice content!nice history!! boba 😀
Благодарю за потраченное время.
Представляю вам [url=https://rufilmonline.ru/melodrama/]русские сериалы мелодрамы[/url] – это невероятное произведение, которое нравится огромному количество зрителей по всему миру. Русские фильмы и сериалы предлагают уникальный взгляд на русскую культуру, историю и обычаи. Сегодня смотреть русские фильмы и сериалы онлайн стало легко благодаря различным платформам и сервисам. От ужасов до триллеров, от исторических фильмов до современных детективов – выбор безграничен. Окунитесь в захватывающие сюжеты, профессиональную актерскую работу и красивую операторскую работу, смотрите фильмы и сериалы из России не выходя из дома.
Incredible! I recently read your blog post and I’m absolutely amazed. Your perspective on the topic is extremely valuable. I’ve gained a new perspective and am eager to see what you write next. Thanks for sharing!
1. Вибір натяжних стель – як правильно обрати?
2. Топ-5 популярних кольорів натяжних стель
3. Як зберегти чистоту натяжних стель?
4. Відгуки про натяжні стелі: плюси та мінуси
5. Як підібрати дизайн натяжних стель до інтер’єру?
6. Інноваційні технології у виробництві натяжних стель
7. Натяжні стелі з фотопечаттю – оригінальне рішення для кухні
8. Секрети вдалого монтажу натяжних стель
9. Як зекономити на встановленні натяжних стель?
10. Лампи для натяжних стель: які вибрати?
11. Відтінки синього для натяжних стель – ексклюзивний вибір
12. Якість матеріалів для натяжних стель: що обирати?
13. Крок за кроком: як самостійно встановити натяжні стелі
14. Натяжні стелі в дитячу кімнату: безпека та креативність
15. Як підтримувати тепло у приміщенні за допомогою натяжних стель
16. Вибір натяжних стель у ванну кімнату: практичні поради
17. Натяжні стелі зі структурним покриттям – тренд сучасного дизайну
18. Індивідуальність у кожному домашньому інтер’єрі: натяжні стелі з друком
19. Як обрати освітлення для натяжних стель: поради фахівця
20. Можливості дизайну натяжних стель: від класики до мінімалізму
ціна натяжних стель [url=https://natjazhnistelitvhyn.kiev.ua/]https://natjazhnistelitvhyn.kiev.ua/[/url] .
Семейное гнездышко в окружении природы
18. Современный дом из бруса 9х12: дизайнерские решения
проект дома из бруса одноэтажный 9х12 [url=https://domizbrusa9x12spb.ru/]https://domizbrusa9x12spb.ru/[/url] .
Incredible! I just finished reading your article and I’m blown away. Your insight on the topic is incredibly insightful. I’ve gained a new perspective and am eager to read more. Keep up the great work!
Incredible! I just finished reading your post and I’m thoroughly impressed. Your perspective on the topic is incredibly insightful. I’ve learned so much and am eager to see what you write next. Thanks for sharing!
phising
Amazing! I just finished reading your post and I’m blown away. Your insight on this subject is spot-on. I’ve learned so much and am eager to see what you write next. Keep up the great work!
воєнторг
14. Военные товары высокого качества
військовий одяг інтернет магазин [url=https://voentorgaseh.kiev.ua/]військові магазини в києві[/url] .
1. Почему берцы – это обязательный элемент стиля?
2. Как выбрать идеальные берцы для осеннего гардероба?
3. Тренды сезона: кожаные берцы или замшевые?
4. 5 способов носить берцы с платьем
5. Какие берцы выбрать для повседневного образа?
6. Берцы на платформе: комфорт и стиль в одном
7. Какие берцы будут актуальны в этом году?
8. Маст-хэв сезона: военные берцы в стиле милитари
9. 10 вариантов сочетания берцов с джинсами
10. Зимние берцы: как выбрать модель для холодного сезона
11. Элегантные берцы на каблуке: идеальный вариант для офиса
12. Секреты ухода за берцами: как сохранить первоначальный вид?
13. С какой юбкой носить берцы: советы от стилистов
14. Как подобрать берцы под фасон брюк?
15. Берцы на шнуровке: стильный акцент в образе
16. Берцы-челси: универсальная модель для любого стиля
17. С чем носить берцы на плоской подошве?
18. Берцы с ремешками: акцент на деталях
19. Как выбрать берцы для прогулок по городу?
20. Топ-5 брендов берцев: качество и стиль в одном
берці військові [url=https://bercifrdt.kiev.ua/]купити берці військові[/url] .
Revolutionize Your Plastic Processing Business with 18ps.ru! Discover cutting-edge solutions for plastic recycling and production equipment. From transforming plastic waste into valuable granules to crafting polymer sand products, our comprehensive range of equipment ensures efficiency and sustainability. Explore our selection today and elevate your production capabilities with top-quality machinery at competitive prices. Whether you’re starting a new venture or upgrading your existing facility, trust 18ps.ru for reliable equipment and unparalleled service. Maximize your ROI and minimize environmental impact – join the plastic processing revolution now!
Eng.18ps.ru – [url=https://eng.18ps.ru/catalog/equipment-for-processing-plastic-and-glass/]buy a plastic processing machine[/url]
lost money
Wow! I recently read your article and I’m blown away. Your perspective on the topic is incredibly insightful. I’ve learned so much and can’t wait to see what you write next. Thanks for sharing!
Amazing! I just read your article and I’m absolutely amazed. Your insight on the topic is incredibly insightful. I’ve gained a new perspective and am eager to read more. Thanks for sharing!
scam
lost money
phising
Ремонт ноутбуков Lenovo – одно из основных направлений деятельности сервисного центра https://servisnyjcentr-lenovo.ru/. Наши сертифицированные инженеры обладают глубокими знаниями и богатым опытом в обслуживании техники этого бренда. Мы осуществляем ремонт любой сложности: от замены неисправных деталей до модернизации железа и настройки программного обеспечения под ваши требования. В арсенале наших мастеров – профессиональное оборудование и инструменты, которые позволяют качественно выполнять работы по ремонту ноутбуков Lenovo всех серий и моделей. Мы внимательно следим за технологиями и регулярно повышаем квалификацию специалистов.
[url=https://servisnyjcentr-lenovo.ru/]сервисный центр lenovo[/url]
замена батареи в ноутбуке леново
замена материнской платы ноутбука lenovo
леново сервис
Использование виртуальной и дополненной реальности arigami.tech
По вопросу [url=https://arigami.tech/3d_widget]интерактивные виджеты[/url] Вы на верном пути. Мы даем гарантию на отличное реалистичное качество после перехода его в 3D формат. Весьма просто будет взаимодействовать с виджетом, меняя в настоящем времени модели, цвета, размеры. Особенный подход к продажам может дать Вам известность и исключительность в данной сфере. Многие покупатели подчеркивают, что осуществлять покупки интереснее и проще с подбором при просмотре 3D моделей.
[URL=demo.4admins.ru/viewtopic.php?f=10&t=1549 ]купить диплом во владивоÑтоке[/URL] – Таков способ обрести официальный документ по завершении образовательного учреждения. Диплом раскрывает двери к дополнительным карьерным возможностям и карьерному развитию.
scam
scam
phising
lost money
Применение технологий дополненной реальности arigami.tech
По запросу [url=https://arigami.tech/augmented_reality]разработка приложений виртуальной и дополненной реальности[/url] Вы на верном пути. Мы даем гарантию на высокое реалистичное качество после перехода его в 3D вид. Весьма легко будет взаимодействовать с виджетом, меняя в настоящем времени вариации, цвета, габариты. Уникальный путь к продажам может дать Вам узнаваемость и исключительность в данной сфере. Многие покупатели отмечают, что осуществлять покупки интереснее и быстрее с выбором при просмотре 3D виджетов.
Excellent write-up
Amazing! I recently read your blog post and I’m thoroughly impressed. Your analysis on this topic is extremely valuable. I’ve gained a new perspective and am eager to see your next post. Your work is inspiring!
Создание товаров в 3D arigami.tech
По теме [url=https://arigami.tech/augmented_reality]виртуальная и дополненная реальность в маркетинге[/url] Вы на правильном пути. Мы даем гарантию на отличное реалистичное качество после перехода его в 3D вид. Весьма легко будет коммуницировать с виджетом, меняя в реальном времени вариации, цвета, габариты. Оригинальный путь к продажам может дать Вам известность и исключительность в данной сфере. Многие покупатели подчеркивают, что осуществлять покупки интереснее и проще с подбором при просмотре 3D моделей.
phising
[url=https://ru.pinterest.com/rega0589/]Волна Усиленная теплица от производителя[/url].
[url=https://ru.gravatar.com/teplica52]теплица52 Теплица из поликарбоната купить[/url].
[url=https://www.vo5.org/news/vybor-nadezhnyh-teplits]Теплица из поликарбоната под заказ[/url].
[url=http://postroyka.org/sovetyi-kak-vyibrat-teplitsu-iz-polikarbonata/]Распродажа теплиц[/url].
[url=https://beacons.ai/teplica52]Теплица из поликарбоната ширина 2[/url].
[url=https://kwork.ru/user/sasha818181]teplica52.ru Теплица из поликарбоната цена[/url].
[url=https://www.strata.com/forums/users/teplica52/]Купить теплицу дешево teplica52[/url].
[url=https://www.weblancer.net/users/Teplica52/]Теплицы прямостенные из поликарбоната купить Волна и ВИТА[/url].
[url=http://postroyka.org/sovetyi-kak-vyibrat-teplitsu-iz-polikarbonata/]Стоимость теплицы[/url].
[url=https://github.com/Teplica52]Теплицы 2 5 м ширина[/url].
[url=https://www.drupalgovcon.org/user/479466]Теплица на заказ цена teplica52[/url].
[url=http://postroyka.org/sovetyi-kak-vyibrat-teplitsu-iz-polikarbonata/]Продажа теплиц из поликарбоната Теплица52[/url].
[url=https://lit.link/en/teplica52]Теплицы из поликарбоната распродажа от производителя[/url].
[url=https://speakerdeck.com/teplica52]Теплицы из поликарбоната купить н новгород[/url].
[url=http://postroyka.org/sovetyi-kak-vyibrat-teplitsu-iz-polikarbonata/]Купить теплицу из поликарбоната дешево[/url].
[url=https://hub.docker.com/u/teplica52]Продажа теплиц из поликарбоната[/url].
[url=https://www.pinterest.co.uk/rega0589/]Волна Теплица из поликарбоната под заказ[/url].
[url=https://dzerjinsk.ru/news/gde-zakazat-sovremennye-polikarbonatnye-teplitsy-dlya-ovoshchei-i-yagod]теплица52 Усиленные теплицы из поликарбоната от производителя купить[/url].
[url=https://www.rohitab.com/discuss/user/1200603-теплицы-волна-Рё-вита/]Теплицы прямостенные усиленные[/url].
[url=https://repo.getmonero.org/teplica52]Стоимость теплицы из поликарбоната[/url].
[url=https://linktr.ee/teplica52]Теплицы из поликарбоната от производителя цены[/url].
[url=https://ru.pinterest.com/rega0589/]teplica52.ru Купить теплицу от производителя[/url].
[url=https://ru.gravatar.com/teplica52]Теплицы из поликарбоната по низким ценам teplica52[/url].
[url=https://www.sqlservercentral.com/forums/user/teplica52]Купить теплицу усиленную Волна и ВИТА[/url].
[url=https://beacons.ai/teplica52]Распродажа теплиц[/url].
[url=https://wyksa.ru/2020/07/30/kak-sdelat-teplicu-iz-polikarbonata-svoimi-rukami.html]Купить теплицу в рассрочку[/url].
[url=https://disqus.com/by/teplica52/about/]Купить теплицу с установкой teplica52[/url].
[url=https://www.reddit.com/user/Teplica52/]Теплицы из поликарбоната распродажа от производителя Теплица52[/url].
[url=https://ogorodnikis.ru/vse-dlja-sada-i-ogoroda/teplica-iz-polikarbonata-vita-premium.html]Купите дачную теплицу[/url].
[url=http://kfaktiv.ru/sovety-po-vyboru-teplicy-iz-polikarbonata.html]Теплицы от производителя акции распродажи[/url].
[url=https://public-herring.profeat.site]Теплицы цена доставка[/url].
[url=https://www.reddit.com/user/Effective-Pop2661/comments/okz7qv/как_РЅРµ_нужно_делать_сетку_для_огурцов_РІ_теплицу/]Заказ теплиц[/url].
scam
scam
Эволюция бренда
16. BMW 7 Series: роскошь и инновации в одном автомобиле
x5 bmw 2023 [url=https://avtosalonbmwftnz.dp.ua/]x6 2023[/url] .
Fantastic! I recently read your blog post and I’m absolutely amazed. Your perspective on the topic is extremely valuable. I’ve learned so much and can’t wait to see what you write next. Thanks for sharing!
Разработка vr приложений в Москве w2w.group
По запросу [url=https://w2w.group/solutions]разработка ar приложений заказать[/url] Вы на верном пути. Наши консультанты готовы дать обратную связь и ответить на любые вопросы. Если у Вас есть личные образцы, которые нужно дополнить реальностью, пришлите их нам и мы сумеем понять, годятся ли они. Если их не существует, мы сами готовы их воплотить, учитывая все Ваши желания. Оформление личного сайта с помощью виртуальной и дополненной реальности не только удерживает клиента, но и даёт оценить показатель подхода к ведению бизнеса. Тем самым Вы можете отличиться среди других на рынке и громко заявить о себе.
Incredible! I just finished reading your post and I’m absolutely amazed. Your analysis on this topic is spot-on. I’ve gained a new perspective and can’t wait to see what you write next. Thanks for sharing!
lost money
Fantastic! I just finished reading your post and I’m thoroughly impressed. Your insight on this topic is extremely valuable. I’ve gained a new perspective and can’t wait to see your next post. Keep up the great work!
lost money
lost money
Холодильник Stinol baraxlit? Звоните в [url=https://stinol-servise.ru/fridge/thermostat/]stinol-servise.ru[/url], и наши мастера быстро вернут его в рабочее состояние.
сервисный центр стинол
сервисный центр стинол москва
замена вентилятора в холодильнике stinol
Разработка vr приложений в Москве w2w.group
Относительно [url=https://w2w.group/solutions]разработка vr приложений стоимость[/url] мы Вам обязательно окажем помощь. Вот уже более 6ти лет мы трудимся в данной сфере, имеем множество положительных отзывов и радостных покупателей, также успели воплотить более сотни успешных проектов и всё с помощью новейших современных технологий. Ознакомиться с портфолио возможно также на вышеупомянутом веб портале.
лазерная сварка купить ручной аппарат цена [url=http://apparat-ruchnoy-lazernoy-svarki.ru/]http://apparat-ruchnoy-lazernoy-svarki.ru/[/url] .
Разработка vr приложений в Москве w2w.group
По поводу [url=https://w2w.group/solutions]разработка приложений дополненной реальности услуги[/url] мы Вам непременно поможем. Вот уже свыше 6ти лет мы реализуем работу в представленной сфере, имеем много положительных отзывов и счастливых клиентов, также успели воплотить более 120ти отличных проектов и всё с помощью новых современных технологий. Ознакомиться с галереей работ можно также на вышеупомянутом веб портале.
Мобильное приложение дополненной реальности w2w.group
По поводу [url=https://w2w.group/solutions]ar разработка[/url] мы Вам обязательно окажем помощь. Вот уже свыше 6ти лет мы реализуем работу в представленной сфере, имеем множество положительных комментариев и довольных покупателей, также успели реализовать более 120ти отличных проектов и всё с помощью новейших прогрессивных технологий. Ознакомиться с галереей работ можно также на вышеупомянутом сайте.
Fantastic! I just read your blog post and I’m absolutely amazed. Your perspective on the topic is incredibly insightful. It really made me think and am eager to see your next post. Keep up the great work!
алюминиевый плинтус для пола [url=https://alyuminievyj-plintus-msk.ru/]плинтус с кабель каналом[/url] .
1. Как выбрать идеальный гипсокартон для ремонта
строительные материалы купить [url=https://gipsokarton-moskva.ru/]стяжка пола[/url] .
Fantastic! I recently read your post and I’m thoroughly impressed. Your insight on this topic is incredibly insightful. I’ve learned so much and can’t wait to see what you write next. Your work is inspiring!
Wow! I recently read your post and I’m blown away. Your perspective on this subject is spot-on. I’ve gained a new perspective and am eager to see your next post. Thanks for sharing!
Вывоз старой мебели ecologia-t.ru
По вопросу [url=https://ecologia-t.ru/info/vivoz_musora_konteinerom_8m3/]вывоз мусора контейнер 8 кубов москва[/url] Вы на правильном пути. Вывозим отходы разного вида: смешанные отходы, твердые бытовые отходы, макулатуру, грунт, снег и подобные. Также проводим работу 24 часа в сутки, без выходных и даже в праздничные дни. Делаем как разовые вывозы, так и ежедневные по определенным дням или часам, в зависимости от планов клиента. Подходим к любой проблеме персонально, чтобы выполнять работу супер качественно.
I will right away seize your rss as I can not in finding your email subscription link or newsletter service. Do you have any? Please allow me recognise so that I may subscribe. Thanks.
купить диплом магистра [url=https://www.1diplomy-grupp.ru]https://www.1diplomy-grupp.ru[/url] .
[URL=https://www.global-telecom.ru/astr/ ]Нея диплом купить[/URL] постановляется заданием для многих, кому встречается с потребностью иметь законное подтверждение об образовании.
Текущие технологии или развитие интернет-рынка позволяют найти массу возможностей для покупки документа.
Однако, выбор надежного провайдера превращается ключевым фактором этого процесса.
Часть фирмы продают изготовление аттестатов с минимальными затратами от заказчика, впрочем не всегда качество таких документов соответствует.
Важно выбирать гарантированные и рекомендованные поставщики, где доступно приобрести аттестат с уверенным качественной степенью и подлинностью.
Одновременно, нужно учитывать не только стоимость, но имидж компании, рецензии клиентов и возможность получить консультацию эксперта до покупкой.
Адекватный выбор поможет предотвратить нежелательных последствий действий и гарантирует достоверность в своем имеющейся требуемого документа.
купить коляску для [url=https://detskie-koljaski-moskva.ru/]коляски прогулочные[/url] .
Превосходное впечатление получил, посетив сервисный центр техники Zanussi, который располагается по ссылке [url=https://zanussi-rem.ru/]zanussi ремонт[/url]. Команда центра продемонстрировали высочайший класс профессионализма, осуществив быстрый и точный ремонт моей посудомоечной машины.
Мастера центра, в силу своей квалификации и богатому опыту, точно сумели диагностировать проблему и устранить ее в кратчайшие сроки. Дополнительно, я был приятно удивлен отличным обслуживанием и разумными ценами на услуги.
Без сомнения, сервисный центр техники Zanussi является надежным выбором для тех, кто ценит качество и эффективность. Сердечно рекомендую этот сервисный центр всем, кто столкнулся с проблемами со своей техникой Zanussi.
Професійні рекомендації
12. Як зберегти здоров’я зубів у період вагітності
ваша стоматологія [url=https://stomatologiyatrn.ivano-frankivsk.ua/]ваша стоматологія[/url] .
1. Идеи для дизайна интерьера
2. Топ-20 трендов в дизайне на 2021 год
3. Цветовые решения
4. Дизайн-проект
5. Инновационные подходы к дизайну: отражение современности
6. Шаг за шагом: создание уютного дизайна спальни
7. Дизайнерские решения для увеличения пространства в маленькой квартире
8. Природный дизайн
9. Баланс цветов и форм: основы хорошего дизайна
10. Дизайнерский бизнес
11. Дизайн в XXI веке
12. Дизайн кухни
13. Тенденции в сфере дизайна мебели: вдохновляющие идеи
14. Дизайн гостиной
15. Искусство минимализма: создание современного дизайна в своем доме
16. Дизайн сада: принципы оформления участка с учетом ландшафта
17. Декорирование дома с использованием текстиля: советы и идеи
18. Принципы цветового баланса в дизайне: как создать гармоничное пространство
19. Топ-10 книг по дизайну интерьера, которые стоит прочитать
20. Дизайн подростковой комнаты
дизайн интерьер [url=https://studiya-dizajna-intererov.ru/]https://studiya-dizajna-intererov.ru/[/url] .
аттестат купить окончание [url=http://www.2diplom-grupp.ru]http://www.2diplom-grupp.ru[/url] .
сплит кондиционер купить [url=https://split-sistema-kupit.ru/]сплит кондиционер купить[/url] .
Получение аттестата по высшему образованию представляется значимым действием в жизни многочисленных индивидуумов, обеспечивая возможности в свежим возможностям и путям.
Тем не менее, вовсе не все время учеба в вузе доступен или подходит из разных причин.
В таких случаях вопросик где приобрести аттестат, становится существенным.
Нынешние технологические достижения и виртуальный рынок предоставляют разнообразные пути для приобретения документа, но существенно выбирать надежных поставщиков, обеспечивающих высокое качество и подлинность документа.
При наличии отборе нужно обращать внимание не только на стоимость, но и на представление компании, мнения клиентов и возможность получения консультации.
[URL=http://newsofmebel.ru/page/2 ]http://boxer-forum.ru/topic3422.html?&p=75483 [/URL] купить диплом РІ выборге – подразумевает инвестировать в свое свое будущее, поэтому отбор поставщика рекомендуется относиться ответственно.
[URL=http://www.ezoteriklove.ru/viewtopic.php?f=5&t=3372&start=20]https://vik.clan.su/news/2011-04-24-74[/URL] – – Это способ обрести официальный удостоверение по завершении образовательного учреждения. Диплом открывает пути к последующим карьерным перспективам и профессиональному росту.
Когда моя стиральная машина Bosch начала протекать, я сразу же обратился в местный сервис. Мастер быстро приехал и обнаружил, что проблема в уплотнителе дверцы. Замена заняла немного времени, и машина снова стала как новая. Сервис оказался на высоте: все сделали быстро и качественно, дали гарантию на замененные детали. Очень удобно, что не пришлось никуда ехать — все сделали на дому.
Бош-Ремонт.рф – [url=https://xn—-9sbn2afcdnw7c.xn--p1ai/]бош сервисный центр[/url]
Мой холодильник Bosch внезапно перестал морозить, что вызвало немало хлопот. Я вызвал мастера из знакомого сервиса, и они быстро прибыли на место. Проблема оказалась в термостате, который нуждался в замене. Работа была выполнена на дому, без лишних хлопот для меня. Сервис использовал только оригинальные запчасти, что дало мне дополнительную уверенность в качестве проведенного ремонта.
Бош-Ремонт.рф – [url=https://xn—-9sbn2afcdnw7c.xn--p1ai/стиральных-машин]ремонт стиральных машин bosch на дому[/url]
casino sweet bonanza [url=http://www.sweet-bonanza-game.ru]http://www.sweet-bonanza-game.ru[/url] .
Секреты быстрой и качественной установки кондиционера
установить кондиционер [url=https://ustanovka-kondicionera-cena.ru/]https://ustanovka-kondicionera-cena.ru/[/url] .
Полезные советы
2. Шаг за шагом: установка кондиционера своими руками
3. Важные моменты при установке кондиционера в квартире
4. Специалисты или самостоятельная установка кондиционера?
5. 10 шагов к идеальной установке кондиционера
6. Подробная инструкция по установке кондиционера на балконе
7. Лучшие методы крепления кондиционера на стену
8. Как выбрать место для установки кондиционера в комнате
9. Секреты успешной установки кондиционера в частном доме
10. Рассказываем, как правильно установить сплит-систему
11. Необходимые инструменты для установки кондиционера
12. Какие документы нужны для оформления установки кондиционера?
13. Топ-5 ошибок при самостоятельной установке кондиционера
14. Установка кондиционера на потолке: особенности и нюансы
15. Когда лучше всего устанавливать кондиционер в доме?
16. Почему стоит доверить установку кондиционера профессионалам
17. Как подготовиться к установке кондиционера в жаркий сезон
18. Стоит ли экономить на установке кондиционера?
19. Подбор оптимальной мощности кондиционера перед установкой
20. Какие бывают типы кондиционеров: сравнение перед установкой
оборудование кондиционирования [url=https://prodazha-kondcionerov.ru/]оборудование кондиционирования[/url] .
кондиционер 7 [url=https://multisplit-sistemy-kondicionirovaniya.ru/]кондиционер 7[/url] .
Мне хотелось бы от всего сердца поблагодарить [url=https://taplink.cc/remontnoutbukovvbalashihe/]ремонт ноутбуков[/url] за их высококвалифицированную работу. Мой ноутбук был отремонтирован в кратчайшие сроки, и он снова функционирует безупречно. Сотрудники были вежливыми и внимательными, объяснив все детали ремонта. Я очень доволен результатом, и с радостью порекомендую этот сервисный центр своим друзьям и знакомым
Когда мой холодильник Гаггенау внезапно сломался, я немедленно обратился в официальный сервисный центр. Мастер приехал уже на следующий день. Он провел тщательную диагностику, благодаря чему быстро нашел причину поломки — неисправность в системе охлаждения. Ремонт был выполнен на месте с использованием оригинальных запчастей, что гарантирует дальнейшую бесперебойную работу устройства. Отличное сервисное обслуживание и профессиональный подход мастера оставили только положительные впечатления.
Gaggenau-Remonty.ru – [url=https://gaggenau-remonty.ru/]ремонт кофемашины gaggenau[/url]
online blackjack for real money in kenya [url=casinoonline.co.ke]casinoonline.co.ke[/url] .
Моя стиральная машина Bosch начала издавать необычный шум во время работы. Обратившись в надежный сервис, я получил выгодное предложение по ремонту с использованием оригинальных запчастей. Мастера приехали в удобное для меня время, провели качественный ремонт по доступной цене, благодаря чему машина была восстановлена до идеального состояния. Эффективность и экономичность услуги сделали этот опыт исключительно положительным.
Бош-Ремонт.рф – [url=https://xn—-9sbn2afcdnw7c.xn--p1ai/]бош сервисный центр[/url]
Современные посудомоечные машины Gaggenau — это сложные устройства, требующие квалифицированного подхода при ремонте. Если у вас возникли проблемы с посудомоечной машиной этой марки, специалисты сервисного центра готовы оперативно прийти на помощь. Они проведут полную диагностику аппарата, точно определят источник проблемы и заменят неисправные детали на оригинальные запчасти, что гарантирует длительность и надежность ремонта. После ремонта машина будет работать как новая, сохраняя все функции и качество мойки.
Gaggenau-Remonty.ru – [url=https://gaggenau-remonty.ru/]ремонт холодильника gaggenau[/url]
Когда мой холодильник Bosch внезапно перестал работать, на помощь пришли мастера из местного сервисного центра. Они оперативно приехали на дом и срочно провели диагностику. Неисправность была быстро устранена, благодаря профессионализму и опыту специалистов. Весь ремонт занял несколько часов, и мой холодильник снова функционировал, как новый, все это — без лишней тревоги и ненужных хлопот.
Бош-Ремонт.рф – [url=https://xn—-9sbn2afcdnw7c.xn--p1ai/]мастер по ремонту стиральных машин бош[/url]
Когда в моей посудомоечной машине Bosch возникли проблемы, я вызвал мастера на дом в Москве. Специалист прибыл в удобное для меня время, и вскоре после диагностики приступил к ремонту. Проблема оказалась в насосе, который был успешно заменен на оригинальный. Цена ремонта была весьма приемлемой, а также мне предоставили гарантию на год, что добавило дополнительного спокойствия.
Бош-Ремонт.рф – [url=https://xn—-9sbn2afcdnw7c.xn--p1ai/посудомоечных-машин]ремонт посудомоечных машин bosch на дому[/url]
Шалені знижки на тактичні кросівки
тактичні кросівки купити [url=https://vijskovikrosivkifvgh.kiev.ua/]https://vijskovikrosivkifvgh.kiev.ua/[/url] .
Для владельцев техники Gaggenau, официальный сервисный центр в Москве предоставляет полный спектр услуг по ремонту и обслуживанию. Посетив официальный сайт, можно легко заказать выезд мастера на дом, получить консультацию или ознакомиться с условиями гарантийного обслуживания. Сервис гарантирует использование оригинальных запчастей и предоставляет высококвалифицированных специалистов для решения любых проблем с вашей бытовой техникой.
Gaggenau-Remonty.ru – [url=https://gaggenau-remonty.ru/]сервис гаггенау в москве[/url]
Если вы ищете профессиональный сервис по восстановлению работоспособности холодильников известной марки в Москве, то вы обратились по правильному адресу. Наш сервисный центр специализируется на ремонте холодильного оборудования данной марки и имеет большой опыт в данной области. Мы гарантируем высокое качество работ, применение фирменных запасных частей и конкурентные тарифы. Свяжитесь с нами немедленно, и мы поможем вашему агрегату Whirlpool снова заработать на все 100%! Для получения дополнительной информации посетите [url=https://remont-holodilnikov-whirlpool.taplink.ws]ремонт холодильников whirlpool в москве[/url].
Если ваш духовой шкаф Gaggenau вышел из строя, не беспокойтесь. В Москве есть профессионалы, которые способны выполнить ремонт в течение всего двух дней. Сервисные центры, специализирующиеся на продукции Gaggenau, обеспечивают высокое качество ремонтных работ благодаря опытным мастерам и использованию оригинальных запчастей. Вам гарантируется быстрое и эффективное устранение любых неисправностей, возвращение вашего устройства в идеальное рабочее состояние с сохранением всех функций и характеристик.
Gaggenau-Remonty.ru – [url=https://gaggenau-remonty.ru/]ремонт гагенау в москве[/url]
Выбор сервиса Gaggenau в Москве обещает не только качественный ремонт вашей техники, но и дополнительные преимущества. Клиенты могут воспользоваться бесплатной диагностикой, что значительно упрощает процесс определения неисправности без начальных затрат. Кроме того, на все виды ремонтных работ распространяется гарантия, что дает дополнительную уверенность в надежности и долговечности выполненных ремонтных работ. Это идеальный вариант для тех, кто ценит профессионализм и стремится обеспечить долгосрочную работоспособность своей техники.
Gaggenau-Remonty.ru – [url=https://gaggenau-remonty.ru/posudomoechnyh-mashin]ремонт посудомоечных машин gaggenau[/url]
Los mejores casinos en linea de Peru para jugadores exigentes
mejor casino online peru [url=https://mejores-casinos-online-peru.com/]online casino games peru[/url] .
Доступные цены
– Кран-буксировщик для душа: плюсы и минусы
краны шаровые [url=https://krany-sharovye-nerzhaveyushie-msk.ru/]краны шаровые[/url] .
Як підібрати тактичний рюкзак для активного відпочинку
Екстремальні подорожі
тактичні рюкзак [url=https://ryukzakivijskovibpjgl.kiev.ua/]тактичні рюкзак[/url] .
бетера букмекерская контора [url=https://sport-bk.by]https://sport-bk.by[/url] .
Сравнение лучших букмекеров
букмекерская контора бетвиннер [url=https://bukmeker-bk.ru/]официальные букмекерские[/url] .
Если вам 18 лет и вам срочно нужны деньги, обратите внимание на МФО, предлагающие займы без отказа на карту. Такие организации ориентированы на молодежную аудиторию и предлагают простые условия получения финансовой помощи. Заявки рассматриваются быстро, благодаря автоматизированным системам, что позволяет получить необходимую сумму без длительного ожидания. Это отличная возможность для тех, кто только начинает свой путь во взрослую жизнь и ещё не успел наработать кредитную историю.
Фин-Мир [url=https://dzen.ru/a/Zg_bqxHnsk6e4u7i]займ на карту мфо[/url]
[url=https://arusak-attestats.ru/]https://arusak-attestats.ru/[/url] – Купить аттестат за 9 – ключ к твоему будущему. В нашем сервисе все вы можете без труда и оперативно приобрести аттестат, обязательный для последующего обучения или трудоустройства. Наши консультанты обеспечивают качество и конфиденциальность предоставления услуг. Приобретайте школьный сертификат в нашем сервисе и откройте другие возможности для того, чтобы своего профессионального роста и карьеры.