Verdens sykeste lidelser
«Tremannen» er ikke den eneste som har en gripende historie og lidelse.
(SIDE2): Ekstreme og sjeldne lidelser har gjort et knippe mennesker kjente opp gjennom tidene. Mange har fått hjelp etter mye medieomtale, og flere av personene sier de lever et normalt liv, på tross av sin besynderlige sykdom. Her er noen av de mest kjente og uvanlige lidelsene:
«ULVEMANNEN»: Fajardo Aceves Jesus Manuel fra Mexico er kjent som «Ulvemannen» på grunn av sin ekstreme hårvekst. Tilstanden er et resultat av «hiper tricosis», en sykdom som fører til overflod av hår. Fajardo Aceves Jesus Manuel mener han har arvet lidelsen etter sin oldemor. Foruten at han ser annerledes ut, hevder han at han lever et helt normalt liv.
«TREMANNEN»: Indonesiske Dede lider av en sykdom der store deler av kroppen hans er dekket av barkeliknende vorter. Hudspesialist tror det kan være en kombinasjon av viruset som forårsaker vorter og en genetisk sykdom som gjør at immunforsvaret hans svekkes. Les mer om historien til Dede her.
«HORNMANNEN»: Opp fra bakhodet til kinesiske Ma Zhong Nan stikker det opp et ti centimeter langt horn. Det begynte å vokse for om lag fem år siden, etter at han fikk et lite kutt mens han kjemmet håret. Les mer om hornmannen her.
TRICHOPHAGIA: Lidelsen er meget sjelden og innebærer at vedkommende spiser på sitt eget hår. Dette kan gi store hårballer i magesekken. Les mer hos Wikipedia.
En 18 år gammel kvinne som gjennom hele livet hadde hatt god helse opplevde plutselig ubehag. Etter fem måneder med magesmerter, kvalme, hevelser og vekttap oppsøkte hun eksperter. Da legene undersøkte kvinnens mage fant de utrolig nok en hårball på 4,5 kilo. Les saken her: Fant kjempehårball i kvinnemage
EKSTREM OVERVEKT: Meksikanske Manuel Uribe veide 556 kilo i september i fjor. På grunn av sin høye vekt hadde han ikke kunnet forlate sengen på fem år. I sommer var det første gang Uribe var utendørs på lang tid. Han kunne endelig forlate rommet sitt etter å ha fullført en streng diett. Les mer: Redd verdens feteste
MIKROSKOPISKE KRANIER :
Microcephaly er en nervelidelse hvor omkretsen av hodet er betydelig mye mindre enn normalt for en persons alder og kjønn. Lidelsen kan være medfødt eller utvikle seg i de første leveårene. Den kommer av ulike tilstander som medfører anormal vekst av hjernen eller fra syndromer som assosieres med kromosom abnormitet.
Zip og pip eller Jennie Lee og Elvira Snow som de egentlig het ble kalt for «The Snow Twins». De led av microcephaly og var som de fleste som lider av dette syndromet mentalt tilbakestående. De reiste sammen med World Circus Sideshow i første halvdel av 1900-tallet sammen med mange andre mennesker som hadde unormale lidelser, men hadde tilholdssted på Coney Island.
«ARMLESS WONDER»: Martha Morris ble født tidlig på 1900-tallet i Chicago. Hun manglet begge armene, og deler av beina. Kvinnen, som fikk navnet «Armless Wonder», var ikke i stand til å kle på seg selv eller gå. Hun kom seg rundt ved hjelp av at folk trillet henne i en rullestol. Hun ble kjent for å holde ulike utstillinger. Da demonstrerte Morris imponerende ferdigheter ved å skrive med tærne som om det skulle vært fullt utviklet fingre.
Histopathology Brain– Alzheimer disease
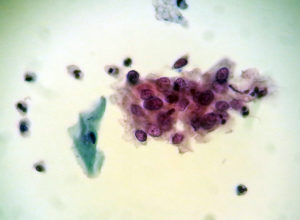 Gruppe med maligne celler.
Gruppe med maligne celler.
Sykdom. PDF.
Vitenskap, kultur eller begrep for sårbarhet, håp, mestring og mot?
Bjørn Hofmann, Høgskolen i Gjøvik, Universitetet i Oslo.
History of Morgellons disease: from delusion to definition. PDF.
(Er dette en “ekte” sykdom? Ingen patologer har vel sett dette? Har noen denne i Norge?)

Fra Folkehelseinstituttet:
Hva er sesonginfluensa?
Hvert år i perioden desember til april rammes befolkningen på den nordlige halvkule av influensaepidemier. Dette kalles sesonginfluensa. To typer av influensavirus, type A og B, er årsak til disse epidemiene. Fra år til år utvikler hvert av influensavirusene nye varianter som gjør vår immunitet mot de gamle variantene utdatert.
Influensapandemier i historien
Epidemier som ser ut til å ha vært influensa, er kjent langt tilbake i tid. Det er særlig de ekstraordinært store epidemiene som har blitt nedtegnet. Til sammen 19 slike svære influensaepidemier er kjent fra historien. Vi kaller dem influensapandemier. Bare influensa A kan gi en pandemi.
Den første pandemien ble beskrevet i 1580. Den største var Spanskesyken i 1918-19, som førte til 25-40 millioner dødsfall i verden. I Norge døde cirka 15 000 personer.
De tre siste større pandemiene var:
- Asiasyken i 1957
- Hong Kong-syken i 1968
- H1N1-pandemien i 2009/2010 (svineinfluensa)
Varianten av A(H1N1)-virus som førte til pandemien i 2009/2010, kalles ofte svineinfluensa siden de nærmeste slektningene vi kjenner er svineinfluensavirus. Varianten utgjør i dag ett av de vanlige sesonginfluensavirusene og har siden pandemien blitt kalt A(H1N1)pdm09.
Forekomst av sesonginfluensa
Andelen som blir syke med influensa varierer fra sesong til sesong, avhengig av immuniteten i befolkningen mot sirkulerende virus, vaksinasjonsdekning og de sykdomsfremkallende egenskaper til selve viruset. I år med større epidemier kan 10-30 prosent av den norske befolkningen bli smittet. I en gjennomsnittlig sesong vil 5-10% av alle voksne og 20-30% av alle barn bli smittet.
Tidspunktet for influensautbruddene er illustrert ved kurven under. Den viser at influensautbruddene vanligvis starter rundt jul/nyttår, og varer i omtrent 12 uker. Influensatoppen har de siste sesongene falt enten rundt nyttår eller omkring vinterferien. Dette kan imidlertid variere mellom sesonger.

Estimater basert på data fra perioden 1975-2004 tyder på at omtrent 900 dødsfall i året kan knyttes til influensa i Norge. Dette er imidlertid et gjennomsnitt og kan variere mye fra år til år. Det er alt overveiende eldre og personer med alvorlig bakenforliggende sykdom som dør som følge influensa.
Fakta om koronavirus (coronavirus)
Publisert Oppdatert
Det nye koronaviruset ble oppdaget i januar 2020 og ny kunnskap tilkommer stadig. Hva vet vi om utbruddet, viruset, sykdommen og risiko? Informasjon og råd vil bli justert og oppdatert i henhold til utbruddets utvikling og kunnskap om infeksjonen.

Om utbruddet
Et tidligere ukjent koronavirus har forårsaket et pågående utbrudd av luftveisinfeksjon. Utbruddet startet i storbyen Wuhan i Hubei-provinsen i Kina i desember 2019, og viruset ble identifisert av kinesiske helsemyndigheter 7. januar 2020.
Det nye viruset er et koronavirus (coronavirus) og har etterhvert fått navnet SARS-CoV-2 som kan gi luftveisinfeksjon hos mennesker. I mange tilfeller har det kun gitt mild sykdom, men det rapporteres også om tilfeller av alvorlig sykdom og dødsfall.
Utbruddet startet i millionbyen Wuhan i Hubei-provinsen sentralt i Kina. Smitten ble i begynnelsen knyttet til et stort matmarked i byen, Huanan Seafood Wholesale Market, med sjømat og ulike typer levende dyr, noe som kan tyde på at smitten opprinnelig kommer fra dyr.
Sykdommen smitter mellom mennesker og det er sett smitte til helsepersonell og andre nærkontakter. Det rapporteres stadig flere tilfeller fra andre kinesiske områder og andre land, også i Europa.
For oppdatert informasjon om utbruddet i Europa og hele verden:
- Det europeiske smittevernbyråets (ECDCs) nettsider om koronavirusutbruddet
- Verdens helseorganisasjons (WHOs) nettside om koronavirusutbruddet
30. januar 2020 erklærte WHO utbruddet som en “alvorlig hendelse av betydning for internasjonal folkehelse”. Erklæringen er ment å styrke Verdens helseorganisasjon (WHO) sin koordinering av innsatsen mot videre spredning av sykdom forårsaket av det nye viruset.
Om viruset
Det nye koronaviruset er et tidligere ukjent virus, men har visse genetiske likheter med SARS-viruset (Severe Acute Respiratory Syndrome) som også tilhører koronavirusfamilien. Viruset som forårsaker MERS (Middle East Respiratory Syndrome) er et annet koronavirus.
Man antar at smittemåten er dråpe- og kontaktsmitte som for andre koronavirus.
Koronavirusfamilien omfatter mange ulike virus som kan gi luftveisinfeksjon. Mange koronavirus gir bare forkjølelse, mens andre kan gi mer alvorlig sykdom og i noen tilfeller forårsake dødsfall. Koronavirus finnes også hos dyr. I sjeldne tilfeller kan slike koronavirus utvikle seg slik at de kan smitte fra dyr til mennesker og mellom mennesker, slik man så under SARS-epidemien i 2002. Da kom smitten antagelig fra flaggermus via sivettkatter og andre dyr. Dromedarer og kameler er smittekilde for sykdommen MERS som ble oppdaget i 2012.
From Biopsy to Diagnosis: How Pathologists Diagnose Cancer and Other Diseases.
This video provides a glimpse into the world of pathology by showing how pathologists and other laboratory professionals help to diagnose patients’ biopsies. This behind-the-scenes look into the University of Michigan Department of Pathology shows how tissue is prepared to be viewed under a microscope and what pathologists are looking for in order to determine a diagnosis.
Mammografiscreening er kontroversielt!
Mange mener det er viktig å finne små svulster så tidlig som mulig, og behandle disse for å få god overlevelse. Imidlertid oppstår det sannsynlig flere ganger naturlig små svulster som imidlertid forsvinner “naturlig” i kroppen, og også i bryst. En mulig årsak til dette kan bl.a.henge sammen med autofagi (selv-spising) som er sentralt i kroppens eget forsvarsystem for å bekjempe visse sykdommer. Se også Nobelprisen i medisin 2016 til japaneren Yoshinori Ohsumi.
Med mammografiscreening får man også en del uheldige sideefekter som bl. a. overbehandling og pasientfeil.
Mange mener det hjelper i forhold til overlevelse, selv om bildet trolig er noe anderledes:
Dr. László Tabár is synonymous with innovation and mammography.
László Tabár, M.D., F.A.C.R. (Hon) har nok bidrad sterkt til den svært positive tankegang om at man bør innføre mammografiscreening! Se hans hjemmesider som er “Big business“.
Statestics: Dessverre Mye error/ biasfeil!
“The average human being has 1 breast and 1 testicle”- (Des McHale)”.
Copyright© 2020, StatPearls Publishing LLC.
This book is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, a link is provided to the Creative Commons license, and any changes made are indicated.
In this Page
Similar articles in PubMed
- ReviewInsights into the Recent 2019 Novel Coronavirus (SARS-CoV-2) in Light of Past Human Coronavirus Outbreaks.[Pathogens. 2020]
- Novel coronavirus 2019-nCoV: prevalence, biological and clinical characteristics comparison with SARS-CoV and MERS-CoV.[Eur Rev Med Pharmacol Sci. 2020]
- ReviewSevere acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges.[Int J Antimicrob Agents. 2020]
- ReviewThe origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak – an update on the status.[Mil Med Res. 2020]
- Abelson Kinase Inhibitors Are Potent Inhibitors of Severe Acute Respiratory Syndrome Coronavirus and Middle East Respiratory Syndrome Coronavirus Fusion.[J Virol. 2016]
Recent Activity
Abstract
The severe acute respiratory syndrome (SARS)-coronavirus-2 (CoV-2) outbreak in Wuhan, China has now spread to many countries across the world including the UK with an increasing death toll. This will inevitably lead to an increase in the number of suspected coronavirus disease 2019 (COVID-19)-related deaths at autopsy. The Royal College of Pathologists has responded to this concern with the release of a briefing on autopsy practice relating to COVID-19. The following article is a summary and interpretation of these guidelines. It includes a description of hazard group 3 organisms, the category to which SARS-CoV-2 has been assigned, a brief description of what is currently known about the pathological and autopsy findings in COVID-19, a summary of the recommendations for conducting autopsies in suspected COVID-19 cases and the techniques for making the diagnosis at autopsy. It concludes by considering the clinicopathological correlation and notification of such cases.
Statistics from Altmetric.com
Introduction
The coronavirus family comprises several zoonotic viruses that cause serious human diseases including Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS).1In late 2019, a new coronavirus outbreak due to SARS-coronavirus-2 (SARS-CoV-2) began in Wuhan, China, which causes an illness in humans designated as coronavirus disease 2019 (COVID-19) by the WHO.2COVID-19 pathobiology remains incompletely understood and significant efforts are underway to study it worldwide.
SARS-CoV-2 continues to spread as the number of deaths continue to increase.3The mode of transmission of the virus is thought to be largely by inhalation of respiratory droplets although acquisition via the skin surface is another possibility.4Fever is usually the first symptom with subsequent development of respiratory symptoms.5 6Most patients with COVID-19 have a mild disease course; however, approximately 20% develop severe disease with high mortality rate and is associated with older age and immunosuppression.5 6
In February 2020, the Royal College of Pathologists (RCPath) released guidance on postmortem examinations for mortuary workers in suspected COVID-19 cases.4This article summarises those guidelines. It is based on our current understanding of COVID-19 in early March 2020 and may change as more information becomes available.
Hazard group categorisation of COVID-19
In the UK, pathogens are categorised according to their risk to humans by the Advisory Committee on Dangerous Pathogens (ACDP) within the Health and Safety Executive.7ACDP guidance is largely aimed towards staff in clinical and research-related microbiology laboratories, however given the potential risk to the health of mortuary staff, autopsy practice has been adapted to reflect the risk of transmission of infectious pathogens during and after the postmortem examination.8These hazard groups (HG1–4) are assigned according to the risk of human infection, the likelihood spread and access to treatment or prophylaxis (seetable 1). Control of substances hazardous to health (COSHH) defines four containment levels for working with these, which correspond directly to the organism’s hazard group (ie, containment level 3 should be used for hazard group 3 organisms).
Table 1
ACDP hazard group definitions: table adapted from ref7
The SARS-related and MERS-related coronaviruses are both considered HG3 pathogens, while most otherCoronavirinaeare HG2.7SARS-CoV-2 has recently been categorised as a HG3 organism.4 7 9Other viruses within HG3 include rabies, poliovirus, dengue virus, hepatitis virus B, C, D and E, and HIV 1 and 2 among others.4Certain HG3 organisms (eg, hepatitis B, C, D and E virus) may be used at containment conditions less than those outlined by COSHH for HG3 organisms; however, SARS-CoV-2 is not on this list.7Once appropriate precautions are in place, the risk to mortuary workers dealing with these HG3 organisms are slight, and a well-staffed mortuary with experienced personnel should be capable of handling cases with any of these HG3 pathogens.
Risk reduction during postmortem examination in HG3 organisms
In general, conducting an autopsy on a patient with a suspected HG3 organisms requires four areas of attention: risk assessment, understanding of the pathology that may be found, universal standard precautions and any standard operating procedures for specific HG3 pathogens. The effective utilisation of universal precautions mitigates against inaccurate or incomplete information used in the risk assessment on an individual case basis.
Mortuary workers performing autopsies have a duty outlined by COSHH to perform risk assessments in every postmortem examination for the safety of themselves and their colleagues. Before the time of the autopsy, this may include a review of the clinical history on the consent form or coroner request form, information attained from the patient’s doctors, laboratory records and hospital infection control and external examination findings.
It must be stressed at this point that if the death is considered to be due to a confirmed COVID-19 infection, an autopsy is unlikely to be necessary and a Medical Certificate of Cause of Death should be given. However, if the infection is involved in a forensic case, then legal duty impels the proper performance of a full appropriate autopsy. The issue of whether a person dies with or from COVID-19 infection may not always be straightforward, and ongoing epidemiology and available investigation data may shed light on this.
Pathological findings in COVID-19
Information regarding the pathological findings in COVID-19 is limited, although several case reports have been published in recent weeks.10 11
Clinical features: Public Health England (PHE) has outlined criteria to assess possibility of COVID-19 infection in patients.12These criteria are the same when the patient is deceased with the exception that the timelines given in the guidance refer to the time prior death or onset of relevant symptoms before death where known.
If it is considered that COVID-19 may have been related to death by these criteria, the choice of either to perform a full postmortem or an examination is limited only to retrieving the samples required to verify COVID-19 infection. This decision must be made according to the individual case and should include the requirements of the coroner or any pertinent individuals. A staged postmortem may also be considered. This involves taking only diagnostic samples initially and later considering or a more complete autopsy after the results of these diagnostic tests are available. This staged technique is recommended if possible.
Macroscopic features: the macroscopic features of COVID-19 are likely to be in the chest and may include pleurisy, pericarditis, lung consolidation and pulmonary oedema. Lung weight may be increased above normal. It should be noted a secondary infection may be superimposed on the viral infection that can lead to purulent inflammation more typical of bacterial infection.4
Microscopic findings:a recent article described the early histopathological features in COVID-19 in two patients who underwent surgical resections for lung adenocarcinoma but were later discovered to have had COVID-19 at the time of the operation.11The findings were non-specific and included oedema, pneumocyte hyperplasia, focal inflammation and multinucleated giant cell formation while no hyaline membranes were seen. Given that these patients were asymptomatic with respect to COVID-19 at the time of the operation, these are likely to reflect only early changes of acute lung injury in the infection.11In another case, a 50-year-old man died from severe COVID-19 infection and more marked histopathological findings were noted.10Samples were taken by postmortem biopsy, and a description of the gross postmortem findings is not given, although multiple ground glass opacities were noted on chest X-ray. The microscopic findings included diffuse alveolar damage with exudates.10The inflammation was predominantly lymphocytic, and multinucleated giant cells were seen alongside large atypical pneumocytes, although no definitive viral inclusions were noted. Microvesicular steatosis with mild inflammation was noted in the liver, although it was unclear whether this was related to the virus or iatrogenic. The features are very similar to those seen in SARS and MERS-coronavirus infections.13 14
Mortuary factors
Adequate ventilation is need where HG3 autopsies are being performed with enough separation from the rest of the mortuary. Either whole room ventilation or down-drafts at the work stations are acceptable.15Any electric bone saws used should have a vacuum that isolates aerosolised particles. It is preferable to have an isolated high-risk facility for performing HG3 autopsies, although this is not compulsory.8All essential equipment should be brought at the start of the postmortem examination (eg, sample receptacles, culture bottles and so on) to eliminate the need to leave and re-enter the workspace. Further information may be found in the appropriate National Health Service guidance documents.16
The team available for these high-risk, postmortem examinations best includes the pathologist, the anatomic pathology technician (APT) and a third circulator. The presence of a circulator is beneficial but not essential.8Suitably experienced autopsy pathology trainees (as assessed by senior staff) may be involved in HG3 autopsies with adequate supervision. No specific infection risk to pregnant trainees has been identified; however, they may decide not to undertake autopsy work, and this should be discussed with the deanery.
Performance of the autopsy in HG3 infections
Several techniques undertaken at autopsy can reduce the risks encountered by HG3 infections. Personnel must be adequately trained. For APTs, this exposure should be in line with their curriculum and standardised training under the Royal Society for Public Health. For pathology trainees, this is at the discretion of senior staff. Sharps injuries can be reduced by minimising sharps in the workspace, using round-ended scissors, blunt-ended PM40 blades and having only a single operator working in the body cavity at a time.4Fresh organs should be sliced while being stabilised with a sponge on a solid surface. Needles should be placed in sharps bins and never resheathed.
Personal protective equipment (PPE) is vital. Certain PPE is universally used in postmortem examinations (box 1); however, the typical surgical masks are not considered sufficient protection. Valved fold flat and moulded protection masks are over 95% effective and are suitable for use in anticipated COVID-19 cases.4Whole-body suits with individual respirators seem to provide almost complete protection, although these are impractical and not necessary.15
Box 1
List of minimum personal protective equipment (PPE) in suspected coronavirus disease 2019 (COVID-19) cases
Universal PPE
-
Surgical scrubs.
-
Scrub hat.
-
Clear face visor.
-
Waterproof gown to cover whole body and forearms (typically a surgical gown).
-
Plastic apron.
-
Rubber boots with metal toecaps.
-
Protective kevlar or neoprene cut-resistant under-gloves.
-
Single use disposable non-latex gloves.
Additional protective equipment in suspected COVID-19 cases
-
Fold flat and moulded protection mask or whole-body suit.
A staged postmortem is recommended by RCPath.4Where a more detailed examination is necessary, a limited postmortem may be considered. In widespread infections, a minimally invasive postmortem examination can be performed to provide necessary fluid and tissue samples;17however, regional or localised infections may be difficult to identify with this technique alone, although they may be improved by combination with postmortem imaging.
Postmortem CT (PMCT) examination:histopathologists with local agreements regarding access to scanning facilities may contemplate PMCT to demonstrate significant pulmonary findings or identify causes of death in a case with incidental SARS-CoV-2 infection. A recently multicentre study (n=101) showed ground-glass lung opacities in 86% of cases, or mixed ground-glass and consolidation in 64% of patients examined.18Vascular enlargement and traction bronchiectasis are also described (71% and 53%, respectively). The changes were more likely to be bilateral, peripherally distributed and involving the lower lobes. The decision to undertake native PMCT or ventilated PMCT would be for the reporting radiologist following consultation: use of an existing clinical airway, or creation of a tracheostomy postmortem, would provide access for ventilation and potentially lower respiratory tract sampling.
Diagnosis of COVID-19
The samples required to diagnose COVID-19 at autopsy are the same as those required during life. They include a 5 mL sample of plain blood (no additive) for serology, upper aerodigestive tract swabs (nose and throat) and lower respiratory tract samples (bronchoalveolar lavage or sputum).19For full and up-to-date details on how to submit these samples, one should follow the links in the references.19It should be noted that the submission of each sample requires a specific E28 form. The specific advice from PHE is that one swab should be used for the upper aerodigestive tract and another swab for the lower respiratory tract.4
RCPath recommends that standard samples, such as respiratory tract swabs and tissue samples, should also be sent to local microbiology departments simultaneously to detect pathogens in the differential diagnosis. Where possible, a complete set of tissue histology is also recommended along with other specific investigations as outlined on a case-by-case basis. Standard formalin-fixation inactivates known coronaviruses and SARS-CoV-2 is believed to be similarly affected.20
Ready-to-use swabs can collect respiratory tract samples. Blood, urine and cerebrospinal fluid samples, where deemed appropriate, should be taken prior to opening the body cavity and under as sterile conditions as possible to reduce contamination. This may be achieved using alcohol-containing disinfectant to clean the skin. Blood cultures should be taken preferentially from the subclavian vein, jugular vein or left ventricle to reduce contamination from the bowel.
Conclusion
We have outlined the hazard group categorisation, pathological features and approach to the autopsy in suspected COVID-19 cases to aid mortuary workers. If on clinicopathological correlation, COVID-19 is deemed to be primary cause of death, then it should be given at the last line of part 1 in the Office of National Statistics format. If there is another primary cause of death and COVID-19 is considered to be contributory, then it can be put in part 2. Finally, it is worthwhile to notify PHE of any cases where COVID-19 is confirmed, although this should also be done by the laboratory that made the diagnosis.
References
Footnotes
-
Handling editorTahir S Pillay.
-
Correction noticeThis article has been corrected since it appeared Online First. The abstract has been replaced, and a new sentence and reference added “Standard formalin-fixation inactivates known coronaviruses and SARS-CoV-2 is believed to be similarly affected[20]”. Several minor text changes have been made throughout.
-
ContributorsBH designed and planned the manuscript performed the literature review, wrote the paper and collaborated with other authors in review. SBL, EY and BS reviewed manuscript drafts and contributed several paragraphs. MO designed and planned the manuscript and was involved in review of drafts along with collaborating between coauthors for multidisciplinary input.
-
FundingThe authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
-
Competing interestsNone declared.
-
Patient consent for publicationNot required.
-
Provenance and peer reviewCommissioned; internally peer reviewed.
Request Permissions
If you wish to reuse any or all of this article please use the link below which will take you to the Copyright Clearance Center’s RightsLink service. You will be able to get a quick price and instant permission to reuse the content in many different ways.